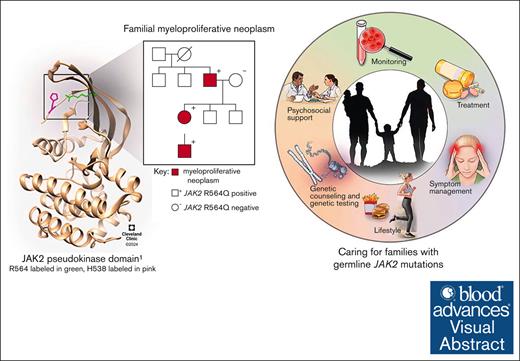
Key Points
Germ line variants in JAK2, R564Q and R564L, cause hereditary thrombocythemia, potentially increasing risk of other hematologic neoplasms.
The symptomatic burden for individuals harboring germ line JAK2 R564Q/L variants can be comparable with those with the canonical JAK2V617F hit.
Visual Abstract
The Janus kinase 2 (JAK2) V617F mutation activates the transcription pathway and has been well characterized as a driver of myeloproliferative neoplasms (MPNs). Recently, there has been a heightened interest in understanding germ line predisposition to hematologic malignancies such as MPN, including several reports of familial MPN. Here, we retrospectively analyzed medical records and data from genetic testing to describe 12 patients with germ line variants at amino acid position 564 of JAK2. This includes 3 supportive cases adding to prior literature regarding the germ line JAK2 R564Q association with hereditary thrombocythemia, as well as confirmation of JAK2 R564L as a germ line variant associated with MPN. Importantly, the symptomatic burden for many of the individuals in this series is comparable with that of individuals with the canonical V617F mutation profile. In the JAK2 R564Q cases, we noted a pattern of familial aggregation, presence of congenital thrombocythemia, and co-occurrence with hematologic neoplasms. Identification of germ line predisposition is essential for understanding the pathogenesis of disease, impact on families, and opportunities for preventive care. Continued research is essential to further characterize the penetrance of these conditions and how best to monitor, treat, and optimize management for these families.
Introduction
Janus kinase 2 (JAK2) plays an important role in both megakaryopoiesis and erythropoiesis via the activation of JAK–signal transducer and activator of transcription (STAT) signaling, leading to cell proliferation and avoidance of apoptosis. Mutations in the JAK2 gene are a common cause of BCR::ABL1–negative myeloproliferative neoplasms (MPNs). The canonical activating point mutation, p.V617F, is found as the driver for ∼50% to 60% of essential thrombocythemia (ET) and 95% of polycythemia vera (PV).1 Valine 617 lies in the Jak homology domain 2 (JH2) of the protein, a pseudokinase domain that normally inhibits autophosphorylation by the kinase domain, JH1. The p.V617F mutation disrupts this inhibitory role of JH2, leading to constitutive activation of JH1 and the downstream JAK-STAT signaling cascade.2 Additional clinically significant variants in exon 12 of JAK2 as well as less common variants across exons 12 to 15 within the pseudokinase domain and throughout the entire gene have been identified for patients with MPN.3,4
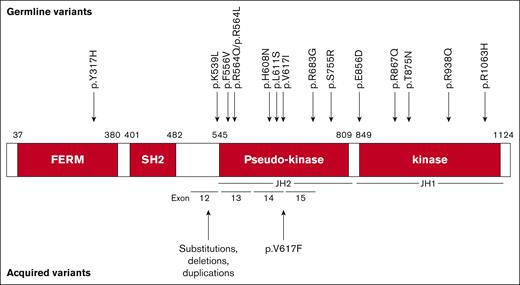
Although most JAK2 variants, including p.V617F, occur as acquired, somatic mutations, germ line JAK2 variants have been reported in association with hereditary thrombocythemia (HT), hereditary erythrocytosis (HE), and familial predisposition to MPN.5-20 These germ line variants are reported throughout the pseudokinase and kinase domains, as shown in Figure 1. MPN associated with germ line JAK2 variants can occur either in the absence of additional variants in JAK2, CALR, or MPL (idiopathic or “triple-negative”)13 or alongside acquired variants, including p.V617F-positive MPN.21 Additionally, the JAK2 46/1 haplotype, consisting of 4 single nucleotide polymorphisms within intronic regions of JAK2, is present in up to 45% of the general population and has been observed to confer a somewhat increased risk of acquiring p.V617F, leading to MPN development (odds ratio, 3.07; 95% confidence interval, 1.73–5.46).22,23
Map of JAK2 protein structure, germ line genetic variants associated with HT and/or HE, and acquired variants occurring in sporadic MPN. Amino acid numbers are given according to the beginning and end of each functional domain. FERM, four-point-one ezrin, radixin, moesin domain; SH2, Src homology domain 2.
Map of JAK2 protein structure, germ line genetic variants associated with HT and/or HE, and acquired variants occurring in sporadic MPN. Amino acid numbers are given according to the beginning and end of each functional domain. FERM, four-point-one ezrin, radixin, moesin domain; SH2, Src homology domain 2.
