Abstract
Mesenchymal stem cells (MSCs), in addition to their multilineage differentiation, have a direct immunosuppressive effect on T-cell proliferation in vitro. However, it is unclear whether they also modulate the immune system by acting on the very first step. In this investigation, we addressed the effects of human MSCs on the differentiation, maturation, and function of dendritic cells (DCs) derived from CD14+ monocytes in vitro. Upon induction with granulocyte-macrophage colony-stimulating factor (GM-CSF) plus interleukin-4 (IL-4), MSC coculture could strongly inhibit the initial differentiation of monocytes to DCs, but this effect is reversible. In particular, such suppression could be recapitulated with no intercellular contact at a higher MSC/monocyte ratio (1:10). Furthermore, mature DCs treated with MSCs were significantly reduced in the expression of CD83, suggesting their skew to immature status. Meanwhile, decreased expression of presentation molecules (HLA-DR and CD1a) and costimulatory molecules (CD80 and CD86) and down-regulated IL-12 secretion were also observed. In consistence, the allostimulatory ability of MSC-treated mature DCs on allogeneic T cells was impaired. In conclusion, our data suggested for the first time that human MSCs could suppress monocyte differentiation into DCs, the most potent antigen-presenting cells (APCs), thus indicating the versatile regulation of MSCs on the ultimate specific immune response.
Introduction
Dendritic cells (DCs), the most potent antigen-presenting cells (APCs), are rare and ubiquitously distributed in the human body. They are derived from CD34+ bone marrow stem cells and can be generated from blood monocytes in vitro by culture with granulocyte-macrophage colony stimulating factor (GM-CSF) and interleukin-4 (IL-4).1-3 DCs play a major role in the uptake, transport, and presentation of antigens with the unique capacity to stimulate naive T lymphocytes.4,5 The ability of DCs to initiate an immune response depends on their transition from antigen-processing to antigen-presenting cells, during which they up-regulate class II major histocompatibility complex (MHC) and T-cell costimulatory molecules (CD80, CD86) in the cell surface, a process referred to as DC maturation.6,7 This transition constitutes an important checkpoint in mounting an immune response because immature DCs not only fail to prime T cells effectively8,9 but also serve to promote tolerance induction.10-13 In addition to their polarizing capacity on naive T cells, they can interact with B cells14 and natural killer (NK) cells.15 Thus, DCs are critical in the initiation of primary immune responses, becoming an important target for immunosuppression to prevent allograft rejection.16-19
Mesenchymal stem cells (MSCs) are multipotential cells, most of which reside within the bone marrow. They can be induced to differentiate into various components of the marrow microenvironment, such as bone, adipose, and stromal tissues.20 Studies conducted in both human and animal models have demonstrated that MSCs are capable of long-term engraftment and multilineage differentiation in vivo.21,22 Furthermore, the intravenous administration of MSCs leads to a modest but significant prolongation of skin graft survival similar to the immunosuppressive agents currently being used clinically. These findings suggest that MSCs may harbor an immunoregulatory activity. Indeed, human MSCs share cell surface markers with thymic epithelium. They express adhesion molecules involved in T-cell interaction, including vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule-1 (ICAM-1), and leukocyte function-associated antigen-3 (LFA-3).20,23 Moreover, MSCs express human leukocyte antigen (HLA) MHC class I and negligible levels of both MHC class II and Fas ligand; they do not express B7-1, B7-2, CD40, or CD40L.24,25 Recently, human and baboon MSCs displayed immunosuppressive properties on T-lymphocyte proliferation in vitro.25-27 However, the induction of immune responses does not rely solely on T cells but also on essential and complicated interactions between DCs and T cells. It is highly possible that MSCs may modulate the generation and antigen-presenting capacity of DCs prior to antigen-specific lymphocyte activation and proliferation.
Based on this hypothesis, we addressed here the negative regulation of MSCs on monocyte-derived DCs regarding from initial differentiation, IL-12 secretion to ultimate stimulatory ability, hence revealing DCs as one of the targets of MSCs displaying immunosupressive effects.
Materials and methods
Generation of human MSCs
The Research Ethics Committees of North Tai Ping Lu Hospital approved human bone marrow samples for research purposes. The health donors gave written consent for the use of bone marrow for research purposes.
