TO THE EDITOR:
The classic Ph-negative myeloproliferative neoplasms (MPNs) are a group of clonal hematopoietic disorders, including polycythemia vera (PV), essential thrombocythemia (ET), and myelofibrosis (MF), caused by a dysregulated JAK/STAT signal transduction because of acquired somatic mutations of JAK2, CALR, or MPL genes.
Chronic inflammation may predispose to MPN development; indeed, higher risk of MPN has been reported for patients with a history of autoimmune diseases, including immune thrombocytopenia, Crohn disease, giant cell arteritis, and Reiter syndrome.1
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection displays extreme interindividual clinical variability, ranging from asymptomatic infection to life-threatening coronavirus disease (COVID-19).2,3 Age is a major risk factor for severe disease, with risk of death doubling every 5 years from childhood onward. The global infection fatality rate is ∼1% but 0.001% at age 5 years and 10% at age 85 years (a 10.000-fold increase). Male sex and medical comorbidities have minor impact.4-6 It was shown that 1% to 5% of patients with life-threatening COVID-19 pneumonia have monogenic inborn errors of TLR3- or TLR7-dependent type I interferon (IFN-I) immunity.7,8 At least 15% of patients with life-threatening COVID-19 pneumonia have neutralizing autoantibodies (AAbs) against IFN-I, which precede SARS-CoV-2 infection.9,10
In patients with MPN, COVID-19 is associated with a mortality of 33%.11 Patients with MF had the highest mortality (48%)12 whereas patients with ET had the greatest risk of venous thromboembolism (16.7%).13
In this study, we searched for AAbs against IFN-I in a cohort of patients with MPN to evaluate their prevalence in the MPN population and investigate clinical correlations, including COVID-19 severity.
This study was approved by the Ethics Committee of Fondazione IRCCS Policlinico San Matteo, Pavia, Italy. The procedures followed were in accordance with the Helsinki Declaration; samples were obtained with patients’ written informed consent.
Plasma samples from 219 consecutively referred patients with MPN were prospectively collected between November 2020 and June 2021 at the Department of Hematology, Fondazione IRCCS Policlinico San Matteo, Pavia. Diagnosis of MPN was made in accordance with 2016 World Health Organization criteria.14 Samples were frozen at −30°C after collection. AAbs against IFN-I subtypes (IFN-α, -β, and -ω) were measured using the enzyme-linked immunosorbent assay and a luciferase-based neutralization assay, as previously reported.9,10 Mutational status was evaluated on DNA extracted from peripheral blood polymorphonuclear cells. Granulocyte JAK2 V617F mutation, CALR exon 9 mutations, and MPL exon 10 mutations were assessed as previously reported.15-17 Statistical analyses were performed as reported in supplemental Materials.
We enrolled 219 patients with MPN, including 101 with ET, 76 with PV, 36 with MF, and 6 with MPN unclassifiable (MPN-u), as reported in Table 1. The median time between diagnosis and sampling was 7.7 years (interquartile range, 3.4-13.4). AAbs neutralizing IFN-α and/or IFN-ω were detected in 29 patients (13.2%; 95% CI, 9.1% to 18.5%), 4 of whom had AAbs neutralizing high concentration (10 ng/mL) and 25 of whom had AAbs neutralizing 100-fold lower concentrations (100 pg/mL) of these cytokines. Prevalence of AAbs was ∼6 times higher in patients with MPN than that observed in the general population10 (age-adjusted PR = 6.2; range, 4.3-8.8; P < .001). One may speculate that such a high prevalence in MPN could be a consequence of IFN-I chronically elevated levels, as happens in systemic lupus erythematosus.18 High levels of IFN-I were, indeed, previously reported in patients with MPN compared with healthy controls.19,20
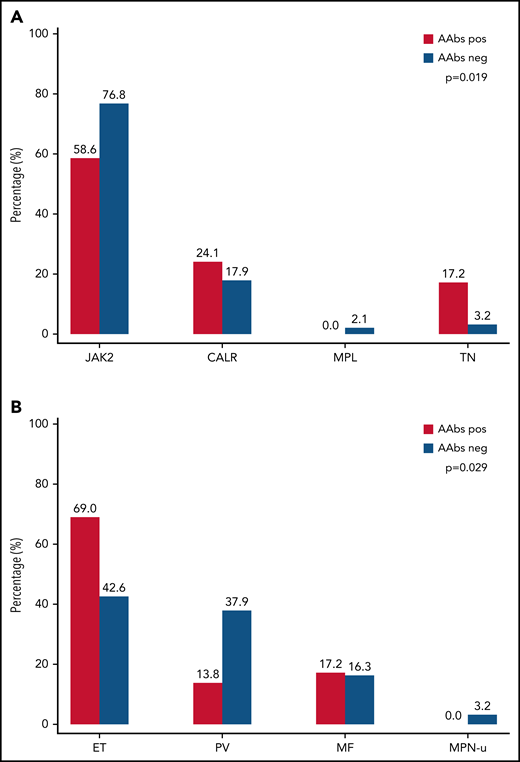
Next, we explored whether the high prevalence of AAbs in MPN correlated with specific clinical or molecular features. We observed a significant difference in terms of distribution of MPN diagnosis (P = .029) and driver mutations (P = .019) between patients with and without AAbs (Figure 1).