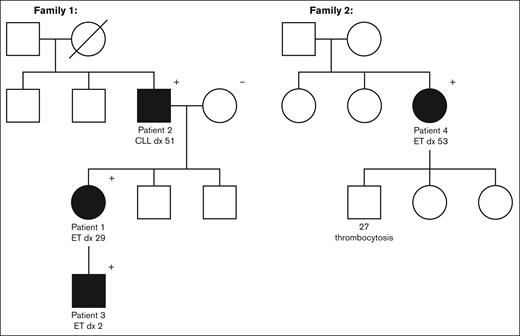
Etheridge et al previously reported a family in which JAK2 p.R564Q segregated with familial ET and demonstrated through functional studies that p.R564Q causes increased activation of the JAK2 protein in platelets and suppression of apoptosis, leading to increased cell growth.17 Here, we describe a family including patients from 3 generations with p.R564Q associated with either congenital idiopathic thrombocythemia or lymphocytosis (Figure 2), as well as 2 patients with idiopathic thrombocythemia and confirmed germ line variants in an alternative variant at the same residue, p.R564L. Additional cases of p.R564L were identified through investigation of our in-house somatic next-generation sequencing (NGS) data. The p.R564L variant has been previously reported and postulated as a germ line variant.4,24-26 The cases reported here confirm a germ line origin for p.R564L and support its role as a cause of HT, with a possible association with other MPNs or hematologic neoplasms.
Three- or 4-generation pedigrees collected for family 1 and family 2. The first illustrates the segregation of p.R564Q with ET and CLL in family 1. The proband in family 2 was identified to have the p.R564L variant. Her son is reported to have thrombocytosis (∼500 × 103/μL) but has not had genetic testing. +, positive for JAK2 variant; –, negative for JAK2 variant; dx, diagnosed.
Three- or 4-generation pedigrees collected for family 1 and family 2. The first illustrates the segregation of p.R564Q with ET and CLL in family 1. The proband in family 2 was identified to have the p.R564L variant. Her son is reported to have thrombocytosis (∼500 × 103/μL) but has not had genetic testing. +, positive for JAK2 variant; –, negative for JAK2 variant; dx, diagnosed.
Methods
The study was approved by the institutional review board of Cleveland Clinic. Clinical germ line genetic testing was performed by a Clinical Laboratory Improvement Amendments–certified, College of American Pathologists–accredited, germ line laboratory via NGS sequencing of DNA extracted from cultured fibroblasts or from peripheral blood when testing relatives for a known family variant. Patients who underwent clinical germ line genetic testing received pretest and posttest genetic counseling to review and discuss benefits and limitations of testing and obtain informed consent.
To determine the incidence and frequency of JAK2 p.R564 variants, a cohort of ∼15 000 consecutive cases tested using a custom hematologic neoplasm NGS gene panel in the Clinical Laboratory Improvement Amendments–certified, College of American Pathologists–accredited Molecular Pathology Laboratory at Cleveland Clinic was queried. Genomic DNA was extracted from peripheral blood collected in EDTA or from bone marrow aspirate collected in EDTA or sodium heparin. The extraction method was either automated on the QIAsymphony (QIAGEN, GmBH), using the DNA midi kit, or manually performed using the QIAGEN Puregene Blood kit. DNA quantity and quality were assessed using Qubit Fluorometer and Nanodrop Spectrophotometer (Thermo Fisher Scientific, Waltham, MA), respectively, and stored at 2 to 8°C before testing.
Genomic DNA (200 ng) from each sample was used for NGS library preparation using a panel of custom primers targeting 63 genes (including JAK2) and Archer VariantPlex reagents for Illumina (Archer, Boulder, CO) following the manufacturers’ recommendation. Briefly, anchored multiplex polymerase chain reaction chemistry was used, in which molecular barcodes and universal primer binding sites were ligated to the DNA library, followed by bead-based fragments selection. This was followed by annealing with gene-specific primers targeting the 63 genes on the panel and polymerase chain reaction amplification. Individual final NGS libraries were quantified using a Qubit fluorometer, pooled, and requantified. Pooled libraries were denatured and sequenced using 2× 151-base pair cycles (with dual indexing) on the NextSeq 550 platform (Illumina Inc, San Diego, CA). Raw sequence was demultiplexed using bcl2fastq software to generate fastq files. Deduplication with molecular barcodes, read alignment, and variant detection including JAK2 p.R564 variants were performed using Archer Analysis software version 6.0.2 (Archer, Boulder, CO). Custom in-house–built scripts were then used for downstream quality control analysis, variant filtering, and case report generation. Additional manual variant filtering and review to evaluate JAK2 variants were performed in cases in which extended JAK2 sequencing was not originally clinically ordered and, thus, masked.
Medical records were retrospectively reviewed for all 12 patients to obtain additional clinical information, bone marrow reports, other laboratory values (including past and present complete blood counts and differentials), and family history, as available. It was noted for each patient whether they had been evaluated by genetics or had confirmation of germ line status via additional testing on a nonhematopoietic sample (cultured skin fibroblasts) or through family studies.
