Abstract
Polyploidization is a part of the normal developmental process leading to platelet production during megakaryocyte (MK) differentiation. Ploidization is mainly involved in cell enlargement, but it is not clear whether gene expression is modified during MK ploidization. In this study, human MKs were grown from CD34+ cells in the presence of thrombopoietin and sorted according to their ploidy level. A pangenomic microarray technique was applied to compare gene expression in 2N-, 4N-, 8N-, and 16N-sorted MKs. Using hierarchical clustering, we demonstrated that 2N and 4N MKs or 8N and 16N MKs are 2 different close populations with 105 discriminating genes. In the second approach, we determined the profile of genes that were continuously down- and up-regulated during polyploidization. Among the 100 down-regulated genes, 24 corresponded to genes involved in DNA replication and repair. The great majority of up-regulated genes corresponded to genes directly involved in platelet functions, such as genes encoding specific platelet glycoproteins and α-granule proteins, actin and microtubule cytoskeleton, factors involved in signaling, and transport proteins. Together, these results suggest that MK polyploidization per se does not regulate gene expression but is intrinsically included in the differentiation process.
Introduction
Most cells are diploid in mammals, but some rare cells are polyploid. The vast majority of polyploid cells are formed by fusion, as for the osteoclast or myoblast. However, other mechanisms leading to an increase in DNA content occur in a few cell types, with the best characterized being polyploidization of hepatocytes and megakaryocytes (MKs).1 One characteristic of MK polyploidization is its occurrence as a normal process during differentiation and in the absence of any hematopoietic stress. Indeed, in the adult, nearly all mature MKs in the bone marrow are polyploid.
Committed MK progenitors initially proliferate by a mitotic process but later during MK differentiation, after the synthesis of platelet-specific protein has begun, cells switch to an endomitotic process to increase their ploidy level.2,3 During normal adult human or murine megakaryopoiesis, polyploidization usually stops at 16N. However, induction of a thrombocytopenia may increase ploidy to a 32N or a 64N level through thrombopoietin (TPO) stimulation.1 After the end of DNA replication, MKs undergo terminal differentiation and markedly increase protein synthesis that leads to granule biogenesis and the development of demarcation membranes required for platelet biogenesis.4-6 Thus, MK differentiation takes place in 3 successive steps—proliferation, polyploidization, and cytoplasm maturation—that finally lead to proplatelet formation and platelet release.
However, there is evidence that polyploidization is not an obligatory phenomenon for the formation of platelets: MKs in the fetus and neonate have a low ploidy level, and some 2N or 4N MKs in the neonate, called micromegakaryocytes (micro-MKs), are able to shed platelets.7,8 Similarly, in myeloproliferative or myelodysplastic syndromes, micro-MKs shedding platelets have been observed.9 The ploidy levels of MK produced in culture from human CD34+ cells are much lower than in the marrow; however, these MKs have the capacity to generate platelets.10
Therefore, it is assumed that the main role of MK polyploidization is to facilitate the increase in cell size without losing the energy required for caryokinesis and cytokinesis. As platelets are MK cytoplasmic fragments, polyploidization increases the overall MK mass, resulting in an increase in platelet formation.
However, polyploidization may have other functions. It has been suggested that MKs from different ploidy levels produce platelets with different functions. In particular, platelets originating from high-ploidy MKs are thought to be more easily activated than platelets generated from MKs with a lower ploidy.11 Therefore, polyploidy may regulate gene expression, MK maturation, and consequently platelet function.12 In favor of this hypothesis, it has been demonstrated that polyploidization up- or down-regulates gene expression of a small number of genes in yeast.13 Whether polyploidization affects gene expression in MKs is not known. We have previously demonstrated that polyploidization was associated with a functional gene amplification14 and was not subjected to epigenetic silencing, as demonstrated in plants.15,16
To determine whether polyploidization governs gene expression in MKs, we cultured human CD34+ cells in the presence of TPO and sorted MKs according to their ploidy level. Gene profiling at different ploidy levels was studied using high-density oligonucleotide microarrays. Surprisingly, only a small number of genes (about 300) were regulated during polyploidization. Down-regulated genes corresponded mainly to immature markers of the MK differentiation, as do genes implicated in DNA replication and repair. In contrast, up-regulated genes segregated with genes involved in platelet function and cytoskeleton organization. These results suggest that polyploidization is implicated in the process of megakaryocyte terminal differentiation and may indirectly regulate platelet functions.
Materials and methods
In vitro growth of megakaryocytes from CD34+ cells
Cytapheresis samples from healthy individuals were obtained with their informed consent. Approval for the study was obtained from Assistance Publique des Hôpitaux de Paris (AP-HP). CD34+ cells were isolated using the immunomagnetic bead technique (Miltenyi Biotec, Paris, France).17 CD34+ cells were grown for 8 days in Iscove modified Dulbecco medium (Gibco BRL, Grand Island, NY) containing penicillin (100 U/mL), streptomycin (100 μg/mL), glutamine (2 mM; Sigma, St Louis, MO), α-monothioglycerol (76 nM; Sigma), 1.5% deionized bovine serum albumin (BSA; Cohn fraction V; Sigma), 1/100 Insulin-Transferrin-Selenium X (Gibco BRL), and sonicated lipids (20 μg/mL).18 The culture medium was supplemented with 10 ng/mL TPO (a generous gift from Kirin Brewery, Tokyo, Japan).
Cell sorting of megakaryocytes
Cells were recovered after 8 days of culture and incubated for 30 minutes at 4°C with the anti-CD41a mAb conjugated with FITC (BD PharMingen, San Diego, CA) in PBS. After washing in PBS-EDTA, cells were incubated for 2 hours in 0.01 mM Hoechst 33342 (Sigma, Saint Quentin Fallavier, France) at 37°C. Morphologic and sorting gates were determined as previously described,3 and CD41+ cells were sorted into 2N and 4N or 8N and 16N cell fractions at 500 cells per second using a FACSDiva flow cytometer (Becton Dickinson, Mountain View, CA) equipped with 2 argon lasers (tuned to 488 and 360 nm, respectively, and operating at 500 mW; Coherent Radiation, Palo Alto, CA) and a 100-μm nozzle.
