TO THE EDITOR:
von Willebrand disease (VWD) is the most common inherited bleeding disorder and can be classified by quantitative or qualitative defects in the von Willebrand factor (VWF).1 VWD affects ∼1 in 100 based on abnormal laboratory parameters; however, ∼1 in 1000 have a bleeding phenotype.2 Bleeding manifestations vary and most commonly present as mucocutaneous bleeding, heavy menstrual bleeding, and easy bruising. If severe enough or left untreated, bleeding can lead to anemia or iron deficiency anemia. Diagnosing VWD is challenging because factor levels and activity are influenced by a multitude of variables including ABO-blood type, age, stress-induced hypothalamic-pituitary-adrenal axis changes, and environmental factors such as inflammation and hormone exposure.3,4 Furthermore, bleeding history can vary based on individual interpretation of bleeding symptoms and whether individuals have experienced hemostatic challenges. An association between hemoglobin, VWF activity, and factor VIII activity has been described in single-institution studies, but further research is needed.5,6
A retrospective review was performed across 5 medical centers to investigate the association between hemoglobin and VWF antigen and platelet binding activity assays. We hypothesized that a negative association exists. Data were collected on nonpregnant participants aged 0 to 21 years who were evaluated for underlying bleeding disorder between January 2001 and October 2022. Demographic information and laboratory results were collected. To be included, participants were required to have laboratory analysis drawn on at least 2 different calendar days. At minimum, each date included values for hemoglobin and a VWF antigen assay (VWF:Ag) or a platelet binding activity assay (ristocetin cofactor [VWF:RCo] or glycoprotein Ib binding [VWF:GpIbM]). Use of hormonal medications was also collected. Finally, information on concurrent medication use was collected, and patients were excluded if they were exposed to desmopressin or VWF concentrates in the 72 hours before laboratory draw. Absolute differences in value between the 2 time points for hemoglobin, VWF:Ag, VWF:RCo, VWF:GpIbM, age, and estrogen exposure were calculated. Simple and multiple linear regressions were performed for statistical analysis. The study was approved by the institutional review board of each participating center. REDCap software was used for data collection, and each institution contributed a minimum of 50 participants (range, 51-200).
In total, 481 participants were included in the analysis (Table 1). Most participants were female (n = 371 [77%]), and the mean age was 11.6 years (range, 0-21). Hemoglobin values were available for all participants, and VWF:Ag values were available for almost all (n = 478 [99%]). VWF:RCo was the more common platelet binding activity assay (n = 401; [83%]) than GpIbM binding activity (n = 38 [8%]). At time of first laboratory draw, VWF:Ag, VWF:RCo, and VWF:GpIbM assays were <50% in 56 (12%), 106 (22%), and 13 (3%) of 481 participants, respectively. All participants with a VWF:Ag <50% had a corresponding activity level <50%, except for 1 participant who did not have activity testing performed. In total, 119 of 481 participants (24.7%) were diagnosed with VWD on first draw. An additional 47 of 481 participants (9.8%) were diagnosed with VWD on the second draw, with an average difference in hemoglobin of 2.8 g/dL between the 2 draws for this subgroup. A total of 459 of 481 participants (95%) had information on estrogen exposure at both time points. Of those 459 participants, 370 (80%) had no exposure at either point, 61 (13%) had no exposure at initial draw but were on estrogen containing therapy at follow-up, and 19 (4%) were on estrogen containing therapy at initial draw but transitioned to progesterone only therapy at time of follow-up. Eighty-eight participants (19%) were on progesterone only therapy at either 1 or both time points.
Participant characteristics
| . | n (%) . |
|---|---|
| Total participants | 481 |
| Sex | |
| Male | 108 (22.5) |
| Female | 371 (77.1) |
| Participants with VWF:Ag | 478 (99.4) |
| Participants with VWF:RCo | 401(83.4) |
| Participants with VWF:GpIbM | 38 (7.9) |
| Participants with information on estrogen use | 459 |
| No use at either time point | 343 (74.7) |
| Use at both time points | 32 (7.0) |
| Use at first time point only | 23 (5.0) |
| Use at second time point only | 61 (13.3) |
| Initial VWF:Ag >100% | 182/478 (38) |
| Initial VWF:RCo >100% | 101/401 (25) |
| Initial VWF:GpIbM >100% | 13/38 (34) |
| . | n (%) . |
|---|---|
| Total participants | 481 |
| Sex | |
| Male | 108 (22.5) |
| Female | 371 (77.1) |
| Participants with VWF:Ag | 478 (99.4) |
| Participants with VWF:RCo | 401(83.4) |
| Participants with VWF:GpIbM | 38 (7.9) |
| Participants with information on estrogen use | 459 |
| No use at either time point | 343 (74.7) |
| Use at both time points | 32 (7.0) |
| Use at first time point only | 23 (5.0) |
| Use at second time point only | 61 (13.3) |
| Initial VWF:Ag >100% | 182/478 (38) |
| Initial VWF:RCo >100% | 101/401 (25) |
| Initial VWF:GpIbM >100% | 13/38 (34) |
| . | n (SD) . |
|---|---|
| Mean age, y | |
| First laboratory time point | 11 (5.5) |
| Second laboratory time point | 12.3 (5.5) |
| Average time between laboratory draws, d | 467 (794) |
| Median hemoglobin first time point, g/dL | 11.6 (2.7) |
| Median hemoglobin second time point, g/dL | 12.6 (2.3) |
| Median change in hemoglobin between draws, g/dL | 0.9 (2.6) (range, −6.5 to 11.0) |
| . | n (SD) . |
|---|---|
| Mean age, y | |
| First laboratory time point | 11 (5.5) |
| Second laboratory time point | 12.3 (5.5) |
| Average time between laboratory draws, d | 467 (794) |
| Median hemoglobin first time point, g/dL | 11.6 (2.7) |
| Median hemoglobin second time point, g/dL | 12.6 (2.3) |
| Median change in hemoglobin between draws, g/dL | 0.9 (2.6) (range, −6.5 to 11.0) |
SD, standard deviation.