Structural analyses were performed for full-length JAK2 (amino acids 1-1132; UniProt ID O60674), modeled using AlphaFold.27,28 The structural quality, as assessed by a pLDDT (per-residue model confidence) score of 87.8, is very high confidence for the entire molecule. The loop regions were further refined using modller.29,30 The regional pLDDT score around R562 was >90. To assess the structural pathogenicity score, AlphaMissense31 was used. Molecular graphics and analyses were performed using PyMOL32 and UCSF Chimera.33 Coulombic, hydrogen-bond, and van der Waals interactions were assessed using distance and angle parameters.
Results
HT case presentations
A 29-year-old female (patient 1) presented to the hematology clinic for evaluation due to a longstanding history of thrombocytosis, dating back to at least age 19 years, as well as intermittent leukocytosis with absolute neutrophilia. Her platelet counts ranged from 400 × 103 to 580 × 103/μL, and her white blood cell count was 20 × 103/μL at the highest, with an absolute neutrophil count of 15.6 × 103/μL. Sequencing of JAK2 exons 12 to 16 was performed on peripheral blood and identified a heterozygous p.R564Q (c.1691G>A) variant at an allele fraction of 47%, and the patient was diagnosed with ET at our affiliate site. She reported a history of chronic headaches and pulmonary embolism at age 16 years in the setting of oral contraceptive use. Bone marrow evaluation showed normocellular to mildly hypocellular marrow and adequate megakaryocytes with unremarkable morphology. Mild reticulin fibrosis (MF-1) was also noted. Flow cytometry showed no evidence of lymphoproliferative disorders. Chromosome analysis revealed a normal, female karyotype. A larger, 63-gene somatic hematologic neoplasm NGS panel test redemonstrated the JAK2 p.R564Q variant at 51% variant allele fraction (VAF) and did not identify any additional variants. Germline genetic testing confirmed p.R564Q as a heterozygous variant found in cultured skin fibroblasts. Concurrently, the proband’s father was being evaluated due to lymphocytosis. His white blood cell count was 14.1 × 103/μL, with an absolute lymphocyte count of 6 × 103/μL; hemoglobin was 15.2 g/dL, and platelet counts were 400 × 103/μL. Genetic testing showed that he was heterozygous for the p.R564Q variant. He was subsequently diagnosed with chronic lymphocytic leukemia (CLL). His somatic hematologic neoplasm NGS testing also showed JAK2 p.R564Q as well as a clinically significant NOTCH1 variant, c.7330C>T (p.Q2444∗), at 51% and 7% VAFs, respectively. Later, the proband’s 2-year-old son was found to have thrombocytosis (544 × 103/μL) in the setting of minor illness. Repeat laboratory tests at his baseline showed persistent thrombocytosis (425× 103 to 514 × 103/μL). Genetic testing for the familial variant revealed that he was also heterozygous for p.R564Q.
Two additional unrelated females presented to our hematology oncology clinic with ET in association with an alternative variant at the same position, p.R564L (c.1691G>T). Patient 4 was a 53-year-old female following with hematology oncology over a period of ∼3 years for persistent thrombocytosis. Her bone marrow evaluation showed a mild increase in megakaryocytes with mostly normal morphology, although some showed nuclear hyperlobulation. There was no significant reticulin fibrosis (MF-0). Her somatic NGS panel testing performed on bone marrow identified the JAK2 p.R564L variant at a VAF of 51%. She underwent germ line genetic testing on cultured skin fibroblasts that confirmed the heterozygous p.R564L variant. Her family history included a 27-year-old son with reported thrombocytosis (∼500 × 103/μL) (Figure 2). Patient 5 was a 49-year-old female with a 13-year history of chronic thrombocytosis (410 × 103 to 520 × 103/μL), seen for evaluation for ET. Somatic NGS testing identified the JAK2 p.R564L (c.1691G>T) variant at a VAF of 49%. This was confirmed as a germ line variant via testing on cultured skin fibroblasts. To our knowledge, she did not have a family history of thrombocytosis or MPN. Her father passed away due to graft-versus-host disease after allogeneic stem cell transplant for non-Hodgkin lymphoma, diagnosed at the age 52 years.