Prevalence of AAbs against type I IFN in the MPN cohort according to MPN driver mutation and MPN subtype. (A) AAbs were present in 17 of 163 (58.6%) patients with JAK2 mutation, 7 of 41 (24.1%) patients with CALR mutation, 0 of 4 (0%) patients with MPL mutation, and 5 of 11 (17.2%) patients who were triple negative. Significant difference was observed in terms of driver mutations (P = .019). Comparing mutational subgroups by pairs, the only significant difference was observed between the percentage of AAbs in patients who were triple negative and that observed in patients with JAK2 mutation (45.5% vs 10.4%; P = .036). (B) AAbs were present in 20 of 101 (69%) patients with essential thrombocythemia (ET), 4 of 76 (13.8%) patients with polycythemia vera (PV), 5 of 36 (17.2%) patients with primary myelofibrosis (MF), and 0 of 6 (0%) patients with myeloproliferative unclassifiable (MPN-u). A significant difference was observed in terms of distribution of MPN diagnosis (P = .029).
Prevalence of AAbs against type I IFN in the MPN cohort according to MPN driver mutation and MPN subtype. (A) AAbs were present in 17 of 163 (58.6%) patients with JAK2 mutation, 7 of 41 (24.1%) patients with CALR mutation, 0 of 4 (0%) patients with MPL mutation, and 5 of 11 (17.2%) patients who were triple negative. Significant difference was observed in terms of driver mutations (P = .019). Comparing mutational subgroups by pairs, the only significant difference was observed between the percentage of AAbs in patients who were triple negative and that observed in patients with JAK2 mutation (45.5% vs 10.4%; P = .036). (B) AAbs were present in 20 of 101 (69%) patients with essential thrombocythemia (ET), 4 of 76 (13.8%) patients with polycythemia vera (PV), 5 of 36 (17.2%) patients with primary myelofibrosis (MF), and 0 of 6 (0%) patients with myeloproliferative unclassifiable (MPN-u). A significant difference was observed in terms of distribution of MPN diagnosis (P = .029).
First, AAbs were observed more frequently in patients without JAK2 mutation compared with those with JAK2 V617F (P = .042). Recent evidence suggests that JAK2 V617F might enhance PD-L1 expression.21 Moreover, research has demonstrated that the PD-1/PD-L1 axis plays a crucial role in regulatory T cell (Treg) development and function by promoting the immunosuppressive ability of Tregs and converting naïve CD4+ T cells to Treg.22 Thus, we can speculate that patients with JAK2 mutation have an increased PD-L1 expression and, consequently, an increased Treg immunosuppressive function that could, in turn, exert a protective role against AAb production.
AAbs were observed more frequently in ET compared with other MPN (P = .009). The higher prevalence observed in patients with ET might be related to genetic background because ∼50% of patients with ET have a JAK2-unmutated genotype. The proportion of females was not significantly different between patients with ET with and without AAbs, thus not supporting the known association between the female sex, which is predominant in ET, and autoimmunity.23
Finally, we found a higher proportion of patients treated with the JAK inhibitor ruxolitinib in the group with AAbs. Using a univariable logistic regression, the probability of having AAbs to IFN-I is roughly 3 times higher in patients treated with ruxolitinib compared with all other patients (odds ratio [OR] = 3.0; 95% CI, 1.1-8.6; P = .036). The association was even higher after adjustment for diagnosis and driver mutation (OR = 4.8; 95% CI, 1.4-15.8; P = .011). Given the well-known role of JAK/STAT pathway in immune system function,24 we might speculate that the immunomodulatory effect of ruxolitinib may influence self-tolerance through a reduction of T-regulatory lymphocytes.25
Overall, 29 patients (13%) developed SARS-CoV-2 infection, 8 of whom (28%) required hospitalization due to severe or critical COVID-19 pneumonia. This proportion is higher than that in the general population for the same age category. AAbs against IFN-I were detected in 4 of 29 patients with SARS-CoV-2 infection. Despite a comparable prevalence of SARS-CoV-2 infection, we reported a rate of hospitalization due to severe COVID-19 that was almost twice that in patients with AAbs (2 of 4 patients, 50%) compared with those without (6 of 25 patients, 24%) and much higher than in the general population for the same age category. However, this difference did not reach a statistical significance, most likely because of the small number of cases. According to previous data,10 the age- and sex-adjusted risk of having critical COVID-19 was higher, even if not significant, in patients with AAbs to IFN-α (OR = 100 pg/mL only: 10.4; range, 0.3-341) than in patients with AAbs to IFN-ω (OR = 100 pg/mL only: 2.4; range, 0.2-27.1).
