Key Points
Exposure to prior cytotoxic therapy in patients with CCUS independently accounts for inferior PFS and OS.
Patients with CCUS who have received cytotoxic therapy have distinct clinical features, which be considered a unique entity called t-CCUS.
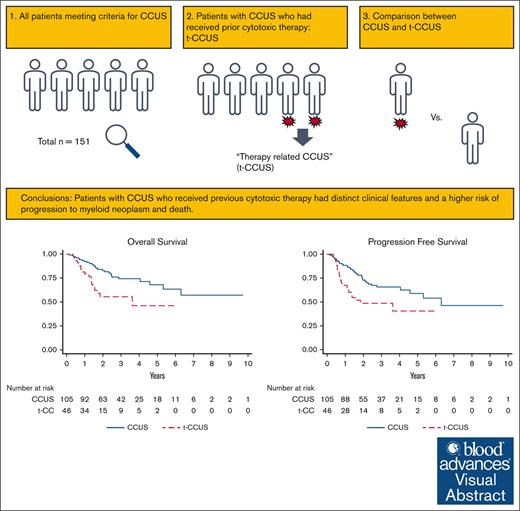
Visual Abstract
Clonal cytopenia of undetermined significance (CCUS) is defined by a myeloid driver mutation in the context of otherwise unexplained cytopenia. CCUS has an inherent risk of progressing to myeloid neoplasm. However, it is unknown how exposure to previous cytotoxic therapy may impact the risk of progression and survival. We stratified patients with CCUS by prior exposure to DNA-damaging therapy. Of 151 patients, 46 (30%) had received cytotoxic therapy and were classified as therapy-related CCUS (t-CCUS), whereas 105 (70%) had de novo CCUS. A lower proportion of t-CCUS had hypercellular marrows (17.8% vs 44.8%, P = .002) but had higher median bone marrow blast percentages. After a median follow-up of 2.2 years, t-CCUS had significantly shorter progression-free survival (PFS, 1.8 vs 6.3 years; hazard ratio [HR], 2.1; P = .007) and median overall survival (OS; 3.6 years vs not reached; HR, 2.3; P = .007) compared with CCUS. Univariable and multivariable time-to-event analyses showed that exposure to cytotoxic therapy independently accounted for inferior PFS and OS. Despite the similarities in clinical presentation between CCUS and t-CCUS, we show that exposure to prior cytotoxic therapies was an independent risk factor for inferior outcomes. This suggests that t-CCUS represents a unique clinical entity that needs more stringent monitoring or earlier intervention strategies.
Introduction
Improvements in sequencing methods has led to increased recognition of precursor states for myeloid neoplasms (MN). These precursor states fall under the umbrella of clonal hematopoiesis (CH), which is defined as clonal expansion in a myeloid driver mutation originating in hematopoietic stem and progenitor cells.1 CH inherently carries a risk for progression to MN.2,3 Cytotoxic therapies are not only associated with a characteristic genetic landscape but also shapes the fitness of the clone.3 However, the proportion of patients with CH developing therapy-related MNs (t-MNs) is relatively low and latency is long.4,5
Clonal cytopenia of undetermined significance (CCUS) is defined as the presence of at least 1 line of unexplainable cytopenia(s) lasting for ≥4 months, along with a clonal abnormality, without evidence of a bona fide MN.1 The risk of leukemic progression from CCUS is higher, and latency shorter. CCUS, therefore, is considered an intermediary premalignant state between CH and MN. Although the impact of therapy on CH and the resultant t-MN development has been studied extensively,3,6 its impact on CCUS remains uncharacterized.
In CCUS, common mutations encountered include TET2 (23.8%), DNMT3A (13.3%), SRSF2 (10.1%), ASXL1 (8.3%), and U2AF1 (4.6%).7 In a previous single institution study, the median progression free survival (PFS) for patients with CCUS was 17.1 months, with an estimated 2-year overall survival (OS) of 73%.8 However, the impact of previous DNA-damaging therapies on characteristics and outcomes of CCUS has not been studied.
t-MNs are aggressive hematological neoplasms that develop as a complication from exposure to DNA-damaging therapies, and carry particularly poor survival. The DNA-damaging agents include cytotoxic chemotherapy used either alone or in combination with radiation or immunosuppressive therapies (eg, azathioprine).9 Although t-MN is rare, with an estimated population incidence of 0.62 per 100 000,10 its incidence is expected to rise as the population ages and the prevalence of cancer survivors increase. Patients who develop t-MN have an estimated median survival of ≤15 months, because they are more likely than de novo acute myeloid leukemia to carry high-risk karyotypes and TP53 mutations.11 While studying patients with t-MN, it was noted that a subset of patients who received DNA-damaging therapies developed unexplained cytopenia but did not have t-MNs. We recently described this group as a clinical entity of therapy-related CCUS (t-CCUS).11 t-CCUS had a significantly better survival compared to World Health Organization–defined t-MN, suggesting that it is a distinct entity from t-MN.11 The International Consensus Classification (ICC) of Hematological Malignancies adapted their “therapy-related” qualifier to CCUS.12 In contrast, the 5th edition of the World Health Organization classification of hematological neoplasms did not comment on the status of t-CCUS.1 This discrepancy suggests lack of consensus, rooted in the lack of relevant data.
Therefore, the aims of this study were to characterize the impact of prior cytotoxic therapy on clinicopathological features and the risk of progression to MN and death.