JAK2 p.R564 variants in a cohort of patients with MPN and hematologic neoplasms
To probe whether JAK2 p.R564 variants were present as a potential cause of MPN in other patients seen at our institution, we assessed NGS data from patients who had a hematologic neoplasm panel test (∼15 000 cases). Seven additional individuals with the p.R564L variant were identified. For all but 1 of these individuals, the VAF for p.R564L was ∼50%, consistent with a germ line origin; the exception was an individual for whom the VAF was 94% for the p.R564L variant and 88% for p.V617F, suggestive of possible loss of heterozygosity. Three of these individuals were diagnosed with PV, 1 with ET, 1 with CLL, 1 with acute myeloid leukemia (AML), and 1 with monoclonal gammopathy of uncertain significance (MGUS). Two of these patients had also acquired the JAK2 p.V617F variant. Patient 12 previously had a splenectomy at age 18 years due to a traumatic sports injury. For at least several months before his diagnosis with AML at age 50 years, he had a history of thrombocytosis (675 × 103/μL) and leukocytosis (75 × 103/μL) with lymphocytosis, neutrophilia, and monocytosis, and at the time of diagnosis, additional acquired clinically significant variants were identified in ASXL1 c.2253dupT (p.A752Cfs∗22), CEBPA c.247delC (p.Q83Sfs∗77), SRSF2 c.284C>A (p.P95H), STAG2 c.1035_1047del13 (p.K346Lfs∗29), and TET2 c.3818G>C (p.C1273S), at VAFs of 48%, 93%, 46%, 95%, and 95%, respectively. His cytogenetics showed a normal, male karyotype, and his AML was consistent with a classification of AML with myelodysplasia-related changes. In addition to a diagnosis of PV, patient 8 also had thrombocytosis, neutrophilic leukocytosis, and monocytosis. In instances in which data were assessable, the majority of the patients harboring JAK2 p.R564 variants exhibited significant scores on the MPN-SAF TSS (MPN symptom assessment form total symptom score; Table 1).
Clinical features of patients with JAK2 R564 variants
| Patient no. . | 1 . | 2 . | 3 . | 4 . | 5 . | 6 . | 7 . | 8 . | 9 . | 10 . | 11 . | 12 . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diagnosis | ET | CLL | ET | ET | ET | ET | CLL | PV | PV | MGUS | PV | AML |
| Age, y | 29 | 51 | 2 | 53 | 49 | 45 | 70 | 73 | 28 | 75 | 61 | 50 |
| Sex | F | M | M | F | F | F | F | M | M | M | M | M |
| Race | Multiracial | Black | Multiracial | White | White | White | White | White | Unknown | Black | White | White |
| JAK2 R564 (VAF) | Q (51%) | Q (51%) | Q (het∗) | L (51%) | L (49%) | L (52%) | L (51%) | L (52%) | L (52%) | L (50%) | L (94%) | L (49%) |
| JAK2 R564X confirmed as germ line | Yes | Yes | Yes | Yes | Yes | No | No | No | No | No | No | No |
| JAK2 V617F (VAF) | No | No | No | No | No | No | No | Yes (11%) | No | No | Yes (88%) | No |
| White blood cell count, ×103/μL | 10.7 | 14.1 | 10.1 | 6.9 | 6.5 | 8.8 | NA | 16.1 | NA | 15.5 | 25.6 | 21.3 |
| Hemoglobin, g/dL | 13.7 | 15.2 | 12.7 | 13.9 | 13.5 | 14.2 | NA | 16.4 | 17.2 | 10.3 | 16.2 | 12.2 |
| Platelet count ×103/μL | 575 | 400 | 503 | 519 | 442 | 438 | NA | 446 | 375 | 75 | 352 | 308 |
| History of thrombosis | PE | No | No | No | No | No | NA | No | No | No | DVT, CRVO | SVT, PVT |
| Splenomegaly | No | No | No | No | No | No | NA | No | No | No | Yes | Splenectomy |
| MPN-SAF TSS | 38 | 34 | NA | 0 | 0 | 53 | NA | NA | 29 | NA | NA | NA |
| Family no. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Family history of cytosis/MPN | ET | ↑Plts | No | PV | NA | No | No | No | No | No | ||
| Family history of HM | CLL | No | NHL | LGL, lymphoma, leukemia | NA | No | No | No | No | Leukemia | ||
| Family history of thrombosis | PE | DVT | DVT, CVA | No | NA | No | No | No | No | No |
| Patient no. . | 1 . | 2 . | 3 . | 4 . | 5 . | 6 . | 7 . | 8 . | 9 . | 10 . | 11 . | 12 . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diagnosis | ET | CLL | ET | ET | ET | ET | CLL | PV | PV | MGUS | PV | AML |
| Age, y | 29 | 51 | 2 | 53 | 49 | 45 | 70 | 73 | 28 | 75 | 61 | 50 |
| Sex | F | M | M | F | F | F | F | M | M | M | M | M |
| Race | Multiracial | Black | Multiracial | White | White | White | White | White | Unknown | Black | White | White |
| JAK2 R564 (VAF) | Q (51%) | Q (51%) | Q (het∗) | L (51%) | L (49%) | L (52%) | L (51%) | L (52%) | L (52%) | L (50%) | L (94%) | L (49%) |
| JAK2 R564X confirmed as germ line | Yes | Yes | Yes | Yes | Yes | No | No | No | No | No | No | No |
| JAK2 V617F (VAF) | No | No | No | No | No | No | No | Yes (11%) | No | No | Yes (88%) | No |
| White blood cell count, ×103/μL | 10.7 | 14.1 | 10.1 | 6.9 | 6.5 | 8.8 | NA | 16.1 | NA | 15.5 | 25.6 | 21.3 |
| Hemoglobin, g/dL | 13.7 | 15.2 | 12.7 | 13.9 | 13.5 | 14.2 | NA | 16.4 | 17.2 | 10.3 | 16.2 | 12.2 |
| Platelet count ×103/μL | 575 | 400 | 503 | 519 | 442 | 438 | NA | 446 | 375 | 75 | 352 | 308 |
| History of thrombosis | PE | No | No | No | No | No | NA | No | No | No | DVT, CRVO | SVT, PVT |
| Splenomegaly | No | No | No | No | No | No | NA | No | No | No | Yes | Splenectomy |
| MPN-SAF TSS | 38 | 34 | NA | 0 | 0 | 53 | NA | NA | 29 | NA | NA | NA |
| Family no. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Family history of cytosis/MPN | ET | ↑Plts | No | PV | NA | No | No | No | No | No | ||
| Family history of HM | CLL | No | NHL | LGL, lymphoma, leukemia | NA | No | No | No | No | Leukemia | ||
| Family history of thrombosis | PE | DVT | DVT, CVA | No | NA | No | No | No | No | No |
Clinical characteristics of patients identified with confirmed or putative germ line JAK2 p.R564 variants. VAF is given for the p.R564 variant identified. For several of the patients, variants were confirmed as germ line via testing on cultured skin fibroblasts or family variant testing. Two patients had concurrent acquired mutation of p.V617F. The MPN-SAF TSS is provided when available as a measure of symptom burden. Family history is provided to the degree it was available upon review of the medical record. Family 1 consists of patients 1, 2, and 3.