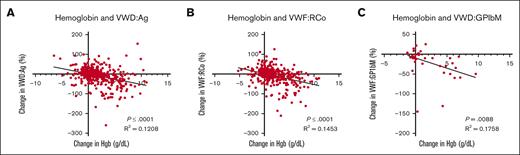
A statistically significant negative association between change in hemoglobin and change in VWF:Ag, VWF:Rco, and VWF:GpIbM binding was found (P < .0001; P < .0001; and P = .0088, respectively, Figure 1) in bivariate linear analysis. Incorporating change in estrogen and change in age into a multiple linear regression model, this association held (P < .001; P < .001; and P < .002). For every 1 g/dL rise in hemoglobin, our model predicts a decrease of 5.3%, 5.6%, and 5.1% in VWF:Ag, VWF:Rco, and VWF:GpIbM values, respectively.
Relationship between hemoglobin values and VWF assays. Simple linear regression of change in (A) hemoglobin and VWF:Ag, (B) hemoglobin and VWF:RCo, (C) hemoglobin and VWF:GPIbM.
Relationship between hemoglobin values and VWF assays. Simple linear regression of change in (A) hemoglobin and VWF:Ag, (B) hemoglobin and VWF:RCo, (C) hemoglobin and VWF:GPIbM.
It has been reported that VWF:Ag and VWF:RCo values >100% have a high negative predictive value for a diagnosis of VWD, with both single and multicenter studies validating these findings.7,8 In our study, 182 of 478 (38%), 101 of 401 (25%), and 13 of 38 participants (34%) presented with values >100% for VWF:Ag, VWF:RCo, and VWF:GpIbM, respectively; so, a bleeding disorder would not have been initially ruled out in the majority of participants using this cutoff. From those patients with initial values >100%, only 5 participants had laboratory tests suggestive of VWD on follow-up testing using a diagnostic threshold of <50% for any VWD assay.9 All 5 participants were anemic with hemoglobin <10 g/dL at presentation. Using a cutoff of 100%, our data showed a negative predictive value of 100% if initial VWF:Ag (n = 76) or VWF:RCo (n = 4) were elevated in isolation. There were no scenarios of isolated VWF:GpIbM activity >100% at presentation; however, fewer participants had VWF:GpIbM testing performed. If any assay was >100% at presentation, the negative predictive value was 97.3%. Five participants presented with all 3 variables >100%, in which a negative predictive value of 100% was found. Using >100% as a cutoff, we found sensitivities of 90%, 97%, and 91% for VWF:Ag, VWF:RCo, and VWF:GpIbM, respectively. Our study supports previous findings regarding the high negative predictive value of VWF assay values >100%. Furthermore, our findings highlight the importance of repeating testing to avoid missing VWD diagnoses in patients who present with elevated VWF in the setting of anemia.
Limitations of our study include the variability of VWF assays used across centers, which reduced the total number of participants available for each specific analysis. The VWF:RCo assay is highly variable with low sensitivity and represented the majority of VWF activity assays used because the newly recommended VWF:GpIbM assay is not yet widely available.10 Furthermore, markers of inflammation were not obtained, which could help identify those patients whose elevated laboratory values were secondary to an underlying inflammatory process, thus confounding the data. We did not collect data on red blood cell transfusion but hypothesize that this association exists independent of the method of increasing hemoglobin. The large age range and inclusion of participants who both do and do not have the potential to menstruate could have also confounded our data. We did perform a subanalysis looking at males of all ages and found a similar association, however, this was not statistically significant. It is difficult to know whether this lack of significance is physiologically based or whether this was due to much smaller number of males (107 vs 374) and much smaller average change in hemoglobin in males (0.6 g/dL vs 1.8 g/dL). All data were collected via retrospective chart review, which presents a limitation because the data rely on participant recall and investigator thoroughness to ensure the most accurate history was collected. Furthermore, additional variables could be present and unaccounted for.
This multi-institutional study found a negative association between hemoglobin values and VWF antigen level, ristocetin cofactor activity, and glycoprotein Ib binding activity. Our data do not demonstrate causation, but if a causal relationship does exist, we hypothesize that it could be secondary to physiologic stress in the setting of anemia or an alternate mechanism. It is known that VWF initiates hemostasis under high flow conditions.11 One of the first physiologic responses to anemia is an increase in cardiac output through tachycardia and high velocity flow, which may, in turn, lead to increased activation of VWF. Because many patients are tested for VWD while anemic, whether acutely or more chronically, these results highlight the potential of missing an underlying VWD diagnosis in patients presenting with anemia and normal VWF levels. Ensuring follow-up for patients with anemia whose history is clinically suggestive is crucial in definitively ruling out VWD.
Contribution: J.S., C.T., A.W., L.A., M.M., Y.C., and M.B. performed the research; and J.S. and A.C.W. designed the research and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Jillian Simoneau, University of Michigan, 1500 E Medical Center Dr, MPB 4202, Ann Arbor, MI 48109; email: jlapin@med.umich.edu.
References
Author notes
Data are available upon reasonable request from the corresponding author, Jillian Simoneau (jlapin@med.umich.edu).
Presented as an abstract at the International Society on Thrombosis and Haemostasis 2023 Congress, Montreal, Quebec, 26 June 2023.