RNA extraction and purification
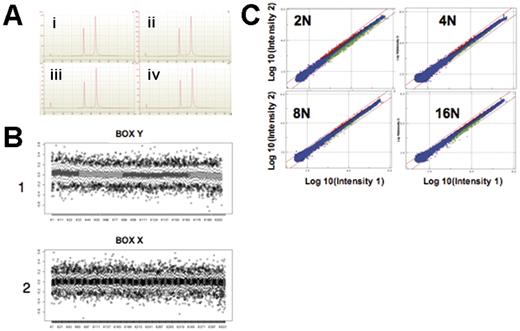
Frozen cell samples in pellets were lysed using 350 μL RLT lysis buffer (RNA lysis buffer containing guanidine isothiocyanate; Westburg, Leusden, The Netherlands) reconstituted with 1% β-mercaptoethanol. Total RNAs were isolated using the RNeasy microkit (QIAGEN, Cortaboeuf, France), as recommended by the manufacturer. RNA concentrations were adjusted precisely at 100 ng/μL and measured with a spectrophotometer (NanoDrop Technologies, Wilmington, DE). Quality of RNA preparations was assessed using the Lab-on-a-Chip 2100 bioanalyzer technology (Agilent Technologies, Palo Alto, CA), based on the 28S/18S ribosomal RNAs ratio (Figure 1A). All samples included in this study displayed a ratio of ribosomal RNAs superior to 1.5.
Quality control of microarray analysis. (A) RNA quality controls made with the 2100 expert software and the 2100 bioanalyzer. The first spike, corresponding to the nanomarker, is an internal control. The second spike corresponds to the 18S RNA and the third spike to the 28S RNA. A good-quality profile is defined by 4 indicators: the basal line must be regular, the 28S spike must be higher than the 18S spike, the 28S/18S ratio must be between 1.5 and 2.5, and the integrity number must be between 1 and 10. The 28S/18S ratio and the integrity number were 1.9 and 10, respectively, for populations 2N (a), 4N (b) and 16N (d), and 2.0 and 10, respectively, for population 8N (c). (B) Example of analysis of the homogeneity level in terms of LogRatio (log of red and green intensities) on the surface of a 44K human whole genome array. Box X2 and Box Y1 correspond to box plot representation (R software) along all vertical (box X) and horizontal (box Y) lines of probes (200 × 400 interleaved). Results show that the mean value of LogRatio is around 0 (due to normalization) without fluctuations of more than 0.04. (C) Representative example of intensities of 2N, 4N, 8N, and 16N populations (Intensity 2) versus the pool (Intensity 1) for combined dye-swap experiments in Resolver software. Probes in red (up-regulated) or green (down-regulated) are calculated at P < .001. Blue probes (unchanged) are considered nondiscriminating between different population samples and the pool. Red lines correspond to levels of fold-change of ± 2. 2N: 887 gene signatures with 561 down-regulated and 326 up-regulated. 4N: 261 gene signatures with 109 down-regulated and 152 up-regulated. 8N: 232 gene signatures with 106 down-regulated and 126 up-regulated. 16N: 362 gene signatures with 241 down-regulated and 121 up-regulated.
Quality control of microarray analysis. (A) RNA quality controls made with the 2100 expert software and the 2100 bioanalyzer. The first spike, corresponding to the nanomarker, is an internal control. The second spike corresponds to the 18S RNA and the third spike to the 28S RNA. A good-quality profile is defined by 4 indicators: the basal line must be regular, the 28S spike must be higher than the 18S spike, the 28S/18S ratio must be between 1.5 and 2.5, and the integrity number must be between 1 and 10. The 28S/18S ratio and the integrity number were 1.9 and 10, respectively, for populations 2N (a), 4N (b) and 16N (d), and 2.0 and 10, respectively, for population 8N (c). (B) Example of analysis of the homogeneity level in terms of LogRatio (log of red and green intensities) on the surface of a 44K human whole genome array. Box X2 and Box Y1 correspond to box plot representation (R software) along all vertical (box X) and horizontal (box Y) lines of probes (200 × 400 interleaved). Results show that the mean value of LogRatio is around 0 (due to normalization) without fluctuations of more than 0.04. (C) Representative example of intensities of 2N, 4N, 8N, and 16N populations (Intensity 2) versus the pool (Intensity 1) for combined dye-swap experiments in Resolver software. Probes in red (up-regulated) or green (down-regulated) are calculated at P < .001. Blue probes (unchanged) are considered nondiscriminating between different population samples and the pool. Red lines correspond to levels of fold-change of ± 2. 2N: 887 gene signatures with 561 down-regulated and 326 up-regulated. 4N: 261 gene signatures with 109 down-regulated and 152 up-regulated. 8N: 232 gene signatures with 106 down-regulated and 126 up-regulated. 16N: 362 gene signatures with 241 down-regulated and 121 up-regulated.
Oligo microarray technology
The Agilent Technologies long oligonucleotide (60 mers) technology is based on a dual-color analysis method, in which probes from samples and the reference are differentially labeled using incorporation of cyanine 5 and cyanine 3, respectively, and with inverse labeling. Dye-swap experiments were performed. Two different complementary rounds of experiments were carried out starting from the same samples. In the first design, the reference was RNA of 2N MKs, and the samples were RNA of 4N, 8N, and 16N MKs; experiments were conducted as dye-swap runs. In the second design, the reference was a pool of RNA obtained by mixing equal amounts of total RNA from each of the 4 samples used in the analysis. The quality of hybridization is shown in Figure 1B-C. The study included a reference versus reference control to assess accuracy of the method.