Methods
This was a retrospective review of all adult patients diagnosed with CCUS at Mayo Clinic Enterprise (Rochester, MN, Jacksonville, FL, and Scottsdale, AZ) between 2010 and 2022. CCUS was defined using ICC criteria12: cytopenia was defined as hemoglobin (Hb) <13 g/dL in males and <12 g/dL in females, absolute neutrophil count of <1.8 × 109/L, and platelets of <150 × 109/L. All patients had presence of myeloid driver mutation or a nonmyelodysplastic syndrome (MDS) defining clonal cytogenetic alteration. MDS-defining cytogenetics were defined per the ICC criteria; that is, patients with isolated 5q deletion, monosomy 7 or 7q deletion, or complex karyotype were determined to have MDS and were excluded from further analysis.12 Pathology was reviewed (D.C.) to ensure that none of the pathologic diagnosis of patients collected met the diagnostic criteria for MN.
In addition, patients were classified as having t-CCUS if they received DNA-damaging agents in the form of either cytotoxic chemotherapy (supplemental Table 1), radiation (including radioligand therapy and field radiation), or a combination of the aforementioned, before CCUS diagnosis.
Demographic and clinical information was extracted at the time of diagnosis, including age, sex, smoking status, blood count, bone marrow blast count and cellularity, type of cytotoxic therapy received, type of therapy after CCUS diagnosis, and next-generation sequencing (NGS). NGS was performed on DNA extracted from bone marrow aspirates at Mayo Clinic using either a clinical targeted myeloid (NGS HemOnc, versions 1-3; supplemental Tables 2-4) or an expanded gene research–based panel (supplemental Table 5). Further information regarding the sensitivity of these panels can be found in supplemental Methods. Of 147 patients with available NGS data, 9 underwent the expanded research panel, with the rest being clinical.
The cause of death was determined based on provider documentation from electronic medical record and were found to be generally (1) due to hematological malignancy; (2) cardiac related; (3) due to infection, or (4) multifactorial or due to indeterminate causes. Because a combination of the aforementioned could contribute to death and there is ambiguity in this determination via provider documentation, hematological malignancy would only be considered the cause of death if the patient progressed to leukemia and chose hospice as a direct result of the new diagnosis or passed away from complications of chemotherapy related to hematological malignancy. Infection or cardiac was considered cause of death only if the infection or cardiac event was not a direct result of active cancer treatment.
Descriptive and summary statistics for numerical values were calculated using Wilcoxon rank sum test. Fisher exact test was used to compare categorical data between groups. Time-to-event outcomes were calculated using a Cox proportional hazard model. Univariable and multivariable analysis for time-to-event data was performed using the same. Subset multivariable analysis was based on the most significant findings from initial univariable analysis. PFS was defined as the interval from the diagnosis to progression to MN or death. OS was defined as the interval from the diagnosis to death from any cause. Statistical analysis was performed using Stata/MP version 16, and survival curves were generated from the same.
This study was reviewed and approved by the institutional review board at Mayo Clinic, Rochester, MN.
Results
Clinical and pathological characteristics
Of 151 patients, 105 (70%) met criteria for CCUS and 46 (30%) for t-CCUS. For those classified as having t-CCUS, 17 patients (37%) received prior cytotoxic chemotherapy, 17 (37%) received combined chemotherapy and radiation, 11 (24%) received radiation therapy alone, and 1 patient received peptide receptor radionuclide therapy. The cancer types for the t-CCUS group are listed in supplemental Table 6. The median age at diagnosis for the entire cohort was 68 years, with the majority being male (60%), and was not different between CCUS and t-CCUS (P = .208). Patients with history of, or active, tobacco use were comparable between the 2 cohorts (41.8% vs 44.4%; P = .857). There were no differences between median blood counts at diagnosis between the 2 groups (Table 1). The median red cell distribution width (RDW) at diagnosis was 15% (range, 11.7%-26.7%) for both CCUS and t-CCCUS. Patients with CCUS were more likely to have hypercellular bone marrows at diagnosis (median, 45% vs 17%; P = .002), whereas patients with t-CCUS were more likely to have hypocellular bone marrows at diagnosis (median, 20% vs 5%; P = .011). t-CCUS was associated with higher bone marrow blasts at diagnosis (median, 1% vs 0%; P = .026). There were no differences in number of patients with ringed sideroblasts on their bone marrow biopsy at diagnosis (median, 14% and 11% for CCUS and t-CCUS, respectively; P = .926). We applied the recently proposed clonal hematopoiesis risk score (CHRS) to our cohort,13 acknowledging that all of our patients already have CCUS, which incurs a higher risk. The median CHRS score was 12 (range, 8.5-15.5). The majority (90%) of patients were evenly distributed between intermediate- and high-risk CHRS, and this was not different between CCUS and t-CCUS (Table 1).
