Key Points
Generation of an “off-the-shelf” CAR EBV-VST cell bank for use in a phase 1 trial is feasible.
This phase 1 clinical trial demonstrates the safety of an allogeneic “off-the-shelf” CAR EBV-VST product.
Visual Abstract
Despite clinical benefit with the use of chimeric antigen receptor (CAR) T cells, the need to manufacture patient-specific products limits its clinical utility. To overcome this barrier, we developed an allogeneic “off-the-shelf” CAR T-cell product using Epstein-Barr virus (EBV)–specific T cells (EBV-VSTs) genetically modified with a CD19-specific CAR (19-28z). Patients with relapsed/refractory (R/R) B-cell malignancies were stratified into 3 treatment cohorts: cohort 1 (n = 8; disease recurrence after allogeneic or autologous hematopoietic cell transplantation [HCT]), cohort 2 (n = 6; consolidative therapy after autologous HCT), or cohort 3 (n = 2; consolidative therapy after allogeneic HCT). The primary objective of this trial was to determine the safety of multiple CAR EBV-VST infusions. Most patients (n = 12/16) received multiple doses (overall median, 2.5 [range, 1-3]) with 3 × 106 T cells per kg determined to be the optimal dose enabling multiple treatments per manufactured cell line. Severe cytokine release syndrome or neurotoxicity did not occur after infusion, and no dose-limiting toxicity was observed in the trial. Median follow-up was 48 months (range, 4-135) with 4 deaths due to disease progression. Overall survival of all patients was 81% at 12 months and 75% at 36 months. Postinfusion expansion and persistence were limited, and CAR EBV-VSTs demonstrated a unique T-cell phenotype compared with autologous 19-28z CAR T cells. Our study demonstrates the feasibility and safety of an allogeneic “off-the-shelf” CAR EBV-VST product with favorable outcomes for patients with CD19+ R/R B-cell malignancies. This trial was registered at www.ClinicalTrials.gov as #NCT01430390.
Introduction
Patients with relapsed/refractory (R/R) B-cell malignancies have a poor prognosis, even after autologous hematopoietic cell transplantation (HCT; auto-HCT) or allogeneic HCT (allo-HCT).1-6 The use of T cells genetically modified to express a chimeric antigen receptor (CAR) targeting tumor-specific antigens has shown efficacy in treating chemotherapy-refractory malignancies.7 Our group has previously published on the development, use, and clinical validation of CD19-specific, second-generation (CD28-containing), autologous CAR T cells (19-28z CAR T cells) in several patient populations.8-13 Several other groups have also reported similar success leading to US Food and Drug Administration approval of several CD19-specific CAR T-cell products for these disease indications.1,8,10,14-28
Despite clinical success, the applicability and broad usage of autologous CAR T-cell products are limited by several major barriers.29-31 The need for patient-specific collection and manufacturing in patients with active malignancies is logistically complex and time consuming. Even with improvements with rapid production platforms, the inability to have a readily available cellular therapy product results in disease progression, need for toxic and potentially ineffective bridging therapy, and, in some cases, loss of adequate performance status and/or organ dysfunction rendering the patient ineligible for treatment with cellular therapy products. Although commercial products have been available for several years, their use is limited due to the complexities of collection, manufacturing availability/success, and capacity constraints of treating centers. The variability in starting material with patient-specific manufacturing results in wide variation/nonhomogeneity in product characteristics, T-cell fitness, incidence of toxicity, and, most importantly, long-term effectiveness of cellular therapy products. Poor CAR T-cell persistence has been associated with inferior outcomes.32 Multidose cellular therapies could address this, but current technology limits multidose production for autologous products.
An “off-the-shelf” allogeneic CAR T-cell approach, derived from healthy donors and stored in large batches, may overcome many of these limitations.33-36 Utilization of healthy donors may confer a more uniform starting material, allowing for more predictable manufacturing and performance of generated cell products, as well as T-cell fitness benefit.37 Allogeneic therapies are readily available and provide an “on-demand” cellular therapy that avoids manufacturing delays/failures, need for bridging therapy, or loss of patient’s eligibility due to worsening clinical status. Single manufacturing runs of an allogeneic product have the potential to treat several patients and/or provide multiple doses for individual patients.37 By increasing the scale of production and creating an inventory/bank of manufactured CAR immune cells, cost per patient decreases whereas access to product increases.37 The use of allogeneic cellular therapy approaches is tempered by the significant morbidity and mortality from graft-versus-host disease (GVHD) if the infusion contains alloreactive T cells and/or the need for significant immunosuppression to enable persistence of a donor cell therapy product.38 The use of autologous and allogeneic CAR Epstein-Barr virus (EBV)–specific T cells (CAR EBV-VSTs) has been previously reported in the setting of R/R neuroblastoma and B-cell malignancies after HCT, respectively.35,39,40 Herein, we report the safe and feasible use of an allogeneic “off-the-shelf” CAR EBV-VST product for patients with R/R B-cell malignancies, including expanding the use of this product as a consolidative therapy after auto-HCT.
Patients and methods
Trial design and oversight
We conducted a single-center phase 1 trial using in vitro expanded allogeneic EBV-VSTs genetically modified with a CD19-specific second-generation (CD28-containing) CAR (19-28z CAR EBV-VSTs) in patients with R/R B-cell malignancies. All clinical investigation was conducted according to the Declaration of Helsinki principles. An informed consent was obtained from all study participants, donors, or their respective legal guardians. Patients received CAR EBV-VST infusions between October 2011 and October 2021. Data cutoff for evaluation of outcomes was 14 February 2024. The primary objective of the study was to determine the safety of allogeneic CAR EBV-VST infusions. Secondary objectives were to determine the efficacy of adoptively transferred CD19-specific T cells on CD19+ malignancy, quantitate the number of CAR+ T cells in the blood at defined intervals after infusion, assess the long-term status of treated patients, assess the feasibility of generating multiple doses of CAR EBV-VSTs from donor EBV-VST cell lines, and describe hematopoietic recovery after auto-HCT or allo-HCT and CAR EBV-VSTs. A minimum sample size of 6 patients per cohort was planned to address the primary objective of safety. If no more than 1 dose-limiting toxicity (DLT) in 6 patients was observed, the treatment was deemed safe for further investigation.
The protocol was approved by Memorial Sloan Kettering Cancer Center’s (MSKCC) institutional review board and registered at ClinicalTrials.gov (identifier: NCT01430390).
Eligibility for this trial included patients of any age with CD19+ R/R B-cell malignancies stratified into 3 treatment cohorts: cohort 1 (disease recurrence after allo-HCT or auto-HCT with any disease status eligible for infusion including no detectable disease), cohort 2 (consolidative therapy after auto-HCT), or cohort 3 (consolidative therapy after allo-HCT). Previous treatment with CAR T cells was not an exclusion criterion in this trial.
Manufacturing (including characterization of virus specificity, lack of alloreactivity, and HLA restriction of the EBV specificity) and clinical use of EBV-VSTs in patients with EBV+ lymphomas have been previously described.41,42 Generation of CAR EBV-VSTs occurred within the MSKCC Cell Therapy and Cell Engineering Facility. A minimum of 2/10 HLA matching was required between recipient and donor CAR EBV-VSTs.