Probes were prepared according to the manufacturer (Low Fluorescent Low Input Linear Amplification kit; Agilent Technologies) from 500 ng total RNA. First, double-stranded cDNAs were synthesized using the MMLV reverse transcriptase and an oligodT-T7 RNA polymerase promoter. Then cDNAs were used as a template for an in vitro transcription process. CTP-Cy3– or CTP-Cy5–labeled cRNAs were fragmented to an average size of 50 to 100 nucleotides by heating the samples at 60°C using the specific fragmentation buffer provided by Agilent Technologies. One microgram of purified cRNA from each sample was mixed with the same amount of reference cRNA. Hybridization was performed on Whole Human Genome Oligo Microarray Kit 44K oligonucleotide microarrays (G4112A; Agilent Technologies) using reagents and protocols provided by the manufacturer.
Feature extraction software provided by Agilent Technologies (version 7.5) was used to quantify the intensity of fluorescent images and to normalize the results using the local background subtraction method according to the manufacturer.
Microarray analysis
The Resolver19 system for gene expression data analysis, developed by Rosetta Inpharmatics (Kirkland, WA), was used to perform analysis of variance (ANOVA). Clusters were created by means of the agglomerative hierarchical clustering algorithm. The Pearson correlation–based distance metric, with the average link as heuristic criteria, was used. The class prediction option of BrB ArrayTools software, version 3.2 (http://linus.nci.nih.gov/BRB-ArrayTools.html), was applied to discover genes that best discriminate the 2N + 4N from the 8N + 16N, at a level of P equal to .001.
Data availability
All data obtained from the microarray analysis have been submitted to Array Express at the European Bioinformatics Institute (http://www.ebi.ac.uk/arrayexpress/) (accession number E-TABM-133). Array Express is a public repository for microarray data that is designed to store well-annotated data in accordance with Microarray Gene Expression Data recommendations (http://www.mged.org).
Quantitative reverse transcriptase PCR
The 7900 HT Microfluidic cards with 384 wells (Applied Biosystems, Foster City, CA) were used. Sixty-one selected genes, shown by microarray analysis to be differentially expressed, were included in the cards, which were divided into 8 sets of assays. The Microfluidic cards were built for this study (correspondence among Gene symbol, RefSeq accession number, and ABI test identity are provided in Table S3, available on the Blood website; see the Supplemental Tables link at the top of the online article).
cDNA was reverse transcribed from 500 ng total ARN of each sample (2N, 4N, 8N, 16N, and a pool of the 4) using random primers and the High Capacity cDNA Archive kit (Applied Biosystems), according to the manufacturer's instructions, under a volume of 20 μL. cDNAs were diluted with 380 μL H2O, combined with 400 μL Universal Mix (Applied Biosystems), and 100 μL was used to fill each of the 8 reservoirs of the card. The last step consisted of sealing the Micro Fluidic card and trimming the reservoirs. Cycle threshold (CT) values were used for analysis. Validation of expression measurements was made by a Pearson correlation between 4 values for 2N, 4N, 8N, and 16N log ratios relative to the pool and 4 values of dCT between CT values of identical ploidy level and pool, respectively.
Immunolabeling for flow cytometric analysis
Cells were fixed at room temperature by 0.5% paraformaldehyde for 15 minutes and subsequently stained for 30 minutes at 4°C with an anti–CD41b-FITC (anti-GPIIb), an anti–CD61-FITC (anti-GPIIIa), an anti–CD34-FITC, and an anti–CD71-FITC (PharMingen, Becton Dickinson, Pont de Claix, France) mAb or with an anti–β2-microglobulin mAb (a gift from Dr Lemonnier, Pasteur Institute, Paris, France). Control cells were incubated with an irrelevant mouse IgG1 antibody, washed twice in PBS, and incubated for 30 minutes at 4°C with FITC-goat anti–mouse IgG (Chemicon International, Paris, France). After labeling, cells were incubated in a 0.9% NaCl solution containing 50 μg/mL propidium iodide (Sigma) and 100 mg/mL RNase A for 4 hours. Cell samples were then analyzed on a FACSort (Becton Dickinson) equipped with an argon laser (15 mW, 480 nm excitation). About 50 000 cells were listed and analyzed with the CellQuest software package (Becton Dickinson). The same protocol was used to stain VWF, β-actin, and GAPDH by means of a mouse anti–human VWF mAb (a generous gift from D. Meyer, INSERM U143, Hôpital de Bicêtre, Kremlin-Bicêtre, France), of a mouse anti–human β-actin mAb (Sigma), or of a mouse anti–human GAPDH mAb (Chemicon International) as a first antibody and an anti–mouse IgG-FITC as a second antibody. Prior to staining, paraformaldehyde-fixed cells were permeabilized by a 4-minute treatment with PBS containing 0.2% Triton X-100.
Results
Acquisition of MK at different ploidy levels
CD34+ cells obtained from cytapheresis samples were grown in serum-free liquid culture in the presence of TPO. In these culture conditions, CD41+ cells were detected as early as day 2, and their number gradually increased until day 9. The presence of polyploid cells was usually detected on day 6, with a variable frequency among samples. After day 9 or 10, MKs became too fragile to be sorted, so we chose day 9 of culture to perform the experiments. In addition, on day 9 the number of CD41+ cells was optimum, peaking at 80% as previously reported,10 and MKs had not yet started to form proplatelets. CD41+ MKs were sorted into 4 ploidy groups (2N, 4N, 8N, and 16N) identified by the Hoechst staining. As MKs with the highest ploidy (8N and especially 16N) were not frequent in the culture (average, 2%), it was necessary to collect MKs from 7 cytapheresis samples to make one pool, and 2 pools were prepared for our study. After sorting MKs in each ploidy class, cells were reanalyzed, and the purity was greater than 95%.
Microarray expression analysis of MKs of different ploidy level
Gene expression profiles from MKs sorted according to their ploidy levels were obtained using the Whole Human Genome Oligo Microarray Kit (Agilent Technologies) 44K oligonucleotide arrays. This technique allows a direct comparison of 2 populations labeled by 2 different dyes (Cy3 and Cy5). Two types of analyses were performed, one using total mRNA from the 2N population and the other using a pool containing an equal quantity of mRNAs from 2N, 4N, 8N, and 16N MKs as the reference populations. The second approach had the advantage of allowing comparison among the 4 sorted populations, whereas the first one excluded the 2N population from the analysis. All experiments were conducted in dye-swap. Genes obtained by an ANOVA from Resolver with P values below .001 were analyzed, and all genes with a very low intensity (signal less than 200) were excluded from the analysis.