Clinicopathological features of patients with de novo CCUS vs t-CCUS
| Variables (median or n; range or %) . | All patients (n = 151) . | CCUS (n = 105) . | t-CCUS (n = 46) . | P value . |
|---|---|---|---|---|
| Age (y), median (range) | 68 (20-99) | 68 (20-99) | 67 (24-83) | .268 |
| Sex (male), n (%) | 91 (60.3) | 67 (63.8) | 24 (52.2) | .208 |
| Active or previous tobacco use, n (%) | 63 (42.6) | 43 (41.8) | 20 (44.4) | .857 |
| Hemoglobin (g/dL), median (range) | 10.9 (6.7-15.9) | 10.6 (6.9-15.9) | 11.4 (6.7-14.8) | .831 |
| MCV (fL), median (range) | 99.2 (80.4-126.7) | 98.4 (80.4-123.2) | 101.4 (86-126.7) | .311 |
| WBC × 109/L, median (range) | 3.5 (0.8-32) | 3.5 (1-32) | 3.5 (0.8-13.4) | .689 |
| ANC × 109/L, median (range) | 1.6 (0.04-19.9) | 1.6 (0.04-19.9) | 1.7 (0.12-9.2) | .854 |
| AMC × 109/L, median (range) | 0.4 (0.01-6.2) | 0.4 (0.01-6.2) | 0.4 (0.03-3.6) | .681 |
| Platelets × 109/L, median(range) | 114 (15-595) | 119 (15-595) | 105 (26-222) | .101 |
| RDW-CV (%), median (range) | 15 (11.7-26.7) | 15 (11.7-26.7) | 15 (12-24.4) | .923 |
| Bone marrow cellularity, n (%) | ||||
| Hypercellular | 55 (36.4) | 47 (44.8) | 8 (17.4) | .002 |
| Hypercellular with atypia | 29 (19.2) | 21 (20) | 8 (17.4) | .824 |
| Hypocellular | 14 (9.3) | 5 (4.8) | 9 (19.6) | .011 |
| Normocellular | 50 (33.1) | 30 (28.6) | 20 (43.5) | .089 |
| Unknown | 3 (1.9) | 2 (1.9) | 1 (2.2) | - |
| Bone marrow blasts (%), median (range) | 0 (0-5)∗ | 0 (0-4) | 1 (0-5) | .026 |
| Abnormal cytogenetics, n (%) | 37 (24.5) | 26 (24.8) | 11 (23.9) | .375 |
| Presence of ringed sideroblasts, n (%) | 26 (13.3) | 15 (14.3) | 5 (10.9) | .926 |
| Number of mutations, median (range) | 2 (0-5) | 2 (0-4) | 1 (0-5) | .551 |
| VAF (%), median (range) | 39 (5-92) | 39 (5-82) | 38.5 (6-92) | .858 |
| Management of disease, n (%) | ||||
| Surveillance only | 112 (74.2) | 75 (71.4) | 37 (80.4) | |
| Hypomethylating agent | 6 (4) | 4 (3.8) | 2 (4.4) | |
| Supportive care (G-CSF, ESA, TPO, and transfusions) | 23 (15.2) | 17 (16.2) | 6 (13) | |
| Immunosuppressive agent | 6 (4) | 6 (5.7) | 0 (0) | |
| Bone marrow transplantation | 0 (0) | 0 (0) | 0 (0) | .544 |
| Vitamin C clinical trial | 4 (2.7) | 3 (2.9) | 1 (2.2) | |
| Progression to MN, n (%) | 28 (17.9) | 16 (15.2) | 12 (26.1) | .171 |
| CMML | 6 (4) | 4 (3.8) | 2 (4.3) | |
| MDS | 15 (9.9) | 8 (7.6) | 7 (15.2) | |
| AML | 6 (4) | 3 (2.9) | 3 (6.5) | |
| BPDCN | 1 (0.6) | 1 (1) | 0 (0) | .491 |
| Follow-up (y), median (range) | 2.2 (0.05-12.6) | 2.5 (0.05-12.6) | 1.5 (0.1-5.9) | .002 |
| Total deaths, n (%) | 44 (29.1) | 26 (24.8) | 18 (39.1) | |
| Malignancy related | 14 (31.8) | 6 (23.1) | 8 (44.4) | |
| Cardiac related | 8 (18.2) | 4 (15.4) | 4 (22.2) | .299 |
| Infection related | 10 (22.7) | 8 (30.1) | 2 (11.1) | |
| Other/multifactorial | 12 (27.3) | 8 (30.1) | 4 (22.2) | |
| CHRS score, median (range) | 12 (8.5-15.5) | 12 (8.5-15.5) | 12 (8.5-15.5) | .574 |
| CHRS risk group, n (%) | ||||
| Low | 16 (10.6) | 9 (8.6) | 7 (15.2) | |
| Intermediate | 70 (46.4) | 50 (47.6) | 20 (43.5) | |
| High | 65 (43.1) | 46 (43.8) | 19 (41.3) | .478 |
| Variables (median or n; range or %) . | All patients (n = 151) . | CCUS (n = 105) . | t-CCUS (n = 46) . | P value . |
|---|---|---|---|---|
| Age (y), median (range) | 68 (20-99) | 68 (20-99) | 67 (24-83) | .268 |