MSCs were generated from normal human bone marrow samples. The mononuclear cells were prepared by gradient centrifugation at 900g for 30 minutes on Percoll (Amersham Biosciences, Uppsala, Sweden) at a density of 1.073 g/mL. Then cells were washed, counted, and plated at 2.0 × 105/cm2 onto 24-well tissue culture plates (Corning Costar, Corning, NY) in low-glucose Dulbecco modified Eagle medium (LG-DMEM; Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (FBS; Hyclone, Logan, UT). Medium was replaced, and the nonadherent cells were removed at 48 hours of initial culture and every 3 or 4 days thereafter. When 70% to 80% confluent, adherent cells were trypsinized (0.05% trypsin at 37°C for 5 minutes), harvested, and expanded in larger flasks. A homogenous cell population was obtained after 2 weeks of culture. Before further expansion and experimental use, MSCs were tested for their ability to differentiate into adipocytes, osteoblasts, and chondrocytes. Adipocytic differentiation was induced by DMEM containing 10% FBS, 0.5 mM isobutylmethylxanthine, 5 μg/mL insulin, 1 μM dexamethasone, and 60 μM indomethacin, whereas 1 mM β-glycerol phosphate, 0.1 μM dexamethasone, and 50 μM ascorbate were used for osteoblastic differentiation. Oil red O and von Kossa dyes were used to identify adipocytes and osteoblasts, respectively. Medium with DMEM containing 2.5% FBS, 50 ng/mL transforming growth factor-β1 (Peprotech, Rocky Hill, NJ), 50 μg/mL ascorbic acid, 1 mM sodium pyruvate, 6.25 μg/mL bovine insulin, 6.25 μg/mL transferrin, 6.25 μg/mL selenious acid, and 1.25 μg/mL bovine serum albumin was used for chondrogenic differentiation. Extracellular matrix, used to assess chondrogenic differentiation, was detected by Alcian blue staining. The identity of MSCs was also confirmed by immunophenotypic criteria based on the expression of CD73 and CD105 and the absence of hematopoietic (with anti-CD45, -CD14, -CD34 antibodies) and endothelial cell (with anti-CD31 antibodies) markers. All antibodies were purchased from BD Pharmingen (San Diego, CA). MSCs were maintained in culture for no more than 15 passages in vitro.
Differentiation of human CD14+ monocytes
Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Paque (1.077g/mL; Invitrogen) density gradient centrifugation. A total of 2 × 107 PBMCs were cultured in 10 mL medium per 75 cm2 flask (Corning). After 2 hours or overnight the cells were gently agitated and the adherent cells collected. Monocytes (more than 85% CD14+) were purified from adherent cells by using the MACS Monocyte Isolation Kit (Miltenyi Biotec, Bergisch Gladbach, Germany), and 1 × 106 cells per milliliter were cultured in 2 mL RPMI 1640, 10% FBS, recombinant GM-CSF (rGM-CSF) (10 ng/mL), rIL-4 (500 U/mL) in 6-well flat-bottomed plates. In some experiments, tumor necrosis factor-α (TNF-α) (20 ng/mL) was added at the initial culture. Irradiated MSCs (15 Gy) were added to the 6-well plates at a density of 2 × 105 cells per well unless otherwise indicated. To prevent the contamination of MSCs when harvested monocyte-derived cells, transwell chambers with a 0.4 μm pore size membrane (Corning) were used. MSCs were cultured on the reverse side of the membrane of the chamber. After obtaining a confluent feeder layer, monocytes were seeded on the upper side of the membrane of the chamber. Sometimes MSCs were cultured in the lower compartment, while monocytes on the upper compartment, to avoid the cell-cell contact. Cytokines were added to the cultures every 3 days. Neutralizing anti-IL-6 and anti-M-CSF monoclonal antibodies (mAbs) were added alone or together at day 0 and day 3 at the concentration of 10 μg/mL (R&D Systems, Minneapolis, MN). To induce maturation of monocyte-derived cells, lipopolysaccharide (LPS) (1 μg/mL) was added for another 48 hours of culture with GM-CSF and IL-4.
FACS analysis
The phenotype of cells was analyzed by flow cytometry using a FACSvantage cytofluorometer (Becton Dickinson, San Jose, CA). For monocyte-derived cells, the following mAbs were used: phycoerythrin (PE)-labeled anti-CD14, anti-CD1a, anti-CD80, anti-CD86, anti-CD83, and anti-HLA-DR (BD Pharmingen). Results are expressed as a percentage of positive cells.
Endocytosis
Endocytosis was measured as the cellular uptake of fluorescein isothiocyanate (FITC)-dextran and was quantified by flow cytometry. Approximately 5 × 105 cells per sample were incubated in medium containing FITC-dextran (1 mg/mL; molecular weight 40 000; Sigma, St Louis, MO) for 60 minutes. After incubation, cells were washed twice with cold phosphate-buffered saline (PBS) to stop endocytosis and remove excess dextran and were then fixed in cold 1% formalin. The quantitative uptake of FITC-dextran by the cells was determined by fluorescence-activated cell sorting (FACS), and the results were expressed in mean fluorescence intensity (MFI) values. At least 10 000 cells per sample were analyzed.
Quantification of human cytokines
IL-6, M-CSF, IL-12, interferon-γ (IFN-γ), and IL-10 were quantified in the cell culture supernatants by enzyme-linked immunosorbent assay (ELISA; R&D Systems). Detection limits were 3.12 pg/mL for IL-6, 31.2 pg/mL for M-CSF, 7.8 pg/mL for IL-12, 15.6 pg/mL for IFN-γ, and 7.8 pg/mL for IL-10. The measurement was done completely according to the protocol of the manufacturer.
Mixed leukocyte reactions (MLRs)
Monocyte-derived cells cultured with GM-CSF plus IL-4 for 7 days were irradiated (30 Gy) and cultured in triplicate at 5 × 103,1 × 104,or2 × 104 cells per 200 μL per well in 96-well flat-bottomed plates with 1 × 105 allogeneic CD4+ T cells purified from PBMCs by using the MACS CD4 isolation kit (Miltenyi Biotec). After 4 days, cells were pulsed during the last 16 hours with 3H-thymidine (1 μCi/well [0.037 MBq/well]). Thymidine incorporation was measured by standard liquid scintillation counting. Results are expressed in counts per minute and shown as means ± SDs of triplicate values.