Lymphodepleting chemotherapy (cohort 1) or HCT conditioning chemotherapy (cohorts 2 and 3) was used for all patients. A cyclophosphamide-based chemotherapy regimen was the preferred conditioning chemotherapy before CAR T-cell infusion for patients with CD19+ malignancy in cohort 1. Lymphodepletion and conditioning were selected by the treating physician based on previous therapy, previous adverse reactions, and the highest likelihood of achieving an optimal response. Two to 14 days after completion of conditioning chemotherapy (cohort 1) or up to 120 days after infusion of autologous/allogeneic hematopoietic stem cells (cohorts 2 and 3), patients received their initial intravenous infusion of CAR EBV-VSTs at the protocol-specified dose. All patients could receive cycle 2 ∼7 to 30 days after cycle 1 and cycle 3 ∼7 to 30 days after cycle 2 if additional doses were available and severe/irreversible toxicities had resolved.
Toxicity assessment
Cytokine release syndrome (CRS) was graded according to American Society for Transplantation and Cellular Therapy (ASTCT) consensus grading.43,44 Severe CRS was defined as grade ≥3. Neurotoxicity was assessed according to National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, and severe neurotoxicity was defined as any seizure or grade ≥3 toxicity of the nervous system (adoption of ASTCT immune effector cell–associated neurotoxicity syndrome [ICANS] consensus grading was not standard institutional practice during conduct of this study and is therefore not available). Acute GVHD was assessed and graded using standard clinical criteria as defined by MSKCC, which are based on the National Marrow Donor Program GVHD consensus panel.45,46 Adverse events were captured for all treated patients until disease relapse, alternative therapy was given, or death. DLTs were defined as new grade 3 or 4 nonhematologic toxicities lasting ≥14 days that were probably or definitively attributed to T-cell infusion, grade 4 neutropenia lasting ≥30 days from the time of conditioning therapy (cohort 1) or autologous/allogeneic HCT (cohorts 2 and 3; in patients with a pretreatment absolute neutrophil count >0.5 × 109/L) unless due to persistent disease or explained from previous treatment, grade 3 or 4 thrombocytopenia that failed to recover to grade ≤2 at 30 days after conditioning chemotherapy (cohort 1) or autologous HCT (cohort 2; in patients with a pretreatment platelet count of >50 000/μL) unless due to persistent disease, grade 3 infusion-related reactions that did not resolve with treatment or grade 4 infusion-related reaction as defined by CTCAE, grade 4 neurotoxicity, grade 4 CRS, or severe CRS not improving after 24 hours of systemic steroids, grade 3 or 4 GVHD after treatment requiring systemic immunosuppression, grade 4 seizure as defined by CTCAE 4.0, or any grade 5 toxicity attributable (definitely, probably, or possibly) to T-cell infusion occurring within 30 days of the final infusion.
Response assessment for leukemia/lymphoma
Responses were assessed as complete remission (CR) or partial response (PR), stable disease, or progressive disease by standard laboratory and pathologic criteria.47 See supplemental Methods for detailed information on response assessment.
Assessment of 19-28z CAR T-cell expansion
Preclinical validation experiments
CAR EBV-VST manufacturing process was developed in the Cell Therapy and Cell Engineering Facility at MSKCC. Preclinical experiments were conducted to establish optimal CAR EBV-VST stimulation, expansion, and transduction. Please refer to supplemental Methods for additional information on preclinical validation.
Statistical analysis of patient clinical end points
Overall survival (OS) was defined as the time from the first dose of CAR EBV-VSTs to death from any cause. Patients alive were censored at their date of last follow-up. Event-free survival (EFS) was defined as the time from the first dose of CAR EBV-VSTs to the first event among nonresponse, relapse, and death from any cause. Patients alive without event were censored at their last follow-up. Nonresponders were counted as having an event on day 0. Survival rates were estimated using Kaplan-Meier estimator. Median follow-up was estimated using the reverse Kaplan-Meier estimator.49 All analyses were done in R version 4.2.1.
Results
Generation of an allogeneic CAR EBV-VST bank
Donor EBV-VST cell lines were generated as previously described.41 Previously manufactured and cryopreserved EBV-VST cell lines were used as starting material for CAR EBV-VST generation. EBV-VST cell lines were selected based on cell number (minimum of 1 × 108 T cells available for transduction) and expression of specific HLA alleles. This included EBV-VST cell lines being homozygous for all 5 HLA alleles (HLA-A, HLA-B, HLA-C, HLA-DR, HLA-DQ; n = 3), for the alleles through which they recognized EBV (n = 2), or for providing HLA compatibility of highly prevalent HLA alleles (n = 9; including EBV-VSTs sharing ≥2 of 12 prevalent HLA-A alleles, 18 prevalent HLA-B alleles, 19 prevalent HLA-C alleles, 15 prevalent HLA-DR alleles, and/or 12 prevalent HLA-DQ alleles). An additional homozygous line was selected based on previously consistently mediating responses when used to treat EBV posttransplant lymphoproliferative disease emerging after hematopoietic or solid organ transplant.
We optimized conditions for stimulation and transduction with our institutional CD19-specific CAR (Figure 1). Transduction efficiencies were measured by flow cytometry (supplemental Figure 1A). Gammaretroviral transduction on days 2 and 3 after the third weekly stimulation with EBV antigen (EBV B lymphoblastoid cell line [B-LCL]) showed higher efficiency than on days 1 and 2 (supplemental Figure 1B). The highest transduction efficiency was observed at the lowest cell density of 2.5 × 10⁵ cells per mL (supplemental Figure 1C). Optimal initial and subsequent stimulation of EBV-VSTs for growth, viability, and transduction efficiency was evaluated demonstrating that cells stimulated with B-LCL and restimulated with B-LCL had the highest proliferation over 15 days in culture (supplemental Figure 1D). Finally, interleukin-2 (IL-2) supplementation alone (compared with IL-2 + IL-15) resulted in higher cytotoxicity of CAR EBV-VSTs (data not shown). Cytotoxicity of CAR EBV-VSTs compared with EBV-VSTs was augmented against Raji and NALM6 at various effector-to-target ratios (supplemental Figure 1E). The final process was validated in vivo (supplemental Figure 1F-G), and a CAR EBV-VST cell bank of 17 cell lines manufactured from 15 healthy donors was created (supplemental Table 2).
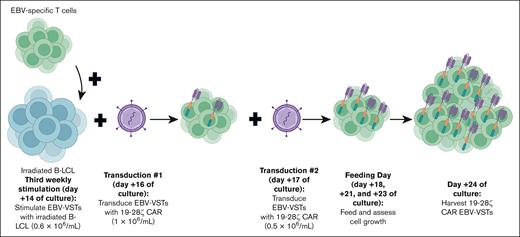
Process development for generating 19-28z CAR EBV-VSTs. Cryopreserved EBV-VSTs (cultured for 28-35 days before cryopreservation) are thawed and stimulated weekly with irradiated B-LCL (days 0, +7, +14). T cells are transduced with 19-28z CAR at a cell density of 1 × 106 cells per mL and 0.5 × 106 cells per mL, respectively, on days +16 and +17. Cells are then fed and evaluated on days +18, +21, and +23 and then harvested and frozen on day +24.