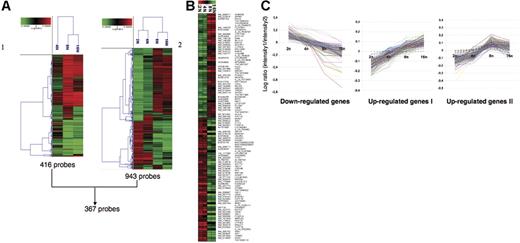
Using the first approach (2N as the reference population), the 3 populations could be grouped into 2 clusters, one corresponding to 4N and the other containing 8N and 16N cells. Four hundred sixteen sequences corresponding to 410 genes were found to distinguish 4N from 8N and 16N populations (Figure 2A, left) and thus provided a signature to the true polyploid MKs. However, the method had a clear limitation. Indeed, in contrast to 8N and 16N cells, the 4N population is not homogenous, since it may contain true polyploid 4N MKs and mitotic cells in the G2/M phase. Precise quantification of the percentage of polyploidy versus mitotic MKs present in the 4N population remains difficult, because there is presently no marker defining a 4N MK from an MK in mitosis. In addition, as previously reported,3 about 20% of the cells are still in mitosis at day 9 of culture. Thus, we performed additional experiments using a pool of mRNAs from MKs with different ploidy as the reference. This approach was more sensitive and allowed grouping of the cells into 2 classes, one class containing 2N and 4N cells and the other containing polyploid 8N and 16N cells. Nine hundred forty-three sequences corresponding to 917 genes were differentially regulated among 2N, 4N, 8N, and 16N populations (Figure 2A, right). Interestingly, the 2 approaches allowed the detection of 367 sequences in common.
Illustration of the microarrays results. Raw data files from the feature extraction software for image analysis were imported into the Resolver system for gene expression data analysis from Rosetta Informatics. Then combined experiments were generated to obtain average values from the replicates and dye-swap experiments in order to avoid dye incorporation bias. Microarray data were processed and combined using the Rosetta Resolver system, as described.19 (A) Hierarchical cluster analysis. (1) 416 probes: expression profiles in the 4N, 8N, and 16N MK populations compared to the 2N MK population. (2) 943 probes: expression profiles in the 2N, 4N, 8N, and 16N MK populations compared with an MK pool. ANOVA was performed on the 41 059 probes of the chips with a P threshold of < .001. These genes were subjected to hierarchical cluster analysis using a calculation based on Pearson correlation and agglomerative method on the average link. Each row represents the combination of 2 dye-swap experimental samples, and each column represents a single accession number. Three hundred sixty-seven probes are common between these 2 analyses. (B) Analysis of the discriminating genes between the 2N-4N and 8N-16N ploidy levels. Combined microarray data were exported from Resolver into BRB ArrayTools. Analysis of discriminating genes between the 2N-4N and the 8N-16N groups was performed using the class prediction module with a random variance model. As data were previously normalized by feature extraction software, no spot filtering and normalization were performed. Genes with greater than 50% missing values were excluded. Finally, 105 genes were selected based on a P threshold of < .001. (red squares) Overexpression versus the pool of all samples. (green squares) Underexpression versus the pool of all samples. Gene names are displayed as RefSeq or GenBank identifiers on the left, whereas they are displayed as gene symbols (or probe names) on the right. (C) Illustration of the 3 clusters of genes with a ploidy-regulated expression. Three clusters from 8 representing the genes for which the expression is down- or up- regulated with ploidy. Up-regulated genes are divided into 2 clusters, one with a linear increase of expression level between different classes of ploidy (cluster I) and another reaching a plateau between 8N and 16N (cluster II). The y axis represents the LogRatio between the intensity of hybridization of each ploidy level (2N, 4N, 8N, or 16N) and the intensity of hybridization of the MK pool. The x-axis represents the different ploidy levels.
Illustration of the microarrays results. Raw data files from the feature extraction software for image analysis were imported into the Resolver system for gene expression data analysis from Rosetta Informatics. Then combined experiments were generated to obtain average values from the replicates and dye-swap experiments in order to avoid dye incorporation bias. Microarray data were processed and combined using the Rosetta Resolver system, as described.19 (A) Hierarchical cluster analysis. (1) 416 probes: expression profiles in the 4N, 8N, and 16N MK populations compared to the 2N MK population. (2) 943 probes: expression profiles in the 2N, 4N, 8N, and 16N MK populations compared with an MK pool. ANOVA was performed on the 41 059 probes of the chips with a P threshold of < .001. These genes were subjected to hierarchical cluster analysis using a calculation based on Pearson correlation and agglomerative method on the average link. Each row represents the combination of 2 dye-swap experimental samples, and each column represents a single accession number. Three hundred sixty-seven probes are common between these 2 analyses. (B) Analysis of the discriminating genes between the 2N-4N and 8N-16N ploidy levels. Combined microarray data were exported from Resolver into BRB ArrayTools. Analysis of discriminating genes between the 2N-4N and the 8N-16N groups was performed using the class prediction module with a random variance model. As data were previously normalized by feature extraction software, no spot filtering and normalization were performed. Genes with greater than 50% missing values were excluded. Finally, 105 genes were selected based on a P threshold of < .001. (red squares) Overexpression versus the pool of all samples. (green squares) Underexpression versus the pool of all samples. Gene names are displayed as RefSeq or GenBank identifiers on the left, whereas they are displayed as gene symbols (or probe names) on the right. (C) Illustration of the 3 clusters of genes with a ploidy-regulated expression. Three clusters from 8 representing the genes for which the expression is down- or up- regulated with ploidy. Up-regulated genes are divided into 2 clusters, one with a linear increase of expression level between different classes of ploidy (cluster I) and another reaching a plateau between 8N and 16N (cluster II). The y axis represents the LogRatio between the intensity of hybridization of each ploidy level (2N, 4N, 8N, or 16N) and the intensity of hybridization of the MK pool. The x-axis represents the different ploidy levels.
