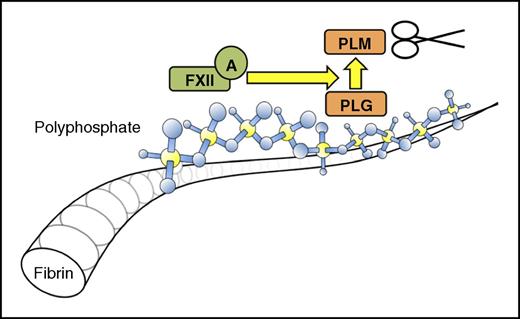
In this issue of Blood, Mitchell et al show how platelet polyphosphate (polyP) amplifies the role of factor XII (FXII) as a plasminogen activator. This negatively charged polymer acts as a “molecular adaptor” that concentrates FXII and plasminogen on the surface of fibrin polymers, eventually resulting in plasmin formation (see figure).1
Intrinsic fibrinolysis when polyP associates with fibrin. FXII, factor XII; PLG, plasminogen; PLM, plasmin.
Intrinsic fibrinolysis when polyP associates with fibrin. FXII, factor XII; PLG, plasminogen; PLM, plasmin.
Since the discovery of polyP in platelets,2 its contribution to thrombosis using in vivo models,3 and its validation as an antithrombotic target,4 scientific attention has been largely focused on its prothrombotic features. Polyphosphate among others accelerates activation of FXI and FV, inhibits anticoagulant pathways,5 and triggers FXII-dependent coagulation.3 Besides these procoagulant features, polyP interferes with fibrinolysis: when it is incorporated into the fibrin meshwork, the resulting fibrin structure is resistant to breakdown by plasmin and subsequently has a reduced capacity to attract tissue-type plasminogen activator and plasminogen for further lysis.6 These are the indirect prothrombotic features of polyP.
In their article, Mitchell et al discover a new antithrombotic feature of polyP: the capacity to stimulate “intrinsic fibrinolysis.” It was previously reported that FXII(a) can directly bind to fibrin.7 This binding does not trigger FXII activation. However, the story changes considerably when platelet polyP is present.
The authors first provide confirmatory evidence for the capacity of short platelet polyP to bind and activate FXII. This property of platelet polyP has been a subject of lively debate. It is particularly interesting that polyP protects FXIIa against “self destruction.” Without polyP, soluble FXIIa cleaves itself into smaller fragments and after ∼2 hours has lost all enzymatic capacity. In contrast, when FXIIa is bound to polyP, FXIIa degradation is reduced and enzymatic activity is preserved. This indicates that platelet polyP binding extends the functional life span of FXIIa and suggests another layer of regulation to keep the activity of FXIIa localized.
In further experiments, the authors found that plasminogen can also bind to platelet polyP. It will be of interest to define the molecular details of this interaction in future studies. Because polyP is known to interfere effectively with plasminogen binding to fibrin,6 plasminogen should have little choice but to bind to polyP when both fibrin and polyP are present at the same time. As a result, a ternary complex forms on the surface of fibrin polymers (FXII, plasminogen, and polyP) to initiate intrinsic fibrinolysis. In clot lysis studies, intrinsic fibrinolytic activity is sufficiently stimulated by platelet polyP to overcome its previously reported antifibrinolytic properties.6 The additional presence of tissue plasminogen activator only modestly further accelerates fibrinolysis.
In comparison with RNA or collagen (both can bind and activate FXII), platelet polyP is particularly effective as a mediator of intrinsic fibrinolysis. This indicates that stimulation of intrinsic fibrinolysis is not a feature that is generally shared by negatively charged polymers. It is imaginable that the capacity of polyP to bind plasminogen makes an important difference.
Subsequently, flow cytometry studies elegantly demonstrate that labeled FXII binds to washed platelets in suspension during their activation. Simultaneously, 4′,6-diamidino-2-phenylindole (DAPI) fluorescence of platelet increases. This nuclear stain is useful for microscopic visualization of intracellular platelet polyP2 and is applied in the current study as a surrogate marker for polyP release. In order for FXII to bind to platelets, fibrin formation is needed: a fibrin polymerization inhibitor (Gly-Pro-Arg-Pro) strongly inhibits FXII binding to activated platelets and simultaneously reduces DAPI fluorescence. This suggests a mechanism in which platelets first sequester polymerizing fibrin on their surface, which in turn recruits secreted soluble polyP polymers, as well as FXII(a). Intriguingly, all of these interactions can take place in the absence of plasma, suggesting that platelets contain sufficient amounts of clotting factors (or fibrin monomer) to independently retain polyP and FXIIa on their surface. Finally, microscopy studies reveal how platelet polyP associates with fibrin polymers and colocalizes with FXII. It will be interesting to unravel the sequence of binding events. Is polyP binding required for effective recruitment and activation of FXII on fibrin? Furthermore, it will be of interest to investigate how plasmin(ogen) is distributed and how intrinsic plasmin activity modifies thrombus morphology. Finally, it will be important to uncover the overall contribution of platelet polyP in physiological hemostasis and its influence on the timing and speed of thrombus breakdown.
The profibrinolytic properties of FXIIa have been the topic of recurrent investigation for decades and have been used to explain the protective role of FXII in various clinical studies on thrombotic and cardiovascular disease. The contribution of this pathway may not be as minor as one might initially expect: a study in healthy human volunteers revealed that intrinsic plasminogen activation accounts for ∼23% of total fibrinolytic activity after administration of endothelial cell agonists.8 It is more than likely that the intrinsic fibrinolytic activity in this study was generated in the absence of platelet polyP or fibrin. Taken together, the novel findings of Mitchell et al show that platelet polyP amplifies intrinsic fibrinolysis, and suggest that this pathway may play a significant role in fibrinolysis of platelet-rich thrombi.
Conflict-of-interest disclosure: The author declares no competing financial interests.