↑, increased; CRVO, central retinal vein occlusion; CVA, cerebral vascular accident; DVT, deep vein thrombosis; F, female; het∗, heterozygous; HM, hematologic malignancy; LGL, large granular lymphocyte leukemia; M, male; NA, not available; NHL, non-Hodgkin lymphoma; PE, pulmonary embolism; Plts, platelets; PVT, portal vein thrombosis; R564X, R564L or R564Q; SVT, superficial vein thrombosis.
Functional effect of p.R564L based on in silico modeling
Multiple in silico predictions and simulations suggest a strong pathogenic potential for the p.R564L variant in JAK2. Structural analysis revealed that the p.R564L mutation disrupts the local structure of the protein, particularly affecting the interaction network involving the residues H538, E566, and Y613, as depicted in Figure 4. The AlphaMissense score calculated for p.R564L is 0.956, corresponding with a prediction of likely pathogenic.
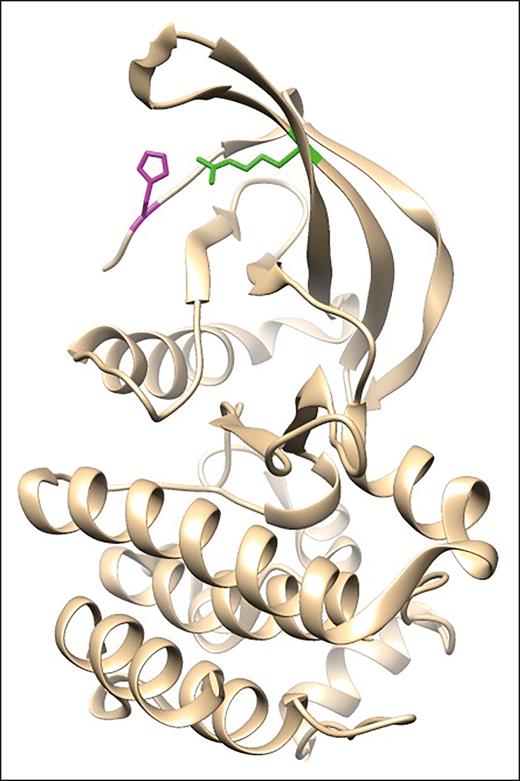
Previously published crystal structure of the JAK2 pseudokinase domain,34highlighting the proposed interaction between p.R564 and p.H538. Adapted from Bandaranayake et al.34
Structural impact of the p.R564L mutation on JAK2 protein.JAK2 p.R564I leads to disruption of local structure. (A) Ribbon diagram of full-length JAK2 (amino acids, 1-1132; UniProt ID O60674) modeled using AlphaFold,27,28 followed by loop refinement using Modeller.29,30 Coloring corresponds with the regional pLDDT score as shown in the key. (B) The amino acids within 5 Å of arginine 564 are highlighted and shown with ball and stick models. Hydrogen-bond and van der Waals interactions are depicted as dashed lines. The p.R564L mutation is expected to disrupt key interactions with nearby residues including His538, E566, Y613, T576, and His574, altering the local structural integrity.
Structural impact of the p.R564L mutation on JAK2 protein.JAK2 p.R564I leads to disruption of local structure. (A) Ribbon diagram of full-length JAK2 (amino acids, 1-1132; UniProt ID O60674) modeled using AlphaFold,27,28 followed by loop refinement using Modeller.29,30 Coloring corresponds with the regional pLDDT score as shown in the key. (B) The amino acids within 5 Å of arginine 564 are highlighted and shown with ball and stick models. Hydrogen-bond and van der Waals interactions are depicted as dashed lines. The p.R564L mutation is expected to disrupt key interactions with nearby residues including His538, E566, Y613, T576, and His574, altering the local structural integrity.