The second analysis demonstrated that 2N and 4N MKs were closely related cell populations that clearly differed from the polyploid 8N and 16N cell populations. Between the 2 clusters (2N, 4N versus 8N, 16N), 105 discriminating genes were identified by the class prediction option of the BrB Array tool with P values below .001 (Figure 2B). Forty-eight genes were found to be up-regulated and 57 to be down-regulated in 8N and 16N MKs in comparison to 2N and 4N MKs. The lists of all genes (Figure 2A-B) are presented in Tables S1 and S2.
The expression profiling data indicate that it is possible to identify polyploid MKs by their gene expression and thus validate our approach. However, our main interest was to characterize the genes regulated during polyploidy and not only to define a signature for MK polyploidy. To precisely determine which genes are regulated during polyploidization, we essentially used the second approach, which allows a comparison of the 4 populations. Nine hundred forty-three sequences were arranged into 8 clusters. Five clusters did not correspond to genes of interest because they varied independently of the ploidy level. Thus, our study was focused on the 3 remaining clusters (Figure 2C).
Validation of the microarray results by QRT-PCR technique
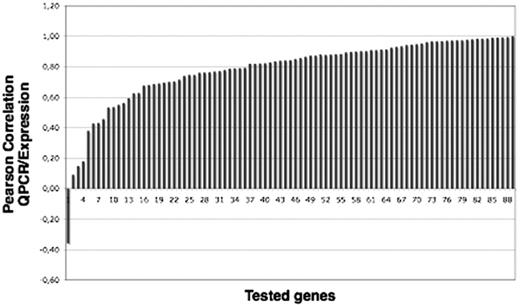
To confirm the results obtained by microarray analysis with a pool of different ploidy MKs as the reference, 61 of 917 differentially expressed genes were selected, and their expression was analyzed by reverse transcriptase–polymerase chain reaction (RT-PCR) using Applied Biosystems 7900 HT Microfluidic cards. The validation of expression measurements was made by a Pearson correlation between 4 values for 2N, 4N, 8N, and 16N LogRatios relative to the pool and 4 values of dCT between values of CT of identical ploidy level and pool, respectively. Due to some multiplicities of probes for each symbol gene (ie, quantitative RT-PCR [QRT-PCR]–selected amplimers), we obtained 88 correlation values for 61 genes (Figure 3), 53 of which were correlated with a coefficient of correlation greater than 0.55. The list of genes and the probes is provided in Table S3.
Validation of microarray results on 61 genes by QRT-PCR. The graph represents the Pearson correlation between data corresponding to the expression levels obtained by QRT-PCR and by microarray analysis for 88 probes representing 61 genes.
Validation of microarray results on 61 genes by QRT-PCR. The graph represents the Pearson correlation between data corresponding to the expression levels obtained by QRT-PCR and by microarray analysis for 88 probes representing 61 genes.
Profile of up- and down-regulated genes during polyploidization
Genes regulated by ploidy were grouped into 3 clusters (Figure 2C) and analyzed according to their biological role. The first cluster corresponded to genes continuously down-regulated during polyploidization (144 sequences, 106 known genes), the second to genes constantly up-regulated during polyploidization (205 sequences, 148 known genes), and the third to genes whose expression increases at 2N, 4N, and 8N to reach a plateau at 16N (206 sequences, 150 known genes). The 2 last clusters were difficult to discriminate; indeed, several oligonucleotides identifying the same genes were deposited on the microarray, and the same gene could be classified in the second or the third cluster, depending on the set of oligonucleotides. Thus, the 2 clusters comprising up-regulated genes were very close and were analyzed together (248 genes).
Down-regulated (106) and up-regulated (248) genes were classified into different categories according to their functions, described in UniGene and PubMed (http://www.ncbi.nlm.nih.gov). Results are shown in Table 1. Among down-regulated transcripts, 24 of 106 corresponded to genes involved in DNA replication and recombination repair. They included all MCM gene transcripts, CDC6, and CDC45L involved in DNA replication. One gene product involved in DNA repair, RAD21, was up-regulated, but RAD21 is also involved in the regulation of the mitotic cohesin complex, and its destruction facilitates chromosome segregation. In addition, some important genes involved in proliferation, such as E2F1, CDC2L1, CDCA7, and MYB, were down-regulated, as were metabolic genes involved in purine synthesis, such as DHFR and MTHFD1. Genes involved in cytokinesis, such as LRPPRC, were also down-regulated. Among the up-regulated transcripts, we identified genes encoding negative regulators of proliferation and mitosis, such as CDKN3, CDKN2D, and cyclin G1 (CCNG1). One marked exception was the up-regulation of cyclin A2 (CCNA2). Together, these observations indicate that each cycle of polyploidization increases the probability of cell cycle exit.
The cluster of major histocompatibility complex (MHC) class II genes (HLA-DR and DP) was also down-regulated during polyploidization. As a mirror, gene products of the MHC class I (HLA-B, -C, -E, -F, -G, and β2 microglobulin) were increased. This may suggest that polyploidization is tightly associated with MK terminal differentiation, because platelets are devoid of MHC class II antigens.20
In agreement with data obtained on MHC genes, most functional clusters of up-regulated genes corresponded to genes involved in proplatelet formation and platelet functions. Several transcripts of platelet glycoproteins, which are receptors for cell matrix or cell-cell interactions, were increased during ploidization. Essentially, they were GPIbα (GP1BA) and GPIbβ (GP1BB) and GPIX (GP9), α6 (ITGA6), β1 (ITGB1) and β3 (ITGB3) integrins, CD31, CD36, CD47, the prion protein (PRNP), CD226, and the prostacyclin receptor (PTGIR). An increase in transcripts of 2 tetraspannins (CD9 and CD63) associated in a network with integrins was also observed. Transcripts of important proteins or glycoproteins present in α-granules and involved in platelet function were also up-regulated, such as VWF, osteonectin (SPARC), thrombospondin (THBS1), CD62 (P-selectin; SELP), latent TGF-β–binding proteins (LTBP1), coagulation factor XIII (F13A1), and PF4V1. Transcripts of other chemokines (CXCL5, CCL5, CKLF), growth factors (angiopoietin [ANPT1], lefty [LEFTY1]), the chemokine receptor CXCR4, and receptors for growth factors, such as the erythropoietin-R (EPO-R) and activin A type I receptor (ACVR1), were also up-regulated.