KLH presentation assay
Keyhole-limpet hemocyanin (KLH) (Sigma) was used to test antigen-specific T-lymphocyte response. The DCs were loaded with 25 μg/mL to 100 μg/mL KLH at 37°C for 60 minutes. A total of 2 × 104 KLH-loaded DCs were cultured with a constant number of 1 × 105 CD4+ T cells for 4 days. During the last 16 hours of each culture period, cells were pulsed with 3H-thymidine (1 μCi/well [0.037 MBq/well]). Results are expressed in counts per minute and shown as means ± SDs of triplicate values.
Western blotting
Cells were resuspended in lysis buffer (Biorad, Hercules, CA). After at least 30 minutes on ice, insoluble fragments were removed by centrifugation (9838g, 4°C). Proteins were separated on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto nitrocellulose membranes. Nonspecific binding was blocked by incubating the membranes with 5% wt/vol nonfat dry milk (for phosphor-p38 mitogen-actviated protein [MAP] kinase antibody, phosphor-p38 (P-p38); Cell Signaling, Beverly, MA) or 5% bovine serum albumin (BSA) (for p38 MAP kinase antibody, p38; Cell Signaling) and TBST (0.05% Tween 20 in Tris [tris(hydroxymethyl)aminomethane]-buffered saline [TBS]). Membranes were incubated with anti-p38 or anti-phosphor-p38 mAb in the appropriate dilution overnight at 4°C. After incubation, membranes were rinsed with distilled water and washed 3 times in wash buffer (TBST). Secondary antibody conjugated to horseradish peroxidase was added to the membranes in 5% nonfat dry milk or 5% BSA in TBST in the appropriate dilution. After 1 hour of incubation at room temperature, membranes were washed 3 times and developed using an enhanced chemiluminescence kit (Pierce, Rockford, IL).
Results
Human MSCs strongly inhibited the initial differentiation of DCs from monocytes
Highly enriched CD14+ monocytes (more than 85% negative for CD1a) were cultured in the presence of GM-CSF (10 ng/mL) and IL-4 (500 U/mL) with (MSC-Mo) or without MSC coculture (Ctr-Mo). After 7 days of incubation, LPS was added for another 48 hours to promote the differentiation and maturation of DCs.28,29 As expected, Ctr-Mo acquired the size, became nonadherent and clustered, and displayed different protruding veils with abundant cytoplasm, a typical morphology of DCs (Figure 1A, upper row). In parallel, these cells displayed increased expression of CD1a in contrast with decreased or absence of CD14 expression. After exposure to LPS, they further differentiated into fully mature DCs characterized by up-regulated expression of CD83 and costimulatory molecule CD80 (Figure 1B, upper row). The presence of MSCs in the initial culture did not affect cell recovery (Ctr-Mo, 52% ± 10%; MSC-Mo, 47% ± 15%). The MSC-Mo's also acquired the size; however, they were round, scattered, lacking in veiled appearance, and developed macrophage morphology with numerous vacuoles (Figure 1A, lower row). In contrast to Ctr-Mo, MSC-Mo retained high CD14+ without acquisition of CD1a and displayed no up-regulation of CD83 and CD80 even under strengthened maturation stimuli—that is, doubled concentration of GM-CSF, IL-4, and LPS to rule out the consumption of cytokines by MSCs (Figure 1B, lower row and data not shown). In some experiments, the same cell dose of MSCs was added to the Ctr-Mo group in the process of immunostaining to counteract the possible difference caused by the presence of MSCs in MSC-Mo. As a matter of fact, MSCs, before or after being cultured with monocytes and cytokines, did not express CD14, CD1a, CD80, and CD86. By using the transwell system (described in “Materials and methods”) the possible contamination of MSCs in collection of cells for phenotype analysis was further excluded. These data clearly suggested that the presence of MSCs was capable of preventing bipotential monocytes from differentiating along the DC pathway.
Human MSCs inhibited the initial differentiation of DCs from CD14+ monocytes. (A) Monocytes cultured in the presence of GM-CSF and IL-4 for 7 days and then LPS for another 48 hours (Ctr-Mo) show clustered and protruding veils resembling mature DCs (top row). In contrast, monocytes with MSC coculture (MSC-Mo) display macrophage-like morphology (bottom row) (for left, middle, and right columns for both rows: original magnifications × 40, × 200, and × 1000, respectively). Cell morphology was evaluated by phase contrast microscopy and Wright-Giemsa staining (Sigma, Saint Louis, MO). Images in panel A were visualized using either an Olympus BH-2 microscope (right column) or a Nikon TE2000-U microscope (left and middle columns) equipped with Plan Fluor 4×/0.13 and 20×/0.45 and Splan oil 100×/1.25 objective lenses (Nikon, Tokyo, Japan). Images were captured with a Nikon Coolpix 995 camera and processed with Adobe Photoshop 7.0 software (Adobe, San Jose, CA). (B) Flow cytometry was employed to analyze the surface molecules expressed by Ctr-Mo (top row) and MSC-Mo (bottom row). Horizontal bars indicate the positive region; the corresponding percentage is shown above each bar. Results shown are representative of 3 independent experiments. (C) CD14 and CD1a expression of monocyte-derived cells in MSC/monocyte cocultures at ratios ranging from 1:10 to 1:200 was assessed by flow cytometry. Results are representative of 2 independent experiments.