In conclusion, we report a higher prevalence of AAbs to IFN-I in patients with MPN compared with the general population, an association with JAK2-unmutated genotype, with ET diagnosis, and a potential higher rate of severe COVID-19. Due to the small number of patients, the lack of mechanistic studies, and the lack of validation, these results are preliminary and need to be confirmed in a larger cohort of patients with MPN. AAb detection may enable early identification of patients who are likely to develop life-threatening COVID-19, which is clinically valuable if we consider the high mortality of SARS-CoV-2 infection in hematological malignancies and the chance of early intervention with monoclonal antibodies.
Acknowledgments
Studies performed at the Department of Hematology, Fondazione IRCCS Policlinico San Matteo and Department of Molecular Medicine, University of Pavia were supported by grants from the Italian Ministry of Health for young researchers (GR-2016-02361272) (E.R.) and by Associazione Italiana per la Ricerca sul Cancro (AIRC: IG 2021 ID 25703) through the project “Actionable targets in clonal progression and systemic spreading of myeloid neoplasms,” (IG 2021 ID 25703) (E.R.) MYNERVA project (Project Code: 21267) (E.R. and L.M.). This work was supported by The European Union’s Horizon 2020 research and innovation program (ATAC, 101003650), the Center for Innovative Medicine at Karolinska Institutet, the Swedish Research Council, the Knut and Alice Wallenberg Foundation (KAW). The Laboratory of Human Genetics of Infectious Diseases is supported by the Howard Hughes Medical Institute, the Rockefeller University, the St. Giles Foundation, the National Institute of Allergy and Infectious Diseases (NIAID) (grants R01AI088364 and R01AI163029), the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health Clinical and Translational Science Award (CTSA) program (UL1TR001866), a Fast Grant from Emergent Ventures, Mercatus Center at George Mason University, the Fisher Center for Alzheimer’s Research Foundation, the Meyer Foundation, the JPB Foundation, the French National Research Agency (ANR) under the “Investments for the Future” program (ANR-10-IAHU-01), ANR grants (ANR-14-CE14-0008-01, ANR-18-CE15-0020-02, ANR-20-CE93-003, ANR-20-CO11-000,1 and ANR-21-COVR-0039), the Integrative Biology of Emerging Infectious Diseases Laboratory of Excellence (ANR-10-LABX-62-IBEID), the French Foundation for Medical Research (FRM) (EQU201903007798), the FRM and ANR GENCOVID project (ANR-20-COVI-0003), ANRS Nord-Sud (ANRS-COV05), the European Union’s Horizon 2020 Research and Innovation Program (grant agreement no. 824110) (EASI-Genomics), the Square Foundation, Grandir-Fonds de solidarité pour l’enfance, the SCOR Corporate Foundation for Science, Fondation du Souffle, Institut National de la Santé et de la Recherche Médicale (INSERM), REACTing-INSERM, and the University of Paris. P.B. received support from the French Foundation for Medical Research (FRM, EA20170638020) and was supported by the PhD program of the Imagine Institute (with the support of the Fondation Bettencourt-Schueller).
Authorship
Contribution: O.B., P.B., J.-L.C., A.B., and E.R. conceived this study, collected and analyzed data, and wrote the manuscript; P.B., J.R., and A.G. evaluated the presence of AAbs against type I IFNs; D.V., I.C.C., and C.T. collected clinical data; D.P. and S.C. conducted molecular investigations; V.V.F. performed statistical analyses; and E.S., L.M., and L.A. finalized the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Elisa Rumi, Department of Molecular Medicine, University of Pavia, Pavia, Italy; e-mail: elisarumi@hotmail.com.
The online version of this article contains a data supplement.
REFERENCES
Author notes
O.B. and P.B. contributed equally to this study.
A.B. and E.R. contributed equally to this study.