Numerous genes encoding proteins involved in the structure or organization of the actin, myosin, and tubulin cytoskeleton were up-regulated. In the actin cytoskeleton, these included structural proteins such as β-actin (ACTB), α-actinin (ACTN1), KLHL5, actin-related protein 10 (ACTR10), and vinculin (VCL) and regulators such as Plekstrin (PLEK), Zyxin (ZYX), component of the Arp2/3 complex (ACTR3), actin-capping protein (CAPZA2), actin-depolymerizing protein (DSTN), and Rho GTPases such as RAC1 and mDia (DIAPH1). Similar results were seen for myosin, with an up-regulation of the heavy-chain MYH9 involved in several platelet diseases, the myosin light-chain MYL6, and tropomyosin (TPM1) or their regulators such as caldesmon 1 (CALD1). Even larger numbers of transcripts involved in the tubulin cytoskeleton, including centrosomes, were up-regulated. These included α1 (TUBA1), α2 (TUBA2) and α6 tubulin (TUBA6), and β1 tubulin (TUBB1), which are directly involved in proplatelet formation and platelet shape, as well as numerous regulators of polymerization such as MAPRE1 and MAPRE2. Several genes coding for proteins associated with centrosome or their regulation were also up-regulated (CKAP5 and caltractin [CETN2]).
One of the most important groups of up-regulated genes concerns molecules involved in signal transduction and protein transport. These comprise several gene transcripts of the Rab gene family (RAB1A, RAB4A, RAB6A, RAB27B), ARF4, BET1, ARL1, and FFFE2 (ARF-GAP) involved in the regulation of vesicle exocytosis. Similarly, kinases (FYN), adaptors (LAT, FYB, GRAP2, IRS2, SH3BP5), GTPase (RAP1B), or their regulators (KIFAP3) essentially involved in the regulation of integrin and GPVI signaling had an increased expression during polyploidization. Several up-regulated transcripts belong to the RAS/RAP1/MAP kinase and Src pathways. An important cluster of functional up-regulated transcripts constitute those involved in metabolism/biosynthesis, with one of them, MAOB, being involved in a platelet disease.
Protein accumulation during polyploidization
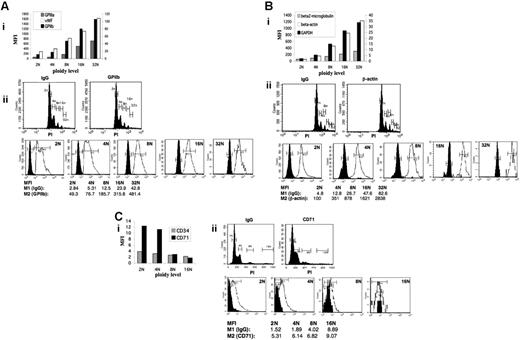
Modulations of transcripts levels occurring during polyploidization should translate into parallel changes at the protein level. To assess this assumption, we selected 6 genes that were up-regulated (GPIIIa [ITGB3], VWF, β-actin [ACTB], β2-microglobulin) or down-regulated (CD71 and CD34) on microarray analysis and 2 other genes for which no statistical data in expression level could be seen (GPIIb [MK specific] and GAPDH [ubiquitous]). Variations in corresponding proteins were quantified as a function of DNA content by flow cytometry. The amount of 3 MK-specific proteins involved in platelet functions—VWF, GPIIb, and GPIIIa (Figure 4A)—and 3 ubiquitous proteins—β-actin, GAPDH, and β2-microglobulin (Figure 4B)—increased proportionally to the ploidy level. Similar results were obtained at the mRNA level for VWF, GPIIIa, and β-actin by microarray, but not for GPIIb (Figure 2). The increase in β2-microglobulin protein level was considerably lower than that of the 5 other proteins but was still linear. An increase in β2-microglobulin at the mRNA level was observed only between 4N and 8N (Table S1; cluster not shown). The protein level of 2 immature MK markers, CD34 and CD71, decreased with ploidy class (Figure 4C).
Relationships among VWF, GPIIb, GPIIIa, β-actin, GAPDH, β2-microglobulin, CD34 and CD71 protein level, and ploidy. The levels of (A) VWF, GPIIb, GPIIIa, (B) β-actin, GAPDH, β2-microglobulin, and (C) CD34 and CD71 in each ploidy class were measured by flow cytometry. (i) Geometric mean fluorescence intensity (MFI) was calculated for each ploidy class as follows. Each ploidy class was first gated (from 2N to 16N) on the propidium iodide (PI) labeling. MFI in FL1 was measured for each specific antibody and its isotype-matched control (IgG). The corrected MFI for each ploidy was calculated by subtraction of the isotype MFI to the MFI obtained with the specific antibody. (ii) Examples of MFI measurement. Cells were stained with specific antibody and PI and gated on the ploidy class (middle panels). MFI designed as M1 for IgG and as M2 for (A) GPIIb, (B) β-actin, and (C) CD71, respectively, was obtained for each ploidy level separately by measuring the fluorescence intensity in FL1 (lower panels). Corrected MFI is equal to the subtraction of M1 from M2. Filled areas represent background fluorescence (M1), whereas open areas represent fluorescence obtained with the specific antibody (M2).