Process development for generating 19-28z CAR EBV-VSTs. Cryopreserved EBV-VSTs (cultured for 28-35 days before cryopreservation) are thawed and stimulated weekly with irradiated B-LCL (days 0, +7, +14). T cells are transduced with 19-28z CAR at a cell density of 1 × 106 cells per mL and 0.5 × 106 cells per mL, respectively, on days +16 and +17. Cells are then fed and evaluated on days +18, +21, and +23 and then harvested and frozen on day +24.
Patients
Sixteen patients were treated with CAR EBV-VSTs: 8 in cohort 1 (B-cell acute lymphoblastic leukemia [B-ALL], n = 6; Burkitt lymphoma, n = 1; chronic lymphocytic leukemia, n = 1), 6 in cohort 2 (primary mediastinal large B-cell lymphoma, n = 2; diffuse large B-cell lymphoma, n = 3; marginal zone lymphoma n = 1), and 2 in cohort 3 (diffuse large B-cell lymphoma, n = 2; Figure 2). Baseline characteristics of patients who received treatment with CAR EBV-VSTs are presented in Table 1. Median age at treatment was 30 years (range, 1-71). All patients were heavily pretreated, having received a median of 5 lines of therapy (including any HCT; range, 3-11) before CAR EBV-VSTs. Lymphodepletion regimens/pretransplant conditioning chemotherapy regimens are presented in Table 2. Hematopoietic recovery (neutrophil and platelet engraftment) after transplant and CAR EBV-VSTs was as expected for patients treated in cohorts 2 and 3.
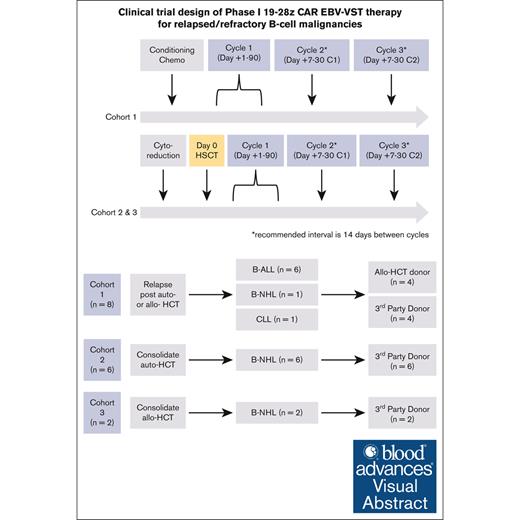
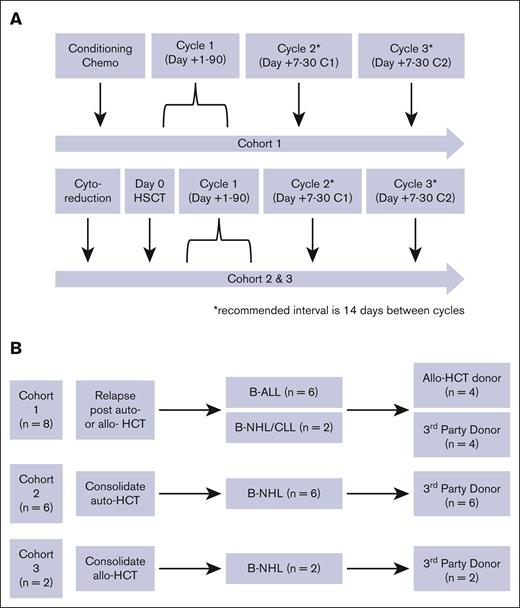
19-28z CAR EBV-VST clinical trial design. (A) Patients in cohort 1 were treated with 1 to 3 cycles of 19-28z CAR EBV-VSTs after relapse after transplant, and patients in cohorts 2 and 3 were treated with 1 to 3 cycles of 19-28z CAR EBV-VSTs as consolidation with hematopoietic cell transplant. (B) Schema showing patients in each cohort along with their diagnosis and transplant donor type. B-NHL, B-cell non-Hodgkin lymphoma; CLL, chronic lymphocytic leukemia.
19-28z CAR EBV-VST clinical trial design. (A) Patients in cohort 1 were treated with 1 to 3 cycles of 19-28z CAR EBV-VSTs after relapse after transplant, and patients in cohorts 2 and 3 were treated with 1 to 3 cycles of 19-28z CAR EBV-VSTs as consolidation with hematopoietic cell transplant. (B) Schema showing patients in each cohort along with their diagnosis and transplant donor type. B-NHL, B-cell non-Hodgkin lymphoma; CLL, chronic lymphocytic leukemia.
Baseline characteristics of 16 patients treated with 19-28z CAR EBV-VSTs
| Characteristic . | Cohort 1, n = 8 . | Cohort 2, n = 6 . | Cohort 3, n = 2 . | Overall, N = 16 . |
|---|---|---|---|---|
| Disease, n (%) | ||||
| B-ALL | 6 (75) | 0 (0) | 0 (0) | 6 (38) |
| NHL | 2 (25) | 6 (100) | 2 (100) | 10 (62) |
| Burkitt lymphoma | 1 | 0 | 0 | 1 (6) |
| DLBCL | 0 | 3 | 1 (50) | 4 (25) |
| PMBCL | 0 | 2 | 0 | 2 (12) |
| Marginal zone lymphoma | 0 | 1 | 0 | 1 (6) |
| SLL/CLL --> DLBCL | 0 | 0 | 1 (50) | 1 (6) |
| CLL | 1 | 0 | 0 | 1 (6) |
| Age at treatment (day 0), median (range), y | 13 (1-68) | 62 (17-71) | 49 (48-51) | 30 (1-71) |
| Median prior lines therapy, median (range) | 5 (4-11) | 3.5 (3-6) | 10; 10 | 5 (3-11) |
| Cell dose, n (%) | ||||
| 1 × 106 T cells per kg | 5 (62) | 0 (0) | 0 (0) | 5 (31) |
| 3 × 106 T cells per kg | 2 (25) | 6 (100) | 2 (100) | 10 (62) |
| 1 × 107 T cells per kg | 1 (12) | 0 (0) | 0 (0) | 1 (6) |
| Number of cycles, n (%) | ||||
| 1 | 3 (38) | 1 (17) | 0 (0) | 4 (25) |
| 2 | 3 (38) | 1 (17) | 0 (0) | 4 (25) |
| 3 | 2 (25) | 4 (67) | 2 (100) | 8 (50) |
| CAR EBV donor, n (%) | ||||
| Third party | 4 (50) | 6 (100) | 2 (100) | 12 (75) |
| HCT donor (sibling) | 2 (25) | 0 (0) | 0 (0) | 2 (12) |
| HCT donor (URD) | 2 (25) | 0 (0) | 0 (0) | 2 (12) |
| HLA matching, n (%) | ||||
| 10/10 | 3 (38) | 0 (0) | 0 (0) | 3 (19) |
| 6/10 | 1 (12) | 1 (17) | 0 (0) | 2 (12) |
| 5/10 | 0 (0) | 1 (17) | 0 (0) | 1 (6) |
| 4/10 | 2 (25) | 1 (17) | 0 (0) | 3 (19) |
| 2/10 | 2 (25) | 3 (50) | 2 (100) | 7 (44) |
| HCT to CAR EBV-VST, median (range), d | 393.5 (99-1128) | 21 (14-60) | 34; 89 | 94 (14-1128) |
| Characteristic . | Cohort 1, n = 8 . | Cohort 2, n = 6 . | Cohort 3, n = 2 . | Overall, N = 16 . |
|---|---|---|---|---|
| Disease, n (%) | ||||
| B-ALL | 6 (75) | 0 (0) | 0 (0) | 6 (38) |
| NHL | 2 (25) | 6 (100) | 2 (100) | 10 (62) |
| Burkitt lymphoma | 1 | 0 | 0 | 1 (6) |
| DLBCL | 0 | 3 | 1 (50) | 4 (25) |
| PMBCL | 0 | 2 | 0 | 2 (12) |
| Marginal zone lymphoma | 0 | 1 | 0 | 1 (6) |
| SLL/CLL --> DLBCL | 0 | 0 | 1 (50) | 1 (6) |
| CLL | 1 | 0 | 0 | 1 (6) |
| Age at treatment (day 0), median (range), y | 13 (1-68) | 62 (17-71) | 49 (48-51) | 30 (1-71) |
| Median prior lines therapy, median (range) | 5 (4-11) | 3.5 (3-6) | 10; 10 | 5 (3-11) |