Human MSCs inhibited the initial differentiation of DCs from CD14+ monocytes. (A) Monocytes cultured in the presence of GM-CSF and IL-4 for 7 days and then LPS for another 48 hours (Ctr-Mo) show clustered and protruding veils resembling mature DCs (top row). In contrast, monocytes with MSC coculture (MSC-Mo) display macrophage-like morphology (bottom row) (for left, middle, and right columns for both rows: original magnifications × 40, × 200, and × 1000, respectively). Cell morphology was evaluated by phase contrast microscopy and Wright-Giemsa staining (Sigma, Saint Louis, MO). Images in panel A were visualized using either an Olympus BH-2 microscope (right column) or a Nikon TE2000-U microscope (left and middle columns) equipped with Plan Fluor 4×/0.13 and 20×/0.45 and Splan oil 100×/1.25 objective lenses (Nikon, Tokyo, Japan). Images were captured with a Nikon Coolpix 995 camera and processed with Adobe Photoshop 7.0 software (Adobe, San Jose, CA). (B) Flow cytometry was employed to analyze the surface molecules expressed by Ctr-Mo (top row) and MSC-Mo (bottom row). Horizontal bars indicate the positive region; the corresponding percentage is shown above each bar. Results shown are representative of 3 independent experiments. (C) CD14 and CD1a expression of monocyte-derived cells in MSC/monocyte cocultures at ratios ranging from 1:10 to 1:200 was assessed by flow cytometry. Results are representative of 2 independent experiments.
Fibroblasts, as previously reported by Chomarat et al,30 switch monocyte differentiation to macrophages rather than to DCs, and full skewing was obtained with a ratio of 1 fibroblast to 4 monocytes. While in the presence of MSCs, as shown in Figure 1C, with the MSC/monocyte ratio of 1:10, the differentiation of monocytes to DCs was completely blocked with no significant difference, and even the ratio decreased to 1:20 and 1:40 (Figure 1C and data not shown). Such inhibitory effect became minor until the ratio dropped to 1:200. Addition of TNF-α at the onset of the fibroblast/monocyte coculture is reportedly able to reverse the skewing effect of fibroblast, resulting in the generation of cells with DC phenotype.31 As for MSCs here, nevertheless, adding TNF-α at the initial MSC/monocyte coculture, even with doubled concentration, did not result in the generation of cells with DC phenotype. No down-regulation of CD14 and no acquisition of CD1a were found. These results further indicate, as compared with fibroblasts at least, the suppressive effect of MSCs on the initial differentiation of DCs from monocytes was strong.
Suppression of DC generation without intercellular contact at a higher MSC/monocyte ratio
In addition, we determined if the inhibition of MSCs on the initial differentiation of DCs required intercellular contact. For this purpose, the transwell chamber system was used to separate monocytes (upper compartment) from MSCs (lower compartment). As shown in Figure 2A, a higher MSC/monocyte ratio (1:10) could completely prevent monocyte differentiation to DCs, like the coculture system described in “Materials and methods.” As the ratio decreased to 1:20 and 1:50, monocytes were able to generate immature DCs (ie, down-regulated CD14 coupled with up-regulated CD1a expression) in the presence of GM-CSF and IL-4 and underwent maturation (significant expression of CD83, data not shown) upon LPS stimulation. The data implied that MSCs were capable of suppressing DC generation by secreting cytokines at a higher ratio and mainly through cell-cell interaction at a lower ratio.
Human MSCs suppressed the initial generation of DCs from monocytes in the transwell chamber system. In the transwell chamber system separating MSCs from monocytes, CD14 and CD1a expression was investigated at multiple MSC/monocyte ratios (A) and at an MSC/monocyte ratio of 1:10 when supplemented with anti-IL-6 and anti-M-CSF neutralizing mAb. (B) 1, control; 2, anti-IL-6 neutralizing mAb; 3, anti-M-CSF neutralizing mAb; 4, anti-IL-6 and anti-M-CSF neutralizing mAb. Results of panel B are shown as mean ± SD from 3 independent experiments (*P < .05, **P < .01).
Human MSCs suppressed the initial generation of DCs from monocytes in the transwell chamber system. In the transwell chamber system separating MSCs from monocytes, CD14 and CD1a expression was investigated at multiple MSC/monocyte ratios (A) and at an MSC/monocyte ratio of 1:10 when supplemented with anti-IL-6 and anti-M-CSF neutralizing mAb. (B) 1, control; 2, anti-IL-6 neutralizing mAb; 3, anti-M-CSF neutralizing mAb; 4, anti-IL-6 and anti-M-CSF neutralizing mAb. Results of panel B are shown as mean ± SD from 3 independent experiments (*P < .05, **P < .01).
Human MSCs inhibited the initial yield of DCs from monocytes reversibly. Using the transwell chamber system separating monocytes (top row) from MSCs (bottom row), monocytes that have been cultured with GM-CSF plus IL-4 for 7 days in the presence of MSCs (at a ratio of 1:10) were collected and then induced by GM-CSF, IL-4, and LPS for 7 days. Percentage of cells positive for the indicated markers is shown. Results are representative of 3 independent experiments. Horizontal bars indicate the positive region; the corresponding percentage is shown above each bar.