Discussion
Single nucleotide variants at p.R564 are rare, and their impact on JAK2 kinase activity was largely unknown until a decade ago, when the causal role of p.R564 was suggested in the pathogenesis of HT.17 The family studies and cases identified from our sequencing data provide further evidence supporting this causal role for p.R564 in HT. Similar to the prior report, we identified a second family in which 3 family members were affected with p.R564Q associated with MPN. The presence of thrombocytosis in this family further suggests that these germ line variants downregulate the inhibitory role of JH2, leading to constitutive activation of JH1 and the downstream JAK-STAT signaling cascade, akin to V617F. In the first report of familial p.R564Q, Etheridge et al described 3 family members who presented with HT. The authors demonstrated through functional studies that p.R564Q led to increased tyrosine phosphorylation of JAK2 and downstream signaling proteins (STAT1, STAT3, and STAT5). This upregulated intracellular signaling, leading to increased cell proliferation. Although the increase in phosphorylated JAK2 was comparable with that seen for p.V617F, downstream signaling and the proliferative effect were muted compared with p.V617F. It was proposed that p.R564Q can interfere with JH2 inhibition by SH2, leading to increased transphosphorylation of JH1 by disrupting hydrogen bonding hypothesized between p.R564 and p.H538 within JH2, based on the crystal structure of the pseudokinase domain34 (Figure 3).
In addition to p.R564Q, we confirmed the germ line status of p.R564L through germ line genetic testing on cultured fibroblasts for 2 unrelated patients with thrombocythemia in our clinic and identified the presence of the p.R564L variant in several additional patients affected by MPN or hematologic malignancies. The p.R564L variant has been previously identified on somatic NGS testing in patients with MPN.4,24-26 The p.R564L variant has a population allele frequency of 0.00006592 (Genome Aggregation Database v4.1.0, GRCh38). In silico predictions support a deleterious effect for the p.R564L variant, with high CADD (25.6), PhyloP (8.78), and PolyPhen (0.995) scores, suggesting pathogenicity and evolutionary conservation. Similarly, our structural data demonstrate that the p.R564L variant likely abrogates several local interactions, including hydrogen bonding with p.H538, disrupting protein structure and function. Furthermore, the AlphaMissense score of 0.956 corresponds with a likely pathogenic prediction. With the data available at this time, p.R564L would be classified as a variant of uncertain significance by American College of Medical Genetics (ACMG) guidelines35 (PM1, PM5, PP2, and PP3). Although our structural modeling suggests that p.R564L disrupts the local structure of JAK2 and predicts a classification of likely pathogenic, future functional studies are needed to validate our observation. Overall, our findings suggest that p.R564L is a germ line variant that may lead to HT and other hematologic neoplasms.
Genetic risk factors for MPN can be categorized as monogenic causes that are highly penetrant for HT or HE, monogenic inherited predisposition to MPN with incomplete penetrance, or polymorphisms that slightly modify the risk or phenotype for MPN and are associated with a risk of acquiring somatic driver mutations.28 For JAK2 p.R564Q, a pattern of apparently congenital thrombocythemia with thrombocythemia being present in the first decade of life (as early as ages 2, 6, and 9 years in the previously reported family16 and in family 1 reported here) has been observed, affecting multiple first-degree relatives within 2 relatively small families. Although the mutation status of unaffected family members is not available for either family, given the striking pattern of very early-onset thrombocythemia within these families, we hypothesize that the predisposition caused by p.R564Q best fits within the category of highly penetrant or congenital thrombocythemia. Our series also indicates that single, heterozygous p.R564 variants appear sufficient to drive disease pathogenesis, resulting in a readily apparent phenotype without requiring additional “hits” such as p.V617F or mutations in other MPN-associated genes. However, patient 2 was identified to carry an additional mutation in NOTCH1 (at 7% VAF) and was diagnosed with CLL, which is possibly incidental or could suggest that individuals with germ line p.R564Q may acquire additional variants, affecting their disease course.
Our study identified 9 patients with p.R564L in the context of various hematologic phenotypes including HT, PV, CLL, AML, and MGUS. Three of these patients were females with a longstanding history of thrombocytosis, with eventual diagnosis of HT in their late 40s to early 50s. Of the patients diagnosed with PV, 2 had concomitant p.V617F mutation. These were the only 2 patients in the series who also harbored the p.V617F mutation. One of these 2 patients had considerably high VAFs for both p.R564L (94%) and p.V617F (88%), suggestive of possible loss of heterozygosity. Overall, the range of phenotypes demonstrated for patients with p.R564L germ line variants exhibit considerable diversity compared with the p.R564Q variant.