| Cell dose, n (%) | ||||
| 1 × 106 T cells per kg | 5 (62) | 0 (0) | 0 (0) | 5 (31) |
| 3 × 106 T cells per kg | 2 (25) | 6 (100) | 2 (100) | 10 (62) |
| 1 × 107 T cells per kg | 1 (12) | 0 (0) | 0 (0) | 1 (6) |
| Number of cycles, n (%) | ||||
| 1 | 3 (38) | 1 (17) | 0 (0) | 4 (25) |
| 2 | 3 (38) | 1 (17) | 0 (0) | 4 (25) |
| 3 | 2 (25) | 4 (67) | 2 (100) | 8 (50) |
| CAR EBV donor, n (%) | ||||
| Third party | 4 (50) | 6 (100) | 2 (100) | 12 (75) |
| HCT donor (sibling) | 2 (25) | 0 (0) | 0 (0) | 2 (12) |
| HCT donor (URD) | 2 (25) | 0 (0) | 0 (0) | 2 (12) |
| HLA matching, n (%) | ||||
| 10/10 | 3 (38) | 0 (0) | 0 (0) | 3 (19) |
| 6/10 | 1 (12) | 1 (17) | 0 (0) | 2 (12) |
| 5/10 | 0 (0) | 1 (17) | 0 (0) | 1 (6) |
| 4/10 | 2 (25) | 1 (17) | 0 (0) | 3 (19) |
| 2/10 | 2 (25) | 3 (50) | 2 (100) | 7 (44) |
| HCT to CAR EBV-VST, median (range), d | 393.5 (99-1128) | 21 (14-60) | 34; 89 | 94 (14-1128) |
-->, initial to subsequent diagnosis; CLL, chronic lymphocytic leukemia; DLBCL, diffuse large B-cell lymphoma; NHL, non-Hodgkin lymphoma; PMBCL, primary mediastinal large B-cell lymphoma; SLL, small lymphocytic lymphoma; URD, unrelated donor.
Outcomes for 16 patients treated with 19-28z CAR EBV-VSTs
| Patient . | Disease . | Target T-cell dose . | LDC . | Pretreatment disease burden∗ . | Initial response . | Time to death/last follow-up (mo) . |
|---|---|---|---|---|---|---|
| 1 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Flu 25 mg/m2 × 5 (C1) | Morphologic disease | Morphologic disease | 6.7 |
| 2 | B-ALL | 1 × 106 T cells per kg (actual, 0.9 × 106 T cells per kg) | Flu 25 mg/m2 × 5 | Morphologic disease | Morphologic disease | 4.2 |
| 3 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Flu 25 mg/m2 × 5 (C1); Cy 1800 mg/m2 × 1 (C2 and C3) | MRD+/CR | N/E | 135+ |
| 4 | NHL | 1 × 106 T cells per kg (actual, 0.6 × 106 T cells per kg) | Flu 20 mg/m2 × 4; clofarabine 20 mg/m2 × 4 (2 wk prior) | MRD+/CR | N/E | 123+ |
| 5 | B-ALL | 3 × 106 T cells per kg (actual: 2.2 × 106 T cells per kg) | Cy 900 mg/m2 × 2 | MRD+/CR | N/E | 21.7 |
| 6 | CLL | 3 × 106 T cells per kg (actual, 3.7 × 106 T cells per kg) | Cy 900 mg/m2 × 1; Flu 25 mg/m2 × 3 | MRD+/CR | MRD+/CR | 80.3+ |
| 7 | B-ALL | 1 × 107 T cells per kg (actual, 7.5 × 106 T cells per kg) | Clofarabine 40 mg/m2 × 4; Flu 25 mg/m2 × 4 | Morphologic disease | Morphologic disease | 8.1 |
| 8 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Cy 500 mg/m2 × 2; Flu 30 mg/m2 × 4 | MRD+/CR | MRD–/CR | 32.2+ |
| 9 | NHL | 3 × 106 T cells per kg (actual, 3.2 × 106 T cells per kg) | BEAM | PR | CR | 88.1+ |
| 10 | NHL | 3 × 106 T cells per kg (actual, 2.4 × 106 T cells per kg) | BEAM | PR | CR | 74.6+ |
| 11 | NHL | 3 × 106 T cells per kg (actual, 2.7 × 106 T cells per kg) | BEAM | PR | CR | 53.3+ |
| 12 | NHL | 3×106 T cells per kg (actual, 8.6×106) | BEAM | PR | CR | 39.8+ |
| 13 | NHL | 3 × 106 T cells per kg (actual, 2.8 × 106 T cells per kg) | BEAM | PR | SD | 35.1+ |
| 14 | NHL | 3 × 106 T cells per kg (actual, 3.4 × 106 T cells per kg) | BEAM | PR | CR | 30.4+ |
| 15 | NHL | 3 × 106 T cells per kg (actual, 4.0×106 T cells per kg) | Ritux/Flu/Cy/TBI (200 cGy) | CR | PD | 30.3+ |
| 16 | NHL | 3 × 106 T cells per kg (actual, 3.9 × 106 T cells per kg) | Flu/Mel | PR | CR | 27.7+ |
| Patient . | Disease . | Target T-cell dose . | LDC . | Pretreatment disease burden∗ . | Initial response . | Time to death/last follow-up (mo) . |
|---|---|---|---|---|---|---|
| 1 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Flu 25 mg/m2 × 5 (C1) | Morphologic disease | Morphologic disease | 6.7 |
| 2 | B-ALL | 1 × 106 T cells per kg (actual, 0.9 × 106 T cells per kg) | Flu 25 mg/m2 × 5 | Morphologic disease | Morphologic disease | 4.2 |
| 3 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Flu 25 mg/m2 × 5 (C1); Cy 1800 mg/m2 × 1 (C2 and C3) | MRD+/CR | N/E | 135+ |
| 4 | NHL | 1 × 106 T cells per kg (actual, 0.6 × 106 T cells per kg) | Flu 20 mg/m2 × 4; clofarabine 20 mg/m2 × 4 (2 wk prior) | MRD+/CR | N/E | 123+ |
| 5 | B-ALL | 3 × 106 T cells per kg (actual: 2.2 × 106 T cells per kg) | Cy 900 mg/m2 × 2 | MRD+/CR | N/E | 21.7 |
| 6 | CLL | 3 × 106 T cells per kg (actual, 3.7 × 106 T cells per kg) | Cy 900 mg/m2 × 1; Flu 25 mg/m2 × 3 | MRD+/CR | MRD+/CR | 80.3+ |
| 7 | B-ALL | 1 × 107 T cells per kg (actual, 7.5 × 106 T cells per kg) | Clofarabine 40 mg/m2 × 4; Flu 25 mg/m2 × 4 | Morphologic disease | Morphologic disease | 8.1 |
| 8 | B-ALL | 1 × 106 T cells per kg (actual, 0.8 × 106 T cells per kg) | Cy 500 mg/m2 × 2; Flu 30 mg/m2 × 4 | MRD+/CR | MRD–/CR | 32.2+ |
| 9 | NHL | 3 × 106 T cells per kg (actual, 3.2 × 106 T cells per kg) | BEAM | PR | CR | 88.1+ |