Human MSCs inhibited the initial yield of DCs from monocytes reversibly. Using the transwell chamber system separating monocytes (top row) from MSCs (bottom row), monocytes that have been cultured with GM-CSF plus IL-4 for 7 days in the presence of MSCs (at a ratio of 1:10) were collected and then induced by GM-CSF, IL-4, and LPS for 7 days. Percentage of cells positive for the indicated markers is shown. Results are representative of 3 independent experiments. Horizontal bars indicate the positive region; the corresponding percentage is shown above each bar.
Next we defined what kind of factors in the supernatant contributed to the effect observed in the transwell system. Here, IL-6 and M-CSF were investigated because they are essential factors in the molecular control of antigen-presenting cell development. Actually, human MSCs did produce IL-6 (1.2 ng/mL per 2 × 105 MSCs) and M-CSF (2.6 ng/mL per 2 × 105 MSCs) as determined by ELISA. As presented in Figure 2B, the CD14+ population was reduced by supplement of anti-IL-6 and anti-M-CSF neutralizing mAb respectively or combined, but increased expression of CD1a was not observed. The data suggested that MSCs might maintain the CD14 immunophenotype of monocytes/macrophages via secretion of IL-6 and M-CSF, but the cytokines counteracting DC-specific CD1a induction by GM-CSF/IL-4 deserve more investigation.
The inhibitory effect in the MSC/monocyte coculture was reversible
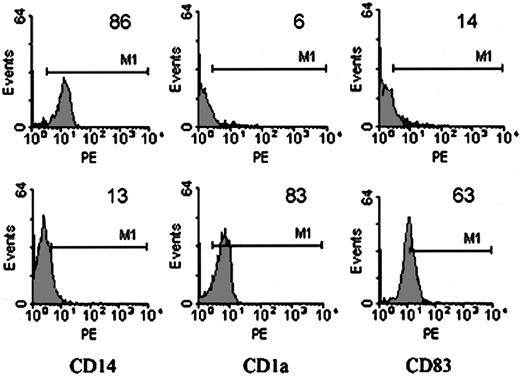
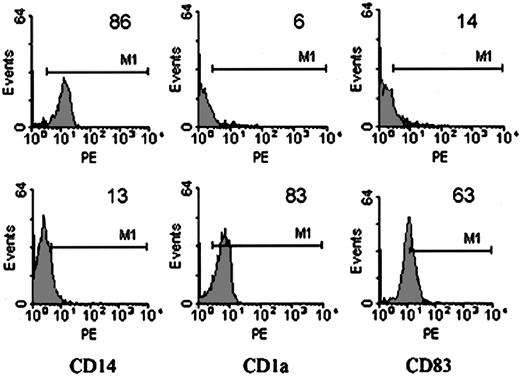
Monocytes are precursors of macrophages and DCs, with each cell type convertible to each other. The environment and the presence of differentiation or other stimulatory signals are the decisive factors.32 Then we studied whether the effect of MSCs on the initial differentiation of DCs from monocytes was persistent or reversible. MSC-Mo's were harvested, washed intensely, and then incubated with GM-CSF, IL-4, and LPS in the absence of MSCs for another 7 days. Although prolonged culture induced cell loss, differentiation and maturation of DCs under these conditions occurred. The down-regulation of CD14 (MSC-Mo versus treated MSC-Mo: 86 versus 13), induction of CD1a (MSC-Mo versus treated MSC-Mo: 6 versus 83), and acquisition of CD83 (MSC-Mo versus treated MSC-Mo: 14 versus 63), as shown in Figure 3, indicated that the suppressive effect in the MSC/monocyte coculture was not refractory but reversible.
Moderate suppression of MSCs on mature DCs
It was important to evaluate whether MSCs, in addition to blocking initial commitment of monocytes to DCs, also affected already differentiated DCs. For this purpose, already differentiated DCs were classified into 2 categories: immature and mature DCs. The former was obtained by culture of monocytes in the presence of GM-CSF and IL-4 for 7 days and the latter under prolonged 2 days of incubation with LPS. As presented in Table 1, treatment of immature DCs with MSCs, though supplemented by GM-CSF, IL-4, and LPS, caused modest up-regulation of membrane CD14 (2.9% ± 0.8% versus 10.9% ± 3.0%) but slight reduction of maturation antigen CD83 (78% ± 12.7% versus 55% ± 15.6%). However, expression of presentation and costimulatory molecules were not altered. Surprisingly, although CD1a and CD83 expression of MSC-treated immature DCs implied increased arrestment of immature DCs, endocytic activity was significantly reduced (MFI of FITC-dextran: 63.5 ± 7.8 versus 33.5 ± 17.7), pointing to a possible inhibition of DC endocytosis by MSCs. The data suggested that some immature DCs might either convert to CD14+ cells (most probably macrophages) or cease to develop into mature DCs.
On the other hand, exposure of mature DCs to MSCs (MSC-mDC), without exogenous cytokines anymore, caused a significantly decreased expression of HLA-DR, CD1a, CD80, and CD86, whereas the CD14 expression and endocytic activity almost remained unchanged. These data implied that MSC coculture could reverse mDCs to an immature state other than to CD14+ monocytes/macrophages.