Interestingly, patient 2 had only mild thrombocythemia but presented with concomitant chronic monoclonal B-cell lymphocytosis progressing to CLL. In addition to patient 2, we observed a family history of lymphoma in patients 5 and 6 and a personal history of MGUS in patient 10 (Table 1). These findings suggest a potential link between the JAK2 p.R564 variants (both p.R564Q and p.R564L) and risk for lymphoid malignancies. The significance of these lymphoid phenotypes is crucial to highlight, particularly because several patients with R564 variants in our small cohort had a family or personal history of lymphoid malignancies. Thus, in addition to its association with MPNs, further exploration is warranted as to whether p.R564 variants also predispose to familial lymphoid malignancies.
Our work demonstrates several cases of MPN associated with germ line JAK2 variants other than at position V617. In addition, we report only the second family adding to Etheridge et al’s description of familial thrombocythemia due to JAK2 p.R564Q.17 We believe this has important clinical implications for the treatment of these rare cases or families. We did not observe a high proportion of major complications (eg, thrombosis, hemorrhage, and evolution to AML), however, we noted that these individuals do suffer from symptoms that profoundly interfere with their quality of life. Patients in our cohort reported a heightened burden of fatigue interfering with their daily functioning, night sweats, and bone pain. As assessed by MPN-SAF TSS (Table 1), patients’ symptoms were comparable with those generally observed in patients with advanced-stage MPN with the canonical somatic JAK2 V617F mutation. There exists a discernible gap in therapeutic options for patients afflicted with familial MPNs, which prompts an imperative for new interventions. The lower dose administration of JAK inhibitors, tailored to alleviate specific symptoms, may represent a promising avenue toward addressing this unmet medical need.
It appears that variants in JAK2 at p.R564 are associated with HT and potentially other myeloproliferative and lymphoproliferative disorders. While this is a small case series, it is of interest that all the females with p.R564 variants had clear HT phenotypes. On the other hand, many of the males did not demonstrate overt thrombocytosis, but rather other hematologic manifestations. The precise mechanisms behind these observations warrant further investigation. One could hypothesize that heightened recognition of iron deficiency and reactive thrombocytosis among females leads to a higher proportion of women being tested and diagnosed with thrombocytosis, potentially indicating a bias in testing and identification practices. Secondly, it is plausible that the augmented risk of MF and malignancy observed in males may skew the timing of identification toward the onset of MF or malignancy.36
Lastly, in terms of management, to date, there are no management guidelines or consensus protocols to direct the treatment of pathogenic germ line variants in JAK2. Although these are distinct from the common p.V617F variant, they may still enhance cell growth similar to the somatic p.V617F lesion.6,7,9,11,16,17 The germ line variants have kinase activity similar to p.V617F; the degree of growth-promoting effects due to counterregulatory mechanisms may differ, however. Although discrete management guidelines are a current unmet need, adopting management approaches from JAK2 p.V617F-positive MPNs (including symptom-directed therapeutic options such as JAK inhibitors) can be a feasible strategy to manage non–p.V617F MPN-associated JAK2 pathogenic variants. In Table 2, we provide our approach to the management of patients with germ line pathogenic variants in JAK2. The presence of germ line JAK2 mutations and a suggestive phenotype (cytosis) emerging as early as year 2 in our series further supports the recommendation that patients with early-onset MPNs should be offered genetic counseling and potentially germ line genetic testing. Overall, it is prudent for individuals with JAK2 germ line variants and their families to work closely with a multidisciplinary health care team, including genetic counselors, hematologists, cardiologists, and other specialists, to develop a personalized and comprehensive management plan, preferably at a major academic center.
Strategies for management of patients with novel germ line JAK2 mutations
| Periodic monitoring |
| Patients with JAK2 p.R564 germ line variants should be regularly monitored for any signs or symptoms of hematologic neoplasms. This should include periodic blood count monitoring, assessment of symptoms of fatigue, or unexplained weight loss. Regular blood tests and physical examinations are crucial for early detection and prompt intervention. |
| Treatment for thrombocythemia |
| If thrombocythemia is present, treatment with aspirin may be prescribed to reduce the risk of thromboses, as well as to decrease vasomotor symptom burden. |
| Mutation-specific treatment |
| JAK2 p.R564Q-expressing cells exhibit heightened sensitivity to the JAK inhibitor (JAKi) ruxolitinib, suggesting potential lower drug doses for treating patients with alternative JAK2 mutations. This understanding offers insights into potential novel treatment approaches to managing germ line cases of disease. Lower doses of ruxolitinib may be employed to suppress symptoms and reduce splenomegaly via inhibition of kinases. |
| Symptomatic treatment |
| In addition to lower dose of JAKi, supportive care can help manage symptoms and improve quality of life. This can include pain management, addressing cytoses and cytopenias, and managing other symptoms associated with the disease and its treatments. |
| Lifestyle modifications |
| We always encourage our patients to maintain a healthy lifestyle, including regular exercise, a balanced diet, and stress management. Avoiding tobacco and excessive alcohol consumption is encouraged which further helps to mitigate risk of thromboses. |
| Genetic counseling |