| 10 | NHL | 3 × 106 T cells per kg (actual, 2.4 × 106 T cells per kg) | BEAM | PR | CR | 74.6+ |
| 11 | NHL | 3 × 106 T cells per kg (actual, 2.7 × 106 T cells per kg) | BEAM | PR | CR | 53.3+ |
| 12 | NHL | 3×106 T cells per kg (actual, 8.6×106) | BEAM | PR | CR | 39.8+ |
| 13 | NHL | 3 × 106 T cells per kg (actual, 2.8 × 106 T cells per kg) | BEAM | PR | SD | 35.1+ |
| 14 | NHL | 3 × 106 T cells per kg (actual, 3.4 × 106 T cells per kg) | BEAM | PR | CR | 30.4+ |
| 15 | NHL | 3 × 106 T cells per kg (actual, 4.0×106 T cells per kg) | Ritux/Flu/Cy/TBI (200 cGy) | CR | PD | 30.3+ |
| 16 | NHL | 3 × 106 T cells per kg (actual, 3.9 × 106 T cells per kg) | Flu/Mel | PR | CR | 27.7+ |
BEAM, carmustine, etoposide, cytarabine, melphalan; C1/2/3, cycle 1/2/3; Cy, cyclophosphamide; Flu, fludarabine; LDC, lymphodepleting chemotherapy; Mel, melphalan; N/E, not evaluable; PD, progressive disease; Ritux, rituximab; SD, stable disease; TBI: total body irradiation.
Disease burden evaluation is before BMT conditioning for cohorts 2 and 3.
Dosing on this protocol was based on total T cells per kg with no maximum T-cell dose specified per protocol. The most common target dose infused was 3 × 106 T cells per kg (range, 1 × 106 to 10 × 106 T cells per kg). Twelve patients (n = 12/16 [75%]) received multiple doses (overall median, 2.5 [range, 1-3]) with 3 × 106 T cells per kg determined to be the optimal dose enabling multiple treatments per manufactured cell line. Products had variable transduction efficiencies with a median of 25.6% (range, 7.4%-41.1%), with a median CAR EBV-VST dose of 0.7 × 106/kg (range, 0.05 × 106 to 2.7 × 106/kg). CAR EBV-VST sources were primary HCT donors (n = 4) and third-party donors (n = 12). Ten of the recipients were EBV seropositive, 4 were seronegative, and 2 were serostatus unknown.
Toxicity
CRS occurred in 1 of 16 patients (6%) and neurotoxicity (irritability or headache) occurred in 3 of 16 patients (19%) after infusion. Severe CRS or neurotoxicity did not occur, and no DLT was noted in the trial (Table 3). No patients in cohorts 2 and 3 exhibited graft failure or rejection after treatment with CAR EBV-VSTs. Seven patients developed diffuse skin rash with 3 patients’ rashes deemed to be GVHD (n = 1, HLA-matched HCT donor; n = 2, third-party donor), which resolved for all 3 patients with topical steroids. Other low-grade toxicities are presented in Table 3.
Toxicities for 16 patients treated with 19-28z CAR EBV-VSTs
| Event (CTCAE grade) . | Overall cohort, n (%) . | Third-party donor cohort, n (%) . |
|---|---|---|
| Number | 16 | 12 |
| CRS (grade 1) | 1 (6) | 1 (8) |
| Neurotoxicity | 2 (13) | 2 (13) |
| Irritability (grade 2) | 1 (6) | 1 (8) |
| Headache (grade 1) | 1 (6) | 1 (8) |
| Skin GVHD (grade 1) | 1 (6) | 1 (8) |
| Skin GVHD (grade 2) | 1 (6) | 1 (8) |
| Skin GVHD (grade 3) | 1 (6) | |
| Non-GVHD skin rash (grade 1) | 2 (12) | |
| Non-GVHD skin rash (grade 2) | 2 (12) | 2 (17) |
| Febrile neutropenia (grade 3) | 1 (6) | |
| Pneumonitis (grade 3) | 1 (6) | 1 (8) |
| Bone pain (grade 1) | 1 (6) | 1 (8) |
| Weight loss (grade 1) | 1 (6) | 1 (8) |
| Vomiting (grade 1) | 1 (6) | 1 (8) |
| Event (CTCAE grade) . | Overall cohort, n (%) . | Third-party donor cohort, n (%) . |
|---|---|---|
| Number | 16 | 12 |
| CRS (grade 1) | 1 (6) | 1 (8) |
| Neurotoxicity | 2 (13) | 2 (13) |
| Irritability (grade 2) | 1 (6) | 1 (8) |
| Headache (grade 1) | 1 (6) | 1 (8) |
| Skin GVHD (grade 1) | 1 (6) | 1 (8) |
| Skin GVHD (grade 2) | 1 (6) | 1 (8) |
| Skin GVHD (grade 3) | 1 (6) | |
| Non-GVHD skin rash (grade 1) | 2 (12) | |
| Non-GVHD skin rash (grade 2) | 2 (12) | 2 (17) |
| Febrile neutropenia (grade 3) | 1 (6) | |
| Pneumonitis (grade 3) | 1 (6) | 1 (8) |
| Bone pain (grade 1) | 1 (6) | 1 (8) |
| Weight loss (grade 1) | 1 (6) | 1 (8) |
| Vomiting (grade 1) | 1 (6) | 1 (8) |
Response rates and long-term outcomes
In cohort 1 (n = 8), 3 patients with morphologic disease before treatment had morphologic disease after treatment (progressive/persistent disease), 3 patients with minimal residual disease–negative (MRD–) CR remained in MRD– CR, 1 patient with MRD+ CR remained in MRD+ CR, and 1 patient with MRD+ CR converted to MRD– CR after treatment (Table 2). In cohort 2 (n = 6), all patients had Deauville 4/PR in response to salvage chemotherapy before auto-HCT making them eligible for auto-HCT50 and consolidation with CAR EBV-VSTs. Five patients achieved CR after treatment with 1 patient having stable disease. In cohort 3 (n = 2), 1 patient with pretreatment CR had progressive disease after treatment and 1 patient with pretreatment PR had a CR after treatment.