| Sex (male), n (%) | 91 (60.3) | 67 (63.8) | 24 (52.2) | .208 |
| Active or previous tobacco use, n (%) | 63 (42.6) | 43 (41.8) | 20 (44.4) | .857 |
| Hemoglobin (g/dL), median (range) | 10.9 (6.7-15.9) | 10.6 (6.9-15.9) | 11.4 (6.7-14.8) | .831 |
| MCV (fL), median (range) | 99.2 (80.4-126.7) | 98.4 (80.4-123.2) | 101.4 (86-126.7) | .311 |
| WBC × 109/L, median (range) | 3.5 (0.8-32) | 3.5 (1-32) | 3.5 (0.8-13.4) | .689 |
| ANC × 109/L, median (range) | 1.6 (0.04-19.9) | 1.6 (0.04-19.9) | 1.7 (0.12-9.2) | .854 |
| AMC × 109/L, median (range) | 0.4 (0.01-6.2) | 0.4 (0.01-6.2) | 0.4 (0.03-3.6) | .681 |
| Platelets × 109/L, median(range) | 114 (15-595) | 119 (15-595) | 105 (26-222) | .101 |
| RDW-CV (%), median (range) | 15 (11.7-26.7) | 15 (11.7-26.7) | 15 (12-24.4) | .923 |
| Bone marrow cellularity, n (%) | ||||
| Hypercellular | 55 (36.4) | 47 (44.8) | 8 (17.4) | .002 |
| Hypercellular with atypia | 29 (19.2) | 21 (20) | 8 (17.4) | .824 |
| Hypocellular | 14 (9.3) | 5 (4.8) | 9 (19.6) | .011 |
| Normocellular | 50 (33.1) | 30 (28.6) | 20 (43.5) | .089 |
| Unknown | 3 (1.9) | 2 (1.9) | 1 (2.2) | - |
| Bone marrow blasts (%), median (range) | 0 (0-5)∗ | 0 (0-4) | 1 (0-5) | .026 |
| Abnormal cytogenetics, n (%) | 37 (24.5) | 26 (24.8) | 11 (23.9) | .375 |
| Presence of ringed sideroblasts, n (%) | 26 (13.3) | 15 (14.3) | 5 (10.9) | .926 |
| Number of mutations, median (range) | 2 (0-5) | 2 (0-4) | 1 (0-5) | .551 |
| VAF (%), median (range) | 39 (5-92) | 39 (5-82) | 38.5 (6-92) | .858 |
| Management of disease, n (%) | ||||
| Surveillance only | 112 (74.2) | 75 (71.4) | 37 (80.4) | |
| Hypomethylating agent | 6 (4) | 4 (3.8) | 2 (4.4) | |
| Supportive care (G-CSF, ESA, TPO, and transfusions) | 23 (15.2) | 17 (16.2) | 6 (13) | |
| Immunosuppressive agent | 6 (4) | 6 (5.7) | 0 (0) | |
| Bone marrow transplantation | 0 (0) | 0 (0) | 0 (0) | .544 |
| Vitamin C clinical trial | 4 (2.7) | 3 (2.9) | 1 (2.2) | |
| Progression to MN, n (%) | 28 (17.9) | 16 (15.2) | 12 (26.1) | .171 |
| CMML | 6 (4) | 4 (3.8) | 2 (4.3) | |
| MDS | 15 (9.9) | 8 (7.6) | 7 (15.2) | |
| AML | 6 (4) | 3 (2.9) | 3 (6.5) | |
| BPDCN | 1 (0.6) | 1 (1) | 0 (0) | .491 |
| Follow-up (y), median (range) | 2.2 (0.05-12.6) | 2.5 (0.05-12.6) | 1.5 (0.1-5.9) | .002 |
| Total deaths, n (%) | 44 (29.1) | 26 (24.8) | 18 (39.1) | |
| Malignancy related | 14 (31.8) | 6 (23.1) | 8 (44.4) | |
| Cardiac related | 8 (18.2) | 4 (15.4) | 4 (22.2) | .299 |
| Infection related | 10 (22.7) | 8 (30.1) | 2 (11.1) | |
| Other/multifactorial | 12 (27.3) | 8 (30.1) | 4 (22.2) | |
| CHRS score, median (range) | 12 (8.5-15.5) | 12 (8.5-15.5) | 12 (8.5-15.5) | .574 |
| CHRS risk group, n (%) | ||||
| Low | 16 (10.6) | 9 (8.6) | 7 (15.2) | |
| Intermediate | 70 (46.4) | 50 (47.6) | 20 (43.5) | |
| High | 65 (43.1) | 46 (43.8) | 19 (41.3) | .478 |
Variables in bold indicate P < 0.05. AMC, absolute monocyte count; AML, acute myeloid leukemia; ANC, absolute neutrophil count; BPDCN, blastic plasmacytoid dendritic cell neoplasm; CMML, chronic myelomonocytic leukemia; CV, coefficient of variation; ESA, erythropoietin stimulating agents; G-CSF, granulocyte-colony stimulating factor; MCV, mean corpuscular volume; TPO, thrombopoietin receptor agonists; WBC, white blood cell count.
One patient with blasts of 5% had received G-CSF treatment before bone marrow biopsy.