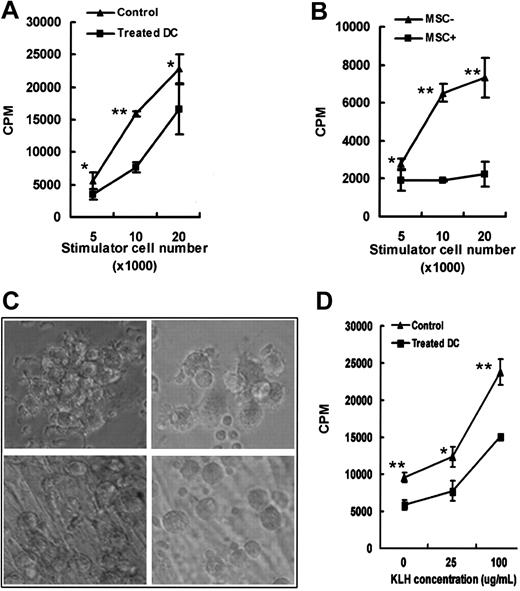
Immunostimulation of allogeneic T-cell proliferation by mature DCs (mDCs) was impaired by MSC treatment. (A) After MSC treatment, mDCs alone were used in graded doses to stimulate allogeneic CD4+ T cells (*P < .05, **P < .01). ▪ indicates treated DCs; ▴, control cells. (B) Graded doses of treated mDCs were used to stimulate allogeneic CD4+ T cells (1 × 105 cells per well) without MSCs retrieved in the MLR culture (*P < .05, **P < .01). ▴ indicates without MSCs; ▪, with MSCs. (C) Notably, untreated mDCs show clustered and protruding characteristics (top row) in the MLR, while treated mDCs are scattered and round (bottom row) (for both rows, original magnification × 100). Images in panel C were visualized using a Nikon TE2000-U microscope equipped with a Plan Fluor 10×/0.30 objective lens (Nikon). Images were captured and processed as for Figure 1A. (D) After MSC treatment, mDCs loaded with KLH were used in graded doses to stimulate allogeneic CD4+ T cells (*P < .05, **P < .01). Symbols are as in panel A. Thymidine incorporation was measured on day 4 by a 16-hour pulse with 1 μCi 3H-thymidine. (A-B,D) Results are shown as mean ± SD of triplicate values. Cell morphology was determined by phase contrast microscopy. One representative experiment from 3 independent experiments is shown.
Immunostimulation of allogeneic T-cell proliferation by mature DCs (mDCs) was impaired by MSC treatment. (A) After MSC treatment, mDCs alone were used in graded doses to stimulate allogeneic CD4+ T cells (*P < .05, **P < .01). ▪ indicates treated DCs; ▴, control cells. (B) Graded doses of treated mDCs were used to stimulate allogeneic CD4+ T cells (1 × 105 cells per well) without MSCs retrieved in the MLR culture (*P < .05, **P < .01). ▴ indicates without MSCs; ▪, with MSCs. (C) Notably, untreated mDCs show clustered and protruding characteristics (top row) in the MLR, while treated mDCs are scattered and round (bottom row) (for both rows, original magnification × 100). Images in panel C were visualized using a Nikon TE2000-U microscope equipped with a Plan Fluor 10×/0.30 objective lens (Nikon). Images were captured and processed as for Figure 1A. (D) After MSC treatment, mDCs loaded with KLH were used in graded doses to stimulate allogeneic CD4+ T cells (*P < .05, **P < .01). Symbols are as in panel A. Thymidine incorporation was measured on day 4 by a 16-hour pulse with 1 μCi 3H-thymidine. (A-B,D) Results are shown as mean ± SD of triplicate values. Cell morphology was determined by phase contrast microscopy. One representative experiment from 3 independent experiments is shown.
Because HLA-DR, CD1a, and costimulatory molecules CD80 and CD86 play an important role in mounting an immune response, we wanted to know if the change of immunophenotype reflected the impairment in the stimulatory activity of MSC-treated mature DCs. To avoid contamination of MSCs in collecting DCs, the transwell system was then used in compliance with aforesaid materials and methods. Compared with mature DCs, there was a 1-fold to 1.5-fold reduction (P < .05) of MSC-mDC-mediated proliferation of allogeneic CD4+ T cells (Figure 4A). Because MSCs have a suppressive effect on T cells, a more significant inhibition (up to 1.5- to 3-fold) was observed in the mixed leukocyte reaction (MLR) without retrieving MSCs (Figure 4B). Interestingly, the morphology of MSC-mDCs in the MLR did change (Figure 4C), becoming round and large rather than veiled. Next, we tested antigen-specific T-cell response induced by DCs loaded with KLH. As shown in Figure 4D, MSC-treated mature DCs showed lower antigen presentation ability. The data suggested that, unlike fibroblasts, MSCs could moderately suppress the function of mature DCs.30
MSCs suppressed IL-12 production by DCs
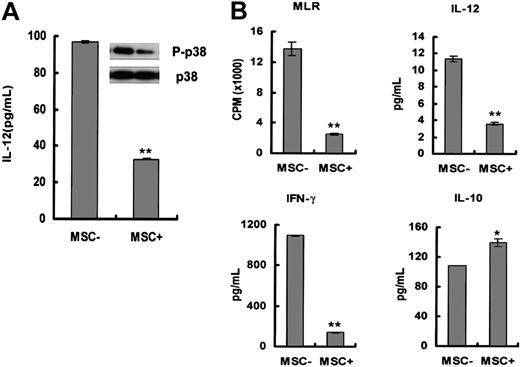
Because the secretion of IL-12 is critical for the maturation and function of DCs, we next investigated whether MSCs could interfere with IL-12 production by DCs. Immature DCs were washed and stimulated by LPS with or without the presence of MSCs for additional 48 hours, followed by supernatant collection for quantification of IL-12. As shown in Figure 4A, IL-12 production was significantly decreased when MSCs were present (96.5 ± 0.65 versus 33.5 ± 0.56 pg per 1 × 106 cells per mL, P < .01). LPS stimulation is reported to be able to induce phosphorylation of p38, an intracellular signaling pathway postulated important in the positive control of IL-12 secretion. Figure 5A shows that the phosphorylation of p38 under LPS stimulation was greatly reduced in the presence of MSCs.
