Background: Over 300 somatic molecular variants in hematologic diseases are either specified as diagnostic criteria in the World Health Organization (WHO) Classification of Tumors of Hematopoietic and Lymphoid Tissues, recognized as potentially actionable biomarkers in the National Comprehensive Cancer Network (NCCN) compendia, or supported by published well-powered clinical studies. Moreover, new molecular alterations with potential clinical implications in hematologic disease are continuously emerging in the scientific literature. These have critical use for a wide spectrum of clinicians, including hematopathologists who diagnose patient-specific hematologic malignancies, heme-oncologists who direct patient care, and clinical trial nurses who assist patients in finding appropriate clinical trials. Importantly, the utility of this information critically depends on the clinician's ability to interpret the significance of variants in a point-of-care setting. Therefore, there is an urgent and unmet need for a clinical decision support system that 1) distills the clinical implications associated with molecular alterations into a standardized and easily interpretable format and 2) democratizes access of this information to all members of the heme-oncology community.
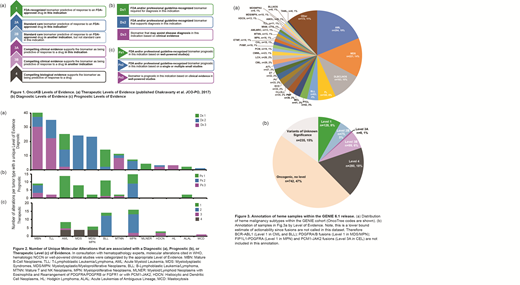
Methods: OncoKB is an established expert-guided precision oncology knowledge base that annotates the oncogenic effect and therapeutic implications of somatic molecular alterations (Chakravarty, D. et al., JCOPO, 2017). Previously, OncoKB was focused primarily on solid tumor mutation annotation. Recently, we expanded OncoKB to include alterations in hematologic malignancies. The heme-specific annotation efforts were guided by heme-oncology and hematopathology physician scientists at Memorial Sloan Kettering (MSK). Supplementing the previously published therapeutic levels of evidence (Fig. 1a), we further added level of evidence systems for diagnostic and prognostic implications (Fig. 1b, c). These three sets of evidence levels are consistent with the criteria set forth by the joint consensus of the ASCO/CAP/AMP guidelines (Li, MM. et al., J Mol Diagn, 2017). We assigned the newly curated heme-specific molecular alterations with diagnostic, prognostic or therapeutic levels of evidence, when applicable. Finally, we annotated and analyzed 1569 hematologic tumor samples from the AACR Project GENIE (release 6.1) with these levels of evidence.
Results: In addition to alterations with both solid and heme clinical implications already curated in OncoKB, we annotated 288 unique heme-specific mutations, fusions, and copy number alterations in 156 newly curated cancer-associated genes. Based on MSK-expert consensus, the WHO and NCCN guidelines, and the scientific literature, we identified a total of 192 alterations with unique diagnostic levels of evidence, 65 alterations with unique prognostic levels of evidence and 55 alterations with unique therapeutic levels of evidence across 13 major hematologic tumor types (Fig. 2).
To test the utility of OncoKB, we annotated all genomic events in 1569 heme cancer samples in 89 hematologic malignancies in the AACR GENIE cohort (V6.1) (Fig. 3a). Thirty-eight percent of samples harbored at least one potentially actionable alteration, and 8% were predictive of clinical benefit from an FDA-approved drug (Fig. 3b).
Conclusions: OncoKB heme data is publicly available both through the web resource http://oncokb.org and through incorporation into the cBioPortal for Cancer Genomics. Heme-specific molecular alterations are used to make an accurate diagnosis, inform prognosis, optimize the use of stem cell transplant, and to link patients with the optimal mechanism-based therapies in the clinical trial setting and in routine clinical practice. This is the first study to annotate and analyze actionability of heme samples. In this proof-of-principle study, we demonstrate the ability to annotate clinical samples with their diagnostic, prognostic and therapeutic implications in a point-of-care setting.
Roshal:Celgene: Other: Provision of Services; Auron Therapeutics: Equity Ownership, Other: Provision of services; Physicians' Education Resource: Other: Provision of services. Ho:Invivoscribe, Inc.: Honoraria. Knorr:Fate Therapeutics: Patents & Royalties. LaFave:Epizyme: Patents & Royalties. Arcila:Invivoscribe, Inc.: Consultancy, Honoraria. Berger:Roche: Consultancy. Solit:Pfizer: Consultancy; Lilly Oncology: Honoraria; Vivideon Therapeutics: Consultancy; Loxo Oncology: Consultancy, Equity Ownership; Illumina: Consultancy. Dogan:Celgene: Consultancy; Seattle Genetics: Consultancy; Corvus Pharmaceuticals: Consultancy; Roche: Consultancy, Research Funding; Novartis: Consultancy; Takeda: Consultancy. Levine:C4 Therapeutics: Membership on an entity's Board of Directors or advisory committees; Qiagen: Membership on an entity's Board of Directors or advisory committees; Isoplexis: Membership on an entity's Board of Directors or advisory committees; Loxo: Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Research Funding; Novartis: Consultancy; Gilead: Consultancy; Imago Biosciences: Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria; Lilly: Honoraria; Prelude Therapeutics: Research Funding; Roche: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.