Cytogenetic and NGS findings
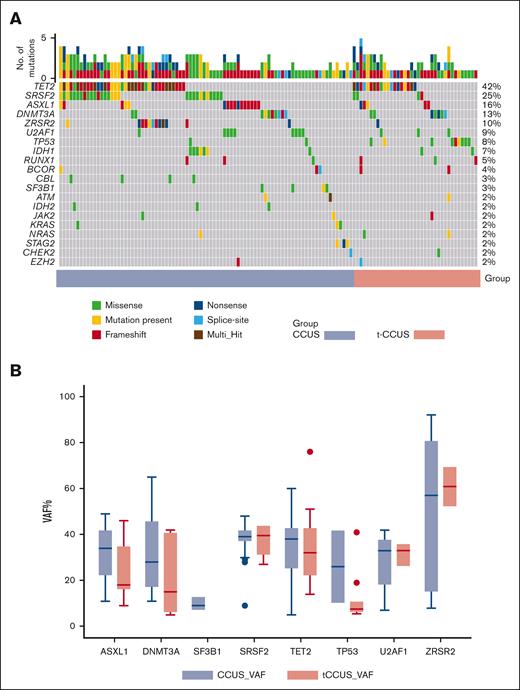
The median percent of patients with abnormal cytogenetics (not meeting criteria for MDS-defining cytogenetics) at diagnosis was 25% and 24% for CCUS and t-CCUS, respectively (P = .375). There were no significant differences in the number of mutations on NGS, with a median of 2 mutations for CCUS and 1 mutation for t-CCUS (P = .551). The most common mutations in the entire cohort were TET2 (37%), SRSF2 (24%), ASXL1 (14%), DNMT3A (12%), ZRSR2 (9%), U2AF1 (8%), and TP53 (7%; Table 2). The CCUS group had more SRSF2 mutations (30% vs 9%, P = .003), whereas t-CCUS was disproportionally enriched in TP53 mutations (20% vs 2%, P = .001; Figure 1A). There were no differences in variant allele frequency (VAF) of all cumulative mutations between the 2 groups (39% vs 38.5% in CCUS and t-CCUS, respectively; P = .858; Table 1). This remained consistent when comparing VAF within each specific gene, including those that were differentially enriched between the 2 groups (supplemental Table 7). For example, the median VAF of SRSF2 mutations was 39% in each cohort (P = .775; Figure 1B). The median VAF in TP53 mutations was numerically higher in CCUS compared with t-CCUS; however, the difference was not statistically significant (26% vs 8%; P = .186). This statistical analysis is limited by the fact that only 2 patients with CCUS harbored TP53mut. Updated myeloid guidelines consider a VAF of >49% as presumptive evidence of biallelic TP53.1,9 All TP53mut patients in our cohort with available cytogenetics (n = 10) had diploid cytogenetics, and none had a VAF of >49%.
NGS results of de novo CCUS vs t-CCUS
| NGS analysis, n (%) . | All patients (n = 151) . | CCUS (n = 105) . | t-CCUS (n = 46) . | P value . |
|---|---|---|---|---|
| Epigenetic regulators | ||||
| TET2 | 56 (37.1) | 38 (36.2) | 18 (39.1) | .855 |
| IDH1 | 9 (6) | 7 (6.7) | 2 (4.4) | .723 |
| IDH2 | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| DNMT3A | 18 (11.9) | 11 (10.5) | 7 (15.2) | .422 |
| Chromatin regulators | ||||
| ASXL1 | 21 (13.9) | 16 (15.2) | 5 (10.9) | .612 |
| Spliceosome factors | ||||
| SRSF2 | 36 (23.8) | 32 (30.4) | 4 (8.7) | .003 |
| SF3B1 | 4 (2.6) | 4 (3.8) | 0 (0) | .314 |
| U2AF1 | 12 (8) | 9 (8.6) | 3 (6.5) | 1.000 |
| ZRSR2 | 13 (8.6) | 11 (10.5) | 2 (4.4) | .346 |
| Transcription factors | ||||
| RUNX1 | 6 (4) | 3 (2.9) | 3 (6.5) | .369 |
| Cell signaling | ||||
| KRAS | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| NRAS | 3 (2) | 1 (1) | 2 (4.4) | .220 |
| CBL | 4 (2.7) | 4 (3.8) | 0 (0) | .314 |
| JAK2 | 3 (2) | 2 (1.9) | 1 (2.2) | 1.000 |
| KIT | 1 (0.6) | 0 (0) | 1 (2.2) | .305 |
| MPL | 2 (1.3) | 0 (0) | 2 (4.3) | .091 |
| NOTCH1 | 1 (0.7) | 0 (0) | 1 (2.2) | .305 |
| WT1 | 2 (1.3) | 2 (1.9) | 0 (0) | 1.000 |
| Tumor suppressor genes | ||||
| TP53 | 11 (7.3) | 2 (1.9) | 9 (19.6) | <.001 |
| Others | ||||
| SETBP1 | 2 (1.3) | 2 (1.9) | 0 (0) | 1.000 |
| ATM | 3 (2) | 2 (1.9) | 1 (2.2) | 1.000 |
| BCOR | 5 (3.3) | 3 (2.9) | 2 (4.4) | .641 |
| STAG2 | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| CHEK2 | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
| PHF6 | 1 (0.6) | 0 (0) | 1 (2.2) | .305 |
| PTEN | 1 (0.6) | 1 (1) | 0 (0) | 1.000 |
| PPM1D | 2 (1.3) | 0 (0) | 2 (4.4) | .091 |
| EZH2 | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
| ITK | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
| NGS analysis, n (%) . | All patients (n = 151) . | CCUS (n = 105) . | t-CCUS (n = 46) . | P value . |
|---|---|---|---|---|
| Epigenetic regulators | ||||
| TET2 | 56 (37.1) | 38 (36.2) | 18 (39.1) | .855 |
| IDH1 | 9 (6) | 7 (6.7) | 2 (4.4) | .723 |
| IDH2 | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| DNMT3A | 18 (11.9) | 11 (10.5) | 7 (15.2) | .422 |
| Chromatin regulators | ||||
| ASXL1 | 21 (13.9) | 16 (15.2) | 5 (10.9) | .612 |
| Spliceosome factors | ||||
| SRSF2 | 36 (23.8) | 32 (30.4) | 4 (8.7) | .003 |
| SF3B1 | 4 (2.6) | 4 (3.8) | 0 (0) | .314 |
| U2AF1 | 12 (8) | 9 (8.6) | 3 (6.5) | 1.000 |
| ZRSR2 | 13 (8.6) | 11 (10.5) | 2 (4.4) | .346 |
| Transcription factors | ||||