Because insufficient IL-12 production of DCs has been implicated in the induction of anergy and tolerance of T cells,33 IFN-γ and IL-10 secretion in the MLR was quantified by ELISA to confirm the event. Notably, IL-12 secretion was also down-regulated in the MLR with MSC coculture (11.4 ± 0.4 versus 3.6 ± 0.1 pg/mL, P < .01). As shown in Figure 5B, CD4+ T cells (2 × 105 per well) stimulated by MSCs/DCs (1 × 105 per well) showed reduced IFN-γ secretion (1091.9 ± 7.3 versus 137.5 ± 0.7 pg/mL, P < .01) but increased IL-10 production (108.7 ± 0.2 versus 139.4 ± 5.5 pg/mL, P < .05), correlating well with impaired proliferation of CD4+ T cells (Figure 5B, MLR: 13 776 ± 900 cpm versus 2459 ± 127 cpm, P < .01).
Discussion
It is well known that MSCs can inhibit T-cell proliferation induced by allogeneic cells in vitro and mediate a systemic immunosuppressive activity in vivo.25-27 In this report, we showed that human MSCs, the primitive cell type with multilineage differentiation ability, inhibited strongly but reversibly the initial differentiation of CD14+ monocytes into CD1a+ DCs via either intercellular contact or soluble factors. In the case of mature DCs, MSCs were able to down-regulate expression of costimulatory molecules CD80 and CD86 and decrease secretion of IL-12, thus suppressing T-cell activation and proliferation. Hence, modulation of DC differentiation and function is relevant to the immunosuppressive effect of MSCs.
MSC coculture reduced IL-12 secretion by DCs. (A) DCs obtained from monocytes after 7 days of induction with GM-CSF plus IL-4 were stimulated by LPS for an additional 48 hours with or without MSC coculture, and then cell-free supernatants were collected and quantified by ELISA for IL-12 production. In the meantime, the cell lysates were blotted with anti-p38 antibody (bottom row) and antiphosphor-p38 antibody (top row). (B) Mature DCs with (MSC+) or without (MSC-) MSC coculture were used to stimulate allogeneic CD4+ T cells. In the MLR, IL-12, IFN-γ, and IL-10 secretion were quantified by Quantikine ELISA. Thymidine incorporation is expressed as mean ± SD of triplicates. Results are representative of 3 independent experiments (*P < .05; **P < .01).
MSC coculture reduced IL-12 secretion by DCs. (A) DCs obtained from monocytes after 7 days of induction with GM-CSF plus IL-4 were stimulated by LPS for an additional 48 hours with or without MSC coculture, and then cell-free supernatants were collected and quantified by ELISA for IL-12 production. In the meantime, the cell lysates were blotted with anti-p38 antibody (bottom row) and antiphosphor-p38 antibody (top row). (B) Mature DCs with (MSC+) or without (MSC-) MSC coculture were used to stimulate allogeneic CD4+ T cells. In the MLR, IL-12, IFN-γ, and IL-10 secretion were quantified by Quantikine ELISA. Thymidine incorporation is expressed as mean ± SD of triplicates. Results are representative of 3 independent experiments (*P < .05; **P < .01).
Bipotential monocytes can give rise to either antigen-presenting DCs or scavenging macrophages when crossing endothelium in vivo, and similar plasticity has been recapitulated in vitro by feeding them with GM-CSF/IL-4 or M-CSF. Like fibroblast and epithelial cell lines identified before,30 MSC-treated monocytes under GM-CSF/IL-4 stimuli showed a larger cell size with numerous vacuoles and displayed low expression of CD1a and persistently high expression of CD14, indicating a skewing from DCs to macrophages. Nevertheless, human MSCs investigated here revealed several unique properties. First, the inhibition was more powerful. The MSC/monocyte ratio that was required for complete suppression of DC commitment was far lower (1:20 and even 1:40) than that of normal skin fibroblast (1:4). Addition of TNF-α,31 a facilitator of DC induction by overriding the IL-6/M-CSF pathway, failed to alter the switch by MSCs. Second, in contrast to fibroblast, MSCs, particularly at a higher ratio (1:10), could reproduce full skewing without cell-cell contact in the transwell system. By comparative analysis of the typical coculture and transwell system, the relative contribution of intercellular contact or supernatant factors could be estimated. Whatever culture methods, a higher MSC/monocyte ratio (1:10) could completely inhibit monocyte differentiation to DCs, thus at least implying the suppression in the coculture system was saturated. As the ratio decreased to 1:20 and 1:40 (data not shown), the inhibitory effect within the transwell system dropped dramatically, but coculture could still fully reserve a typical monocyte/macrophage immunophenotype. Hence, at a lower ratio, the effect of intercellular contact prevailed. Among cytokines proven to exercise a negative control of DC generation from monocytes, IL-6 and M-CSF, rather than IL-10 34 and IFN-γ,35 were indeed produced by human MSCs. Neutralizing their bioactivity, however, failed to generate CD1a+ cells with a unique veiled appearance. As expected, CD14 expression was reduced upon neutralization of either IL-6 or M-CSF, implying MSCs could retain the monocyte or macrophage immunophenotype (CD14) via M-CSF or/and IL-6, but the soluble factors through which MSCs offset CD1a+ DC commitment driven by GM-CSF/IL-4 may need further evaluation.