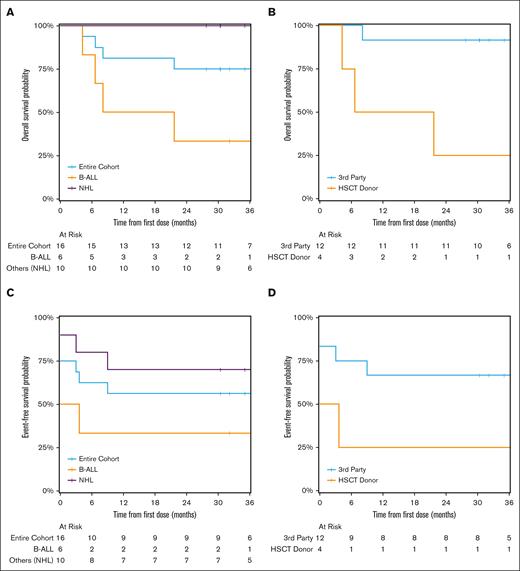
Median follow-up was 47.6 months (range, 4.2-135) with 4 deaths, all due to disease progression and all in cohort 1. OS of all patients was 81% (95% confidence interval [CI], 64-100) at 12 months and 75% (95% CI, 57-100) at 36 months whereas 36-month EFS rate was 56% (95% CI, 37-87). OS at 36 months was 100% for patients with non-Hodgkin lymphoma (n = 10) and 33% for patients with B-ALL (n = 6; Figure 3A). EFS at 36 months was 70% for patients with non-Hodgkin lymphoma and 33% for patients with B-ALL (Figure 3C). OS at 36 months was 92% for patients treated with third-party T cells (n = 12) and 25% for patients treated with their allo-HCT donors’ T cells (n = 4; Figure 3B). EFS at 36 months was 67% for patients treated with third-party T cells and 25% for patients treated with their allo-HCT donors’ T cells (Figure 3D). Our institution previously reported progression-free survival (PFS) of 49% in a similar cohort comparable with cohort 2,50,51 prompting us to evaluate PFS in this cohort, which was 83% at 36 months.
Long-term outcomes of patients treated with 19-28z CAR EBV-VSTs. (A) OS of patients with B-ALL vs NHL. (B) OS of patients with CAR EBV-VST products from their transplant donor vs a third-party donor. (C) EFS of patients with B-ALL vs NHL. (D) EFS of patients with CAR EBV-VST products from their transplant donor vs a third-party donor. Note: patients with no initial response to treatment are included with event time of 0. NHL, non-Hodgkin lymphoma.
Long-term outcomes of patients treated with 19-28z CAR EBV-VSTs. (A) OS of patients with B-ALL vs NHL. (B) OS of patients with CAR EBV-VST products from their transplant donor vs a third-party donor. (C) EFS of patients with B-ALL vs NHL. (D) EFS of patients with CAR EBV-VST products from their transplant donor vs a third-party donor. Note: patients with no initial response to treatment are included with event time of 0. NHL, non-Hodgkin lymphoma.
Biomarkers of response/T-cell phenotypic analysis
CAR EBV-VST persistence was measured in 15 of 16 patients, and CAR was detected in 2 of 16 patients (12%). CAR detection (peripheral blood) was demonstrated at a single time point after infusion for patient 1 (4 months) and patient 2 (2 days). Evaluating the correlation between CAR detection/persistence and EBV viremia, we found that both patients with evidence of CAR detection in the peripheral blood remained EBV– by peripheral blood PCR. However, this correlation is limited given that EBV viremia was not uniformly measured in this trial (11/16 patients had peripheral blood EBV PCR measured during follow-up), with only 1 patient with B-ALL exhibiting viremia (1 time point) that was not re-evaluated due to disease progression.
Peripheral blood B-cell aplasia (BCA) has been used as a surrogate marker of CAR T-cell persistence after CD19-specific CAR T-cell therapy.52 Before treatment, 8 of 16 patients had BCA evaluated with 5 patients demonstrating pretreatment BCA (patients 1, 2, 4, 9, and 15). After treatment, BCA was evaluated in 14 of 16 patients. For the 5 patients with pretreatment BCA, 3 patients ultimately relapsed and the other 2 patients had B-cell reconstitution at days 61 (patient 4) and 118 (patient 9). For the 3 patients who did not have pretreatment BCA, 1 patient had B-cell reconstitution at day 56, 1 patient was not evaluated after treatment, and 1 patient has remained in BCA for over 4 years. For the remaining 8 patients who did not have BCA evaluated before treatment, 1 patient was not evaluated after treatment (relapsed), 2 patients relapsed, 1 patient remained in BCA until 94 days (no subsequent evaluations), and 4 patients had B-cell reconstitution at days 18 (patient 11), 58 (patient 5; relapsed), 110 (patient 13), and 209 (patient 14). Therefore, prolonged BCA was not evident in this cohort, which corresponds to the limited persistence observed.
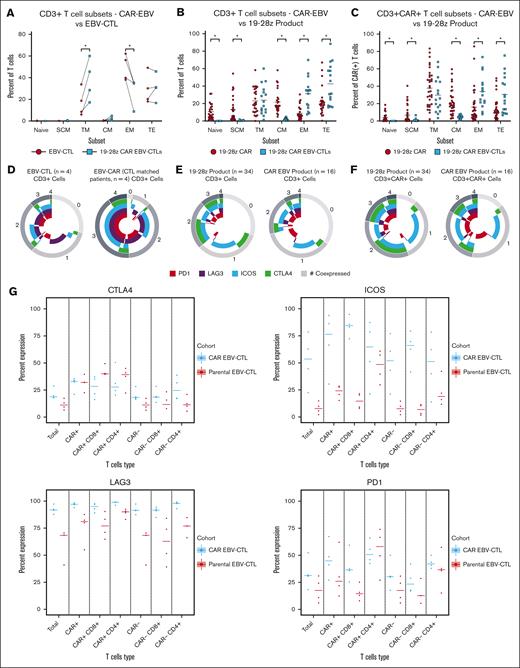
CAR EBV-VST products were compared with their corresponding parental EBV-VST products and to autologous CAR T-cell products expressing the same CD19-specific CAR9 to evaluate differentiation and exhaustion markers. Autologous/patient-derived CAR T-cell products and CAR EBV-VST products had comparable CAR transduction efficiencies (median, 24% and 26%, respectively), vector copy number (all products <4 copies per cell as required by institutional product certificate of analysis), and culture time after viral transduction (median, 7 days) with IL-2 supplementation.48 Total culture time between the 2 manufacturing processes was different with autologous CAR T cells having a median culture time of 11 days (range, 7-16) vs CAR EBV-VSTs, which were all cultured for 24 days after thawing of previously manufactured EBV-VSTs (parental EBV-VSTs were cultured for 28-35 days before cryopreservation; Figure 1). Comparing CAR EBV-VST products with their corresponding parental EBV-VST products (Figure 4A), CAR EBV-VST products had a higher proportion of transitional memory T cells and a lower proportion of effector memory T cells. Compared with CAR T-cell products from an autologous/patient-derived source, the CAR EBV-VST products consisted of a significantly lower proportion of naïve T cells, T stem cell memory, and central memory T cells and a significantly higher proportion of effector memory T cells and terminal effector T cells in both total T cells (Figure 4B) and CAR+ T cells (Figure 4C).