| Patients and families with JAK2 variants have benefited from genetic counseling to review concepts of hereditary and acquired genetic variation, to discuss benefits and limitations of genetic testing, to clarify potential risks for family members, and to talk through genetic testing results and potential uncertainties that may remain given the limited data outlining the implications of germ line JAK2 variants. |
| Psychological support |
| Our patients and their families benefit from psychological support, including counseling or support groups, to cope with the emotional challenges associated with newly diagnosed novel disorders and illnesses. |
| Indications for genetic testing |
| Genetic testing for novel germ line JAK2 variants could be considered for individuals with unexplained chronic thrombocytosis, particularly from a young age <40 y, in the absence of causes for secondary thrombocytosis such as iron deficiency or inflammatory disorders. Testing could also be considered for families including multiple family members with V617F-negative thrombocythemia. For patients identified with a suspected germ line non–V617F JAK2 variant (eg, variant with a VAF near 50%) confirmation of germ line status can be pursed through family member studies or testing on a nonhematopoietic sample such as cultured skin fibroblasts. |
| Periodic monitoring |
| Patients with JAK2 p.R564 germ line variants should be regularly monitored for any signs or symptoms of hematologic neoplasms. This should include periodic blood count monitoring, assessment of symptoms of fatigue, or unexplained weight loss. Regular blood tests and physical examinations are crucial for early detection and prompt intervention. |
| Treatment for thrombocythemia |
| If thrombocythemia is present, treatment with aspirin may be prescribed to reduce the risk of thromboses, as well as to decrease vasomotor symptom burden. |
| Mutation-specific treatment |
| JAK2 p.R564Q-expressing cells exhibit heightened sensitivity to the JAK inhibitor (JAKi) ruxolitinib, suggesting potential lower drug doses for treating patients with alternative JAK2 mutations. This understanding offers insights into potential novel treatment approaches to managing germ line cases of disease. Lower doses of ruxolitinib may be employed to suppress symptoms and reduce splenomegaly via inhibition of kinases. |
| Symptomatic treatment |
| In addition to lower dose of JAKi, supportive care can help manage symptoms and improve quality of life. This can include pain management, addressing cytoses and cytopenias, and managing other symptoms associated with the disease and its treatments. |
| Lifestyle modifications |
| We always encourage our patients to maintain a healthy lifestyle, including regular exercise, a balanced diet, and stress management. Avoiding tobacco and excessive alcohol consumption is encouraged which further helps to mitigate risk of thromboses. |
| Genetic counseling |
| Patients and families with JAK2 variants have benefited from genetic counseling to review concepts of hereditary and acquired genetic variation, to discuss benefits and limitations of genetic testing, to clarify potential risks for family members, and to talk through genetic testing results and potential uncertainties that may remain given the limited data outlining the implications of germ line JAK2 variants. |
| Psychological support |
| Our patients and their families benefit from psychological support, including counseling or support groups, to cope with the emotional challenges associated with newly diagnosed novel disorders and illnesses. |
| Indications for genetic testing |
| Genetic testing for novel germ line JAK2 variants could be considered for individuals with unexplained chronic thrombocytosis, particularly from a young age <40 y, in the absence of causes for secondary thrombocytosis such as iron deficiency or inflammatory disorders. Testing could also be considered for families including multiple family members with V617F-negative thrombocythemia. For patients identified with a suspected germ line non–V617F JAK2 variant (eg, variant with a VAF near 50%) confirmation of germ line status can be pursed through family member studies or testing on a nonhematopoietic sample such as cultured skin fibroblasts. |
Acknowledgments
This work made use of the High-Performance Computing Cluster provided by Cleveland Clinic Lerner Research Institute Computing Services.
B.J. is supported in part by National Cancer Institute grant R01CA257544 and National Institute of Diabetes and Digestive Kidney Diseases grant R01DK131923.
Authorship
Contribution: B.L.S. contributed to the study concept and design, provided patient data to the study, performed data collection and analysis, and wrote the manuscript; H.E.C. contributed to the study design, data interpretation, and revision of the manuscript; A.L.A., H.L., and J.M. provided patient data; Z.J.T. designed the Bioinformatics Pipeline used to analyze next-generation sequencing data; D.S.B. submitted the institutional review board protocol, contributed to data interpretation, and assisted with the revision of the manuscript; A.G. contributed to the study concept, data interpretation, and revision of the manuscript and also provided patient data; E.C. contributed to the study concept and design, performed data collection and analysis, and assisted with data interpretation and revision of the manuscript; B.J. performed in silico structural analysis, data interpretation, original writing of relevant sections, and reviewed and edited the manuscript; J.N. contributed to study concept and design, data analysis, data interpretation, and the writing of the manuscript; A.S. contributed to the study concept and design, data interpretation, and the writing of the manuscript; and all authors have substantially contributed to the content of the article and have agreed to the submission in its current format.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Abhay Singh, Cleveland Clinic, Taussig Cancer Institute, Leukemia and Myeloid Disorders Program, 9500 Euclid Ave, CA-60, Cleveland, OH 44195; email: singha21@ccf.org.
References
Author notes
B.L.S. and H.E.C. are joint first authors.
J.N. and A.S are joint senior authors.
Original data are available on request from the corresponding author, Abhay Singh (singha21@ccf.org).