The most prominent advantage of MSCs over some kinds of terminal differentiated cells lies in their plasticity, essential for creating an appropriate environment upon different stimuli. In fact, development of the immune system is closely related to mesenchymal tissues, and their interactions may occur at multiple levels. Undoubtedly, a specialized microenvironment allows yielding of either DCs or macrophages from monocytes. The best paradigm is given by Randolph et al36 showing that DCs arise from monocytes that migrate across endothelium grown on an extracellular matrix whereas macrophages develop from monocytes that remain in the subendothelial matrix. The immune response is strictly controlled, and the primary mechanisms include interactions between different subsets of immune competent cells and the influence of microenvironments. Zhang et al37 have reported that cellular contact with splenic stromal cells promotes mature DCs to proliferate in a fibronectin-dependent way. Furthermore, both intercellular contact and stromal cell-derived transforming growth factor-β could induce their differentiation into a novel regulatory DC subset. It is conceivable that MSCs, the precursors of stromal cells, together with their descendents in varied and successive differentiation status, provide various niches in different tissues. As a further proof, the osteogenic, chondrogenic, and adipogenic lineages differentiated from MSCs do suppress lymphocyte proliferative responses.38
MSCs differed from previously described fibroblast and epithelial cell lines in that the suppression of DC development is continuous. Normally the life of DCs can be identified into 2 major phases39,40 : an immature stage characterized by a high efficiency in taking up and processing antigens and a mature stage in which the antigen uptake capacity is lost and the function shifts to potent APCs41,42 associated with a high expression of molecules involved in antigen presentation and T-cell stimulation, such as MHC II, CD80, CD86, and CD83. Actually, monocyte-derived DCs and macrophages can readily interconvert into one another until the late stages of their respective differentiation/maturation process. We have shown that, upon removal of MSCs, CD14+ macrophages could be reinduced into CD1a+ DCs. Beyond the stage of immature DCs, however, MSCs were unable to efficiently reverse the DCs into CD14+ monocytes/macrophages (less than 10%). The plausible explanation is that the previously reported reconversion is carried out in a cytokine environment supporting macrophage differentiation (withdrawal of GM-CSF/IL-4) while our finding is under a DC maturation condition (GM-CSF/IL-4 plus LPS). With respect to DC maturation, CD83, the maturation antigen of DCs, was reduced by MSC treatment regardless of whether the starting population was either immature or mature DCs. Such alteration implied that the pool of immature DCs was enlarged at the expense of mature DCs. Nevertheless, the change contradicted with decreased or unchanged endocytosis capacity of DCs. The mechanism is most probably related to direct suppression of DC endocytosis by MSCs.
Whether naive T cells diverge into Th1 or Th2 effectors depends on the cytokine environment (IL-12) and the type and the activation state of DCs. Accordingly, insufficient IL-12 production and decreased expression of MHC class II plus costimulatory molecules of DCs upon MSC treatment might modulate the balance between Th1 and Th2 cells in favor of the latter, which showed lower IFN-γ and higher IL-10 production. Although the direct effect of MSCs on T cells may be included in the MLR, the changed morphology of DCs and the decreased secretion of IL-12 suggested a direct effect of MSCs on DCs, which subsequently regulated T-cell expansion.
Abundant studies have been carried out to show that MSCs perform a direct, strong, and reversible suppression on T-cell proliferation in vitro. Such effect occurs regardless of the species of T cells or MSCs and is independent of APCs or CD4+/CD25+ regulatory T cells. In vivo, administration of MSCs leads to prolonged skin graft survival. Lazarus et al43 have reported a phase 1 bone marrow stromal cell (BMSC) study in patients with advanced hematologic malignancies without significant toxicities. In addition, most patients have experienced a reduction of acute and chronic graft versus host disease (GVHD). Whether MSCs or their offspring participate in the regulation of T-cell proliferation via DCs in vivo calls for further investigation. Fortunately, we have successfully isolated and expanded MSCs from murine bone marrow using a novel and reliable method, which paves the way for studying the effect of MSCs on DCs in vivo.44
In summary, our data suggested that MSCs might modulate the immune system, not only acting directly on T cells but also at the very first step of the immune response through the inhibition of DC differentiation and maturation. This study will be clinically beneficial. Because DCs serve as gatekeepers to the body's immune system, it seems likely that by somehow suppressing DC function, be it through the limitation of antigen uptake, processing, or presentation, the likelihood of graft rejection could be significantly reduced. More valuably, the effects of MSCs on DCs are reversible, which will circumvent the flaws of long-lasting hypoimmune competence following transplantation.
Prepublished online as Blood First Edition Paper, February 3, 2005;DOI 10.1182/blood-2004-02-0586.
Supported by grants from National Natural Science Foundation (nos. 30271245 and 30470640), National Basic Research Program of China (no. G1999054302), and High-tech Research and Development Program of China (no. 2003AA205170).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.