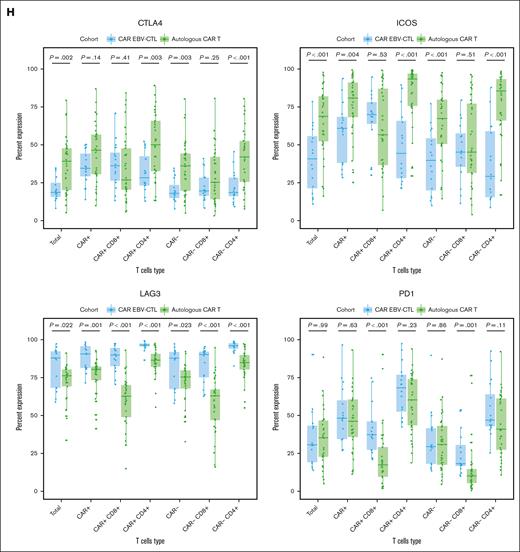
T-cell phenotypic analysis of 19-28z CAR EBV-VSTs. (A) Dot plots comparing CD3+ T-cell subsets of paired parental EBV-VSTs with 19-28z CAR EBV-VSTs. (B) Dot plots comparing CD3+ T-cell subsets of 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (C) Dot plots comparing CD3+ CAR+ T-cell subsets of 19-28z CAR EBV-VSTs vs 19-28z CAR T cells. (D) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ EBV-VSTs with 19-28z CAR EBV-VSTs. (E) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (F) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ CAR+ 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (G) Percent expression of individual exhaustion markers (PD1, LAG3, ICOS, and CTLA4) on T-cell subsets of EBV-VSTs vs 19-28z CAR EBV-VSTs. (H) Percent expression of individual exhaustion markers (PD1, LAG3, ICOS, and CTLA4) on T-cell subsets of 19-28z CAR EBV-VSTs vs 19-28z CAR T cells. P values were determined by Mann-Whitney-Wilcoxon tests.
T-cell phenotypic analysis of 19-28z CAR EBV-VSTs. (A) Dot plots comparing CD3+ T-cell subsets of paired parental EBV-VSTs with 19-28z CAR EBV-VSTs. (B) Dot plots comparing CD3+ T-cell subsets of 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (C) Dot plots comparing CD3+ CAR+ T-cell subsets of 19-28z CAR EBV-VSTs vs 19-28z CAR T cells. (D) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ EBV-VSTs with 19-28z CAR EBV-VSTs. (E) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (F) Ring graphs comparing exhaustion marker (PD1, LAG3, ICOS, and CTLA4) expression of CD3+ CAR+ 19-28z CAR EBV-VSTs with 19-28z CAR T cells. (G) Percent expression of individual exhaustion markers (PD1, LAG3, ICOS, and CTLA4) on T-cell subsets of EBV-VSTs vs 19-28z CAR EBV-VSTs. (H) Percent expression of individual exhaustion markers (PD1, LAG3, ICOS, and CTLA4) on T-cell subsets of 19-28z CAR EBV-VSTs vs 19-28z CAR T cells. P values were determined by Mann-Whitney-Wilcoxon tests.
Exhaustion marker expression was higher in CAR EBV-VST products than in their corresponding parental EBV-VST products (Figure 4D). However, exhaustion marker expression was lower in CAR EBV-VST products than autologous/patient-derived CAR T-cell products in both total T cells (Figure 4E) and CAR+ T cells (Figure 4F). A similar pattern was observed in CAR+CD4+ T cells (Supplemental Figure 2A); however, the opposite effect was observed in CAR+CD8+ T cells (Supplemental Figure 2B). CAR EBV-VST products show more frequent ICOS and LAG3 expression than their corresponding parental EBV-VST products (Figure 4G). Autologous CAR T-cell products showed consistently more frequent expression of CTLA4 and ICOS, whereas CAR EBV-VST products had more LAG3 and more PD1 but only on CAR+/CAR− CD8+ cells (Figure 4H).
Discussion
In this phase 1 trial, we successfully treated 16 patients with an allogeneic “off-the-shelf” CAR EBV-VST product with limited toxicity and no severe CRS, severe neurotoxicity, or DLTs. This trial established the safety and feasibility of multiple infusions/cycles of allogeneic “off-the-shelf” CAR EBV-VSTs in a cohort of heavily pretreated patients with R/R B-cell malignancies.
Our group has extensive experience with third-party–derived EBV-VSTs in the post-HCT setting with limited toxicity or GVHD.9,42 Therefore, we sought to enlist EBV-VSTs as a vehicle for our institutional CD19-specific CAR T cell. The use of CAR EBV-VSTs in the autologous setting has been previously reported with an anti-GD2 CAR in pediatric patients with R/R neuroblastoma.39 GD2-specific CAR EBV-VSTs were noted to persist in greater numbers and for a greater period of time after adoptive transfer than activated T cells expressing the same CAR but lacking viral specificity.39,53,54 The use of allogeneic CAR EBV-VSTs has also been previously reported in the setting of B-cell malignancies after HCT.35,39,40 Importantly, results demonstrated that signaling through the native T-cell receptor (TCR) during viral reactivation enhances the proliferation and function of CAR EBV-VSTs.40 The successful development of an allogeneic viral-specific T-cell product redirected to a tumor via the expression of a Wilms’ tumor antigen 1–specific TCR (TCRC4) has also been previously reported and demonstrated favorable outcomes in the prophylactic setting.55
In contrast to our previous institutional experience of autologous CAR T-cell infusion as consolidation after high-dose chemotherapy and auto-HCT (10/15 [67%] of patients experienced severe neurotoxicity/ICANS), this trial demonstrated minimal toxicity in a near identical cohort (cohort 2).13 Patients in cohort 2 had to meet an inclusion criterion of Positron Emission Tomography (PET)+ disease to be eligible for auto-HCT (PET+ PR), which is a high-risk cohort with an expected PFS of 40% to 50%.50,51 Notably, our cohort of patients demonstrated a PFS of 83% at 36 months. This warrants further investigation of CAR EBV-VSTs in a larger cohort of patients given that the focus of this phase 1 trial was to determine the safety and feasibility of using CAR EBV-VSTs.