Relationships among VWF, GPIIb, GPIIIa, β-actin, GAPDH, β2-microglobulin, CD34 and CD71 protein level, and ploidy. The levels of (A) VWF, GPIIb, GPIIIa, (B) β-actin, GAPDH, β2-microglobulin, and (C) CD34 and CD71 in each ploidy class were measured by flow cytometry. (i) Geometric mean fluorescence intensity (MFI) was calculated for each ploidy class as follows. Each ploidy class was first gated (from 2N to 16N) on the propidium iodide (PI) labeling. MFI in FL1 was measured for each specific antibody and its isotype-matched control (IgG). The corrected MFI for each ploidy was calculated by subtraction of the isotype MFI to the MFI obtained with the specific antibody. (ii) Examples of MFI measurement. Cells were stained with specific antibody and PI and gated on the ploidy class (middle panels). MFI designed as M1 for IgG and as M2 for (A) GPIIb, (B) β-actin, and (C) CD71, respectively, was obtained for each ploidy level separately by measuring the fluorescence intensity in FL1 (lower panels). Corrected MFI is equal to the subtraction of M1 from M2. Filled areas represent background fluorescence (M1), whereas open areas represent fluorescence obtained with the specific antibody (M2).
Discussion
Polyploidization is a rare event in mammals and only concerns some rare types of cells. Generally, polyploidization is not tolerated in humans, leading to spontaneous abortions. In contrast, polyploidy is common in yeasts and plants and even occurs in amphibians and fish. In these species, polyploidization may facilitate flexibility and long-term evolution by gene expression alteration due mainly to epigenetic silencing and the return to diploidization.15,16,21-23
In MKs, polyploidization is related to the requirement of cell enlargement necessary to optimize platelet biogenesis. Therefore, it appears that polyploidization is a way to amplify the genome and thus to increase protein synthesis, facilitating cell growth. One intriguing challenge is to understand whether MK polyploidization regulates gene expression, as demonstrated in yeast, or whether polyploidization is part of the differentiation process that leads to platelet production.
During in vivo marrow MK differentiation, polyploidization is part of the differentiation process but seems to be independently regulated from platelet formation. This assumption is based on several pieces of evidence: (1) during MK differentiation, polyploidization precedes maturation, a phenomenon in parallel with that observed in other hematopoietic lineages in which complete cytoplasmic maturation is associated with postmitotic arrest; (2) some genes regulating platelet production, such as p45NF-E2,24 are not involved in the regulation of polyploidization, whereas genes that increase MK ploidy, such as cyclin D3, do not modify platelet levels25 ; (3) in some mouse background, the modal ploidy level is 32N instead of 16N, without major changes in the platelet number or function26 ; and (4) in human malignant hemopathies or during ontogenesis or in culture from CD34+ cells, it has been shown that some micro-MKs produce platelets.7,8 These 2N and 4N cells are mature MKs with a well-developed demarcation membrane system (DMS) and α-granules, and they clearly differ from 2N MK precursors (promegakaryoblasts) that display only rudimentary DMS and have only begun granule biogenesis.27
Thus, whether polyploidization and terminal MK differentiation are 2 independent processes or 2 differentially regulated but intricate processes remains a matter of speculation. In an attempt to solve this question, we took advantage of an in vitro MK culture system from human CD34+ cells to study gene expression during polyploidization. This culture system allows the entire MK differentiation, including platelet formation, but is not entirely synchronous. In addition, the ploidy level reached at any time of the culture is lower than in the marrow, usually with a modal ploidy of 4N or 8N. This can be intrinsically due to the culture system that, being asynchronous, always includes some immature MKs and/or may favor proliferation of MK progenitors over polyploidization. Nevertheless, there is no alternative to this culture system in humans to perform such an analysis because of the low number of MKs in the marrow. Knowing this limitation, we have sorted MKs on their ploidy level, and we have studied their transcriptome using pan-genomic microarrays. To perform the study, the microarray technique was optimized in 2 ways: to deal with low amounts of RNA and to analyze low changes in gene expression because the majority of fold changes were less than 2.
As a first approach, hierarchical clustering was performed that indicated the presence of 2 cell clusters, one corresponding to 2N and 4N cells, the other to 8N and 16N cells, with 105 discriminating genes by the class prediction option (P < .001) of the BrB Array tool. The most likely hypothesis was that the 2N and 4N cell cluster was composed of a majority of immature cells and that 4N cells were not truly tetraploid but rather cells undergoing mitosis. Among the 105 discriminating genes, none except CD34 and CD33, which were found to be down-regulated in 8N and 16N cells, are known to be related to differentiation. This may imply that 8N and 16N MKs have properties different from 2N and 4N MKs, a phenomenon that could be reminiscent of ontogeny, where fetal MKs have a low ploidy.28
The fact that the clustering was not based on genes regulated by differentiation supports the hypothesis that our approach does not induce a major bias in the study of genes regulated during polyploidization. Therefore, we focused our study on genes that were either continuously down-regulated during polyploidization (whose expression diminishes at each ploidy level) or up-regulated. For the up-regulated genes, it was difficult to discriminate between genes whose expression constantly increased from 2N to 16N from those whose expression increased until 8N and subsequently remained stable. Therefore, these 2 categories of genes were analyzed together. Taking into consideration that changes in expression on RNA level are followed by changes in protein level, this approach led to 2 major conclusions.
The first major conclusion was that a significant fraction of the down-regulated genes corresponded to genes involved in DNA replication, such as CDC6 and MCM genes, proliferation, or DNA repair. MK polyploidization proceeds as normal mitosis through G1, S, G2, and M phases. However, mitosis is incomplete in MKs with an absence of karyokinesis and cytokinesis. Thus, each round of polyploidization is associated with a new S phase,29 indicating that CDC6 and MCM gene products play an important role in the polyploidization process. The finding that CDC6 and MCM genes were down-regulated with a ploidy level could reflect that the probability of increasing ploidy diminishes at each ploidy level. In support of this hypothesis, BrdU labeling shows that the percentage of MKs in S phase diminishes during ploidization (data not shown). However, we cannot eliminate the hypothesis that this decrease in S-phase cells is related to a change in cell cycle during ploidization, with a longer G1 phase and a shorter S phase. Such a change in cell cycle will also lead to a decrease in cells expressing DNA replication genes during polyploidization. Interestingly, none of the gene products presumably involved in the endomitotic process, such as cyclin D3,30 cyclin B1,3,31,32 Aurora B, or survivin,33-35 were down-regulated during ploidization with exception for cyclin A2. From these data, it may be postulated that most genes involved in polyploidization are regulated at the protein level.