| RUNX1 | 6 (4) | 3 (2.9) | 3 (6.5) | .369 |
| Cell signaling | ||||
| KRAS | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| NRAS | 3 (2) | 1 (1) | 2 (4.4) | .220 |
| CBL | 4 (2.7) | 4 (3.8) | 0 (0) | .314 |
| JAK2 | 3 (2) | 2 (1.9) | 1 (2.2) | 1.000 |
| KIT | 1 (0.6) | 0 (0) | 1 (2.2) | .305 |
| MPL | 2 (1.3) | 0 (0) | 2 (4.3) | .091 |
| NOTCH1 | 1 (0.7) | 0 (0) | 1 (2.2) | .305 |
| WT1 | 2 (1.3) | 2 (1.9) | 0 (0) | 1.000 |
| Tumor suppressor genes | ||||
| TP53 | 11 (7.3) | 2 (1.9) | 9 (19.6) | <.001 |
| Others | ||||
| SETBP1 | 2 (1.3) | 2 (1.9) | 0 (0) | 1.000 |
| ATM | 3 (2) | 2 (1.9) | 1 (2.2) | 1.000 |
| BCOR | 5 (3.3) | 3 (2.9) | 2 (4.4) | .641 |
| STAG2 | 3 (2) | 3 (2.9) | 0 (0) | .553 |
| CHEK2 | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
| PHF6 | 1 (0.6) | 0 (0) | 1 (2.2) | .305 |
| PTEN | 1 (0.6) | 1 (1) | 0 (0) | 1.000 |
| PPM1D | 2 (1.3) | 0 (0) | 2 (4.4) | .091 |
| EZH2 | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
| ITK | 2 (1.3) | 1 (1) | 1 (2.2) | .518 |
Variables in bold indicate P < 0.05.
Genetic landscape of clonal cytopenia of undetermined significance (CCUS) stratified by prior therapy. (A) Genetic characteristics and (B) variance allele frequency (VAF) for select genes in patients with de novo or therapy-related CCUS. MCV, mean corpuscular volume.
Genetic landscape of clonal cytopenia of undetermined significance (CCUS) stratified by prior therapy. (A) Genetic characteristics and (B) variance allele frequency (VAF) for select genes in patients with de novo or therapy-related CCUS. MCV, mean corpuscular volume.
Outcomes after CCUS diagnosis
Management of CCUS and patients with t-CCUS
After CCUS diagnosis, the subsequent treatment was at the treating physician’s discretion. Overall, there were no differences in the observed treatment modalities chosen between the 2 groups. A majority in both groups underwent surveillance only (71% vs 80% in CCUS and t-CCUS, respectively), with the second most common modality being supportive care (growth factor support and/or transfusion, 15% vs 6% in CCUS and t-CCUS, respectively). A small number of patients in both groups were treated with a hypomethylating agent (3.8% vs 4.4% in CCUS and t-CCUS, respectively). Finally, 4 patients underwent enrollment in a phase 2 clinical trial assessing IV ascorbic acid in TET2-mutated CCUS (ClinicalTrials.gov identifier: NCT03418038). Of 4 patients in the clinical trial, 1 patient did receive prior cytotoxic therapy and was in the t-CCUS group.
Phenotype upon progression and cause of death
Of 151 patients, 28 (18.5%) patients (16/105 with CCUS and 12/46 with t-CCUS) progressed to MNs. MDS was the most common diagnosis upon progression in both cohorts (7.6% CCUS vs 15% t-CCUS), followed by acute myeloid leukemia and chronic myelomonocytic leukemia (Table 1). Overall, 44 (29%) patients died during follow-up. The causes of death of were evenly distributed between those listed earlier, with 6 (23%) malignancy, 4 (15%) cardiac, 8 (30%) infection, 8 (30%) multifactorial in CCUS. In contrast, the cause of death in t-CCUS cohort was 8 (44%) malignancy, 4 (22%) cardiac, 2 (11%) infection, and 4 (22%) multifactorial (P = .299).
PFS and OS
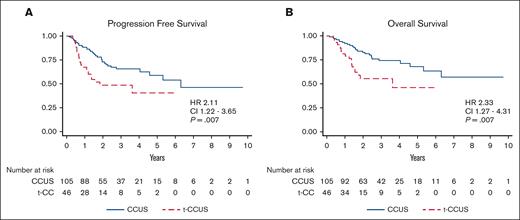
The median length of follow-up of the study cohort was 2.2 years, with the CCUS group having a significantly longer length of follow-up (2.5 vs 1.5 years, P = .002). CCUS had a significantly longer PFS than t-CCUS, (6.3 vs 1.8 years; hazard ratio [HR], 2.1; P = .007; Figure 2A). Similarly, median OS was longer for CCUS than t-CCUS (not reached vs 3.6 years; HR, 2.3; P = .007; Figure 2B).
Survival of clonal cytopenia of undetermined significance (CCUS) cases stratified by prior cytotoxic therapies. Prior cytotoxic therapies is associated with inferior (A) progression-free survival (PFS) and (B) overall survival (OS) in patients with CCUS.
Survival of clonal cytopenia of undetermined significance (CCUS) cases stratified by prior cytotoxic therapies. Prior cytotoxic therapies is associated with inferior (A) progression-free survival (PFS) and (B) overall survival (OS) in patients with CCUS.