T-cell phenotype, including expression of differentiation and exhaustion markers, has been shown to impact response to CAR T-cell therapy.56-60 Apheresis products containing a higher proportion of naïve or memory T cells mediate postinfusion CAR T-cell persistence, and end-of-production (EOP) CAR T cells containing a higher proportion of memory CD8+ T cells have improved responses.58,60 Comparison of apheresis and EOP CAR T-cell products has identified dysfunctional/exhausted signatures that associate with decreased patient responses.56,57 Not surprisingly, we demonstrated increased expression of exhaustion markers in EOP CAR EBV-VSTs compared with corresponding parental EBV-VST products. This is likely due to the additional LCL stimulation to enable gene transfer and extended culture time of CAR EBV-VSTs. However, compared with products derived from patients with B-cell malignancies using CD3/CD28 bead manufacturing with the same 19-28z CAR construct, the CAR EBV-VST products had a lower proportion of T cells expressing exhaustion markers. This suggests that a manufacturing process using LCL stimulation to generate CAR EBV-VSTs from healthy donors has the potential to result in less exhaustion compared with the CD3/CD28 bead–based manufacturing process. Further comparisons between these manufacturing processes are warranted given that PD1 and LAG3 are known markers of T-cell activation, which could result in their elevation after shorter culture time and/or tonic signaling resulting in activation-induced exhaustion.48 Compared with autologous/patient-derived products, the CAR EBV-VST products consisted of a significantly lower proportion of naïve and memory T cells and significantly higher proportion of more differentiated T cells, albeit less exhausted T cells. Therefore, in addition to their allogeneic origin, these characteristics could also contribute to the limited persistence we observed in patients treated with CAR EBV-VSTs compared with the patients treated with CAR T cells. The utilization of multiple infusions of CAR EBV-VSTs or earlier transduction of EBV-VSTs has the potential to overcome this limited persistence.40
Despite our demonstration that allogeneic CAR EBV-VST treatment is feasible and tolerable, several limitations to our study exist. We and others have reported on the importance of the intensity and exposure of lymphodepleting chemotherapy in mediating clinical outcomes in patients treated with CAR T cells.9,61-63 Therefore, future clinical trial designs using third-party CAR EBV-VSTs should use a harmonized lymphodepletion, which may further optimize this cellular therapy platform. Our initial study design used HCT donors as the source of EBV-VSTs, which proved to be logistically cumbersome and failed to establish a readily available product. To overcome this, we transitioned to the use of third-party donor-derived EBV-VSTs. There was a wide variation in the number of CAR T cells per kg administered to each patient as our dosing strategy relied on a total T-cell dose per kg compared with a flat dose or CAR per kg dose. ASTCT ICANS grading was not feasible (absence of Immune Effector Cell-Associated Encephalopathy (ICE) score collection during the time period of study enrollment); however, patients exhibited minimal neurotoxicity, and we have previously reported concordance between CTCAE and ASTCT neurotoxicity grading.64 Cohort 3 failed to achieve target recruitment due to slow enrollment and the eventual closure of the study after the achievement of the primary objective for patients in the trial as a whole. Finally, further investigation of CAR EBV-VSTs in a larger cohort of patients is warranted to determine any signal of efficacy.
This trial determined a manufacturing platform of allogeneic CAR EBV-VSTs to enable repeated dosing for multiple recipients per manufactured cell line. The use of split or sequential CAR T-cell infusions has been successfully applied in the setting of adult patients with R/R B-ALL, and our trial further demonstrates the feasibility of this approach.65 In the cohort of heavily pretreated patients, long-term outcomes after multiple infusions of CAR EBV-VSTs were excellent. In summation, allogeneic “off-the-shelf” CAR EBV-VSTs provide a readily available therapy for patients with limited therapeutic options and provide a safe approach for consolidative therapy after HCT.
Acknowledgments
The authors acknowledge the editorial assistance of Joseph Olechnowicz, Department of Pediatrics, Memorial Sloan Kettering Cancer Center (MSKCC). The authors thank the patients and families who participated in this trial; the staff of the Cell Therapy and Cell Engineering Facility, Virginia Escobedo, Victoria Szenes, Adrienne Catedral, Nicholas Webb, and Keith Kallas; and the Center for Immune Cell Therapies at MSKCC for their help with the clinical trial management of this study.
This work was supported in part by MSKCC support grant/core grant P30 CA008748 and philanthropic support from Comedy vs Cancer.
MSKCC is the sponsor of this study and holds the patent for the technologies used in this study. This technology has been licensed to Atara Biotherapeutics (EBV-VSTs) and Juno/Celgene (19-28z CAR) for additional development. The MSK Institutional Review Board and Research Council influenced the study design in the interest of scientific clarity and human participants protection. Only the authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Authorship
Contribution: S.S. and K.J.C. wrote the manuscript, collected data, and analyzed data; G.C.F. and J.B. collected data; A.M., C.O.W., D.M., and K.H. analyzed data; M.S., R.J.O., N.A.K., R.J.B., I.R., and K.J.C. designed the clinical trial concept and preclinical testing. R.J.O., S.E.P., and R.J.B. designed research and treated patients; M.I.C., A.A.J., A.S., J.J.B., C.S.S., M.-A.P., S.A.G, and K.J.C. treated patients; C.T., J.C., X.W., I.R., and M.S. performed and supervised research; and all authors critically reviewed the manuscript and approved the content.
Conflict-of-interest disclosure: K.J.C. has received research support from Atara Biotherapeutics, Novartis, Celgene, and Cellectis, and has consulted and participated in advisory boards for Novartis. S.E.P. has received support for the conduct of clinical trials through Boston Children’s Hospital from AlloVir, Atara Biotherapeutics, and Jasper; is the inventor of intellectual property related to development of third-party viral-specific T-cell program, with all rights assigned to Memorial Sloan Kettering Cancer Center; and reports honoraria, consulting, or participation in advisory board for Pierre Fabre, Regeneron, Cellevolve, Vor, and Ensomo, DSMB, Stanford University, and New York Blood Center. J.J.B. has consulted for Merck, Sanofi, Sobi, and SmartImmune and received compensation for serving on data monitoring committee DMC (either chair or member) for Advanced Clinical and CTI Clinical Trial Services. N.A.K. holds equity in Amgen, Johnson & Johnson, and Merck. R.B. has licensed intellectual property to and collects royalties from Bristol Myers Squibb (BMS), Caribou, and Sanofi; received research funding from BMS; is a consultant to BMS and Atara Biotherapeutics Inc; and was on the scientific advisory board of Triumvira, Cargo Tx, and CoImmune. R.J.O. received royalties, research support, and consulting from Atara Biotherapeutics. M.S. reports research support from Atara Biotherapeutics and has licensed intellectual property to Juno Therapeutics, Atara Biotherapeutics, Fate Therapeutics, Takeda Pharmaceuticals, Mnemo Therapeutics, and Minerva Biotechnologies. I.R. is a scientific cofounder of Mnemo Therapeutics and reports participation in the advisory board of Center for Commercialization of Cancer.
Correspondence: Kevin J. Curran, Pediatrics, Memorial Sloan Kettering Cancer Center, 1275 York Ave, New York, NY 10065; email: currank@mskcc.org.
References
Author notes
The protocol summary and/or a statistical summary are available on request from the corresponding author, Kevin J. Curran (currank@mskcc.org). Requests for deidentified individual participant data can be made beginning 12 months after publication and for up to 36 months after publication. Deidentified individual participant data reported in the manuscript will be shared under the terms of a data use agreement and may only be used for approved proposals. Requests may be made to crdatashare@mskcc.org. Requests should contain adequate information on the type of analysis to be performed and a full listing of parties that will have access through this request.
The full-text version of this article contains a data supplement.