The second major conclusion was that a large fraction of up-regulated genes is related to MK differentiation, a finding in accordance to previous gene profiling studies aiming to study human MK differentiation.36,37 Indeed, it appears that genes down-regulated during polyploidization were not regulated by MK differentiation, except for the transcription factor MAX. In contrast, a large fraction of genes (F13A1, GRAP2, ITGB3, THBS1, HGD, ITGA6, ABCC3, FYB, FYN, HMMR, GP1BA, NID, GP9, PECAM1, SELP, VWF, VCL, ZYX, RGS10, MAPRE, RIT1) whose expression increased during polyploidization were also found to increase during differentiation in the 2 previous studies.36,37
Recently, gene profiling has been performed on murine MKs at different levels of differentiation.38 MKs were sorted on their forward scatters, and these different MK populations, although not homogenous for a ploidy class, included MKs with different modal ploidy. In accordance with our results, down-regulation of the expression of genes involved in DNA replication and up-regulation of the expression of genes involved in cytoskeleton, cell migration, G-protein signaling, and platelet function were detected.
Interestingly, angiopoietin-1 (ANGPT1), a growth factor essential for blood vessel development but not directly involved in platelet biology, was positively regulated during MK ploidization. Previous works have already demonstrated the presence of ANGPT1 in MKs39 and platelets, as well as its release after the platelet activation by thrombin.40 Together, these results suggest that ANGPT1 synthesized in MKs and released by activated platelets could play an important role both in angiogenesis and in the regulation of hematopoiesis.
Our study also emphasizes the role of the cytoskeleton during MK development and, probably, in proplatelet formation and platelet function. Indeed, several tubulin genes, including β1 tubulin, actin cytoskeleton genes, and both MYH9 and MLC, were found to be up-regulated during ploidization.
These observations strongly suggest that polyploidization is included in the terminal differentiation program of the MK and that an up-regulation of genes involved in platelet biogenesis and function already begins during polyploidization. This result gives some support to the hypothesis that platelets produced from MK with different ploidy levels may have different functions. Alternatively, our results could be explained by a 2-fold increase in probability to enter differentiation at each ploidy level, whereas at the same time the probability to replicate DNA diminishes.
One intriguing result concerns the absence of marked changes during ploidization in the level of the transcription factors involved either in the regulation of MK-specific genes, such as GATA-1, FOG-1, FLI-1, SCL/TAL1, and AML1, or in platelet production, such as p45NF-E2, whereas transcripts of their target genes were up-regulated. This may be due to several non–mutually exclusive mechanisms: translational or posttranslational regulations or changes in multiprotein complexes containing these transcription factors. Further investigations remain necessary to determine how polyploidization and MK differentiation may modify gene expression. In addition, this intertwining between ploidization and expression of MK-specific genes was not obvious from knockout experiments. Disruption of 2 essential MK transcription factors, GATA-1 and Fli-1, leads to both a decrease in ploidy level41,42 and a partial blockage in differentiation with impaired expression of MK-specific genes. In contrast, p45NF-E2 disruption inhibits proplatelet formation but does not alter ploidy level.24 When cyclin D3 and cyclin D1 were overexpressed in transgenic animals, the ploidy level was increased without an increase in platelet production.25,43
Our study clearly demonstrates that MK polyploidization differs from the other models of cell polyploidization. In yeast, it was shown that only 17 genes had a ploidy-dependent expression without correlation with a precise function.13 More recently, it has been shown in rat vascular smooth muscle that polyploidization was associated with aging and with a down-regulation of a few numbers of genes.44 In plants, polyploidization is associated with a down-regulation of some genes by epigenetic silencing.15,16 These data contrast with MK polyploidization that is associated with up- and down- regulation of multiple genes, of which the majority corresponds to genes involved in terminal differentiation and platelet biogenesis. It would be interesting to know whether polyploidization of the hepatocyte has a biological significance similar to MK polyploidization or also corresponds to cell aging.
Our study demonstrates that MK polyploidization leads to the coordinated expression of genes involved in the arrest of DNA replication and in platelet functions and production. Therefore, polyploidization is a cell process that is part of terminal MK differentiation. How the polyploidization process may interact with the transcription machinery in MKs remains to be defined.
Authorship
Contribution: H.R. performed research, analyzed data, and wrote the paper. A.K. performed research, analyzed data, and wrote the paper. D.S. performed microarray experiments. H.R. performed statistical analysis. F.L. performed flow cytometry analysis. T.R. performed microarray and PCR experiments.
D.T.L. discussed results. G.K. discussed results. N.D. performed research. P.D. performed statistical analysis and designed microarray experiments. V.L. performed microarray and PCR experiments and designed microarray experiments. W.V. designed the research and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
H.R. and A.K. contributed equally to this study.
Correspondence: Hana Raslova, INSERM U790, Institut Gustave Roussy, PR1, 39 rue Camille Desmoulins, 94805 Villejuif, France; e-mail: hraslova@igr.fr.
The online version of this article contains a data supplement.
An Inside Blood analysis of this article appears at the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
This work was supported by grants from Ligue Nationale contre le Cancer (équipe labellisée 2004 and 2007) and Agence Nationale de la Recherche (contrat blanc). D.S. was supported by fellowships from the Institut Gustave Roussy and Amgen.
We thank F. Wendling for critical reading of the manuscript and helpful suggestions, K. Brewery (Tokyo, Japan) for the gift of human recombinant thrombopoietin, D. Meyer (INSERM U143, Hôpital de Bicêtre, Kremlin-Bicêtre, France) for a generous gift of anti-VWF mAb, and D. Lemonnier (Pasteur Institute, Paris, France) for a generous gift of anti–β2-microglobulin mAb.