Next, we performed univariable time-to-event analysis (UVA) for PFS and OS including the clinical and genetic variables available at CCUS diagnosis (Table 3). The most frequent aforementioned mutations were analyzed in addition to groupings by different mutational types such as role in cell signaling, spliceosome factors, or epigenetic regulators.14 The grouping of DNMT3A, ASXL1, and TET2 (DAT mutations) was also included, because 62% of cases harbored ≥1 of these 3 mutations. Exposure to prior therapy was associated with inferior PFS (HR, 2.11; 95% confidence interval [CI], 1.22-3.65; P = .007) and inferior OS (HR, 2.33; CI, 1.27-4.31; P = .007; Figure 2). Similarly, a higher CHRS score was associated with inferior PFS (HR, 1.37; CI, 1.18-1.59; P < .001) and OS (HR, 1.29; CI, 1.1-1.51; P = .002). In addition, lower Hb, lower platelet count, higher RDW, higher maximum VAF, higher CHRS score, TP53mut, and SF3B1mut were associated with inferior PFS. Increased age, lower Hb, lower platelets, higher RDW, the presence of abnormal cytogenetics, and TP53mut were associated with inferior OS. Age, sex, and smoking status were not significantly associated with PFS or OS.
Univariable analysis for PFS and OS in patients with CCUS
| Variables . | PFS . | OS . | ||
|---|---|---|---|---|
| HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| Exposure to therapy | 2.11 (1.22-3.63) | .007 | 2.33 (1.27-4.31) | .007 |
| Age | 1.02 (1.00-1.04) | .077 | 1.03 (1.01-1.10) | .046 |
| Sex (male) | 1.28 (0.73-2.24) | .395 | 1.07 (0.57-2.00) | .833 |
| Active or A | 1.10 (0.65-1.87) | .728 | 0.95 (0.52-1.74) | .876 |
| Hb | 0.78 (0.69-0.89) | <.001 | 0.74 (0.63-0.86) | <.001 |
| WBC | 1.00 (0.93-1.08) | .936 | 1.03 (0.96-1.11) | .420 |
| PLT | 0.99 (0.99-0.99) | .022 | 0.99 (0.99-0.99) | .023 |
| ANC | 1.02 0.92-1.12) | .754 | 1.06 (0.96-1.18) | .221 |
| AMC | 1.10 (0.73-1.65) | .667 | 1.25 (0.84-1.87) | .264 |
| RDW | 1.18 (1.10-1.26) | <.001 | 1.20 (1.11-1.29) | <.001 |
| MCV | 1.03 (1.00-1.06) | .022 | 1.03 (0.99-1.06) | .059 |
| Abnormal cytogenetics | 1.55 (0.88-2.73) | .132 | 1.93 (1.04-3.59) | .040 |
| Bone marrow blasts | 1.15 (0.91-1.45) | .236 | 1.24 (0.96-1.59) | .098 |
| Number of mutations | 1.15 (0.90-1.48) | .259 | 1.18 (0.90-1.56) | .237 |
| CHRS score | 1.37 (1.18-1.59) | <.001 | 1.29 (1.10-1.51) | .002 |
| Maximum VAF | 0.98 (0.97-0.99) | .043 | 0.99 (0.97-1.00) | .144 |
| TET2 mutated | 0.97 (0.56-1.68) | .923 | 0.87 (0.46-1.61) | .649 |
| SRSF2 mutated | 1.25 (0.68-2.28) | .478 | 1.23 (0.62-2.43) | .559 |
| ASXL1 mutated | 1.02 (0.48-2.15) | .966 | 1.17 (0.52-2.63) | .704 |
| DNMT3A mutated | 1.07 (0.48-2.37) | .868 | 1.16 (0.49-2.76) | .730 |
| ZRSR2 mutated | 1.19 (0.51-2.79) | .684 | 0.89 (0.32-2.49) | .820 |
| U2AF1 mutated | 2.06 (0.97-4.39) | .061 | 2.15 (0.95-4.83) | .065 |
| TP53 mutated | 2.66 (1.14-6.24) | .024 | 3.29 (1.28-8.43) | .013 |
| SF3B1 mutated | 3.86 (1.2-12.50) | .024 | 0.73 (0.10-5.31) | .755 |
| RUNX1 mutated | 1.46 (0.45-4.67) | .526 | 1.94 (0.60-6.30) | .268 |
| DAT∗ mutation | 0.88 (0.52-1.49) | .633 | 0.78 (0.43-1.41) | .412 |
| Spliceosome factor mutation | 1.68 (0.99-2.84) | .053 | 1.18 (0.65-2.15) | .585 |
| Epigenetic regulator mutation | 0.90 (0.53-1.53) | .702 | 0.85 (0.47-1.53) | .588 |
| Cell signaling mutation | 0.77 (0.33-1.80) | .550 | 0.83 (0.33-2.11) | .695 |
| Variables . | PFS . | OS . | ||
|---|---|---|---|---|
| HR (95% CI) . | P value . | HR (95% CI) . | P value . | |
| Exposure to therapy | 2.11 (1.22-3.63) | .007 | 2.33 (1.27-4.31) | .007 |
| Age | 1.02 (1.00-1.04) | .077 | 1.03 (1.01-1.10) | .046 |
| Sex (male) | 1.28 (0.73-2.24) | .395 | 1.07 (0.57-2.00) | .833 |
| Active or A | 1.10 (0.65-1.87) | .728 | 0.95 (0.52-1.74) | .876 |
| Hb | 0.78 (0.69-0.89) | <.001 | 0.74 (0.63-0.86) | <.001 |
| WBC | 1.00 (0.93-1.08) | .936 | 1.03 (0.96-1.11) | .420 |
| PLT | 0.99 (0.99-0.99) | .022 | 0.99 (0.99-0.99) | .023 |
| ANC | 1.02 0.92-1.12) | .754 | 1.06 (0.96-1.18) | .221 |
| AMC | 1.10 (0.73-1.65) | .667 | 1.25 (0.84-1.87) | .264 |
| RDW | 1.18 (1.10-1.26) | <.001 | 1.20 (1.11-1.29) | <.001 |
| MCV | 1.03 (1.00-1.06) | .022 | 1.03 (0.99-1.06) | .059 |
| Abnormal cytogenetics | 1.55 (0.88-2.73) | .132 | 1.93 (1.04-3.59) | .040 |
| Bone marrow blasts | 1.15 (0.91-1.45) | .236 | 1.24 (0.96-1.59) | .098 |
| Number of mutations | 1.15 (0.90-1.48) | .259 | 1.18 (0.90-1.56) | .237 |
| CHRS score | 1.37 (1.18-1.59) | <.001 | 1.29 (1.10-1.51) | .002 |
| Maximum VAF | 0.98 (0.97-0.99) | .043 | 0.99 (0.97-1.00) | .144 |
| TET2 mutated | 0.97 (0.56-1.68) | .923 | 0.87 (0.46-1.61) | .649 |
| SRSF2 mutated | 1.25 (0.68-2.28) | .478 | 1.23 (0.62-2.43) | .559 |
| ASXL1 mutated | 1.02 (0.48-2.15) | .966 | 1.17 (0.52-2.63) | .704 |
| DNMT3A mutated | 1.07 (0.48-2.37) | .868 | 1.16 (0.49-2.76) | .730 |
| ZRSR2 mutated | 1.19 (0.51-2.79) | .684 | 0.89 (0.32-2.49) | .820 |
| U2AF1 mutated | 2.06 (0.97-4.39) | .061 | 2.15 (0.95-4.83) | .065 |
| TP53 mutated | 2.66 (1.14-6.24) | .024 | 3.29 (1.28-8.43) | .013 |
| SF3B1 mutated | 3.86 (1.2-12.50) | .024 | 0.73 (0.10-5.31) | .755 |
| RUNX1 mutated | 1.46 (0.45-4.67) | .526 | 1.94 (0.60-6.30) | .268 |
| DAT∗ mutation | 0.88 (0.52-1.49) | .633 | 0.78 (0.43-1.41) | .412 |
| Spliceosome factor mutation | 1.68 (0.99-2.84) | .053 | 1.18 (0.65-2.15) | .585 |
| Epigenetic regulator mutation | 0.90 (0.53-1.53) | .702 | 0.85 (0.47-1.53) | .588 |
| Cell signaling mutation | 0.77 (0.33-1.80) | .550 | 0.83 (0.33-2.11) | .695 |
Variables in bold indicate P < 0.05. MCV, mean corpuscular volume; PLT, platelets; WBC, white blood cell count.
DAT mutations: DNMT3A, ASXL1, and TET2.
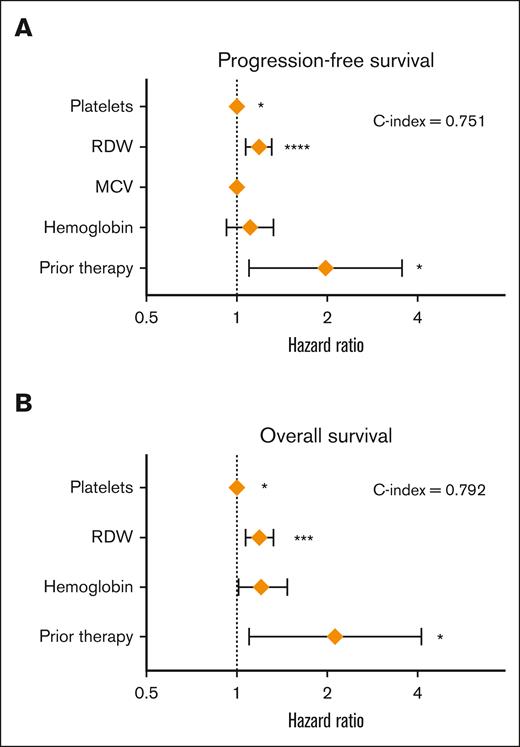
Given the limited number of events, multivariable time-to-event was limited to the inclusion of 3 to 5 factors significant in the univariable analysis (Figure 3). The clinical model consisted only of complete blood count parameters available at diagnosis and a baseline variable of exposure to therapy (Table 4). Exposure to prior therapy was independently associated with an inferior PFS (HR, 2.01; CI, 1.12-3.61; P = .020) and OS (HR, 2.13; CI, 1.10-4.14; P = .026). Next, we investigated whether the inclusion of prior therapy further risk stratifies CHRS and noted that prior therapy remained significant for PFS and OS even in the context of CHRS. Finally, we included statistically significant genetic factors identified on univariable time-to-event analysis to observe that the prior exposure therapy was independently associated with PFS and OS. Interestingly, “clinical” models had the highest C-index statistics for PFS and OS (0.751 and 0.792, respectively) among all the models tested.
Factors associated with survival in clonal cytopenia of undetrmined significane (CCUS). Forest plot of multivariable analysis for (A) progression-free survival (PFS) and (B) overall survival (OS) in CCUS.
Factors associated with survival in clonal cytopenia of undetrmined significane (CCUS). Forest plot of multivariable analysis for (A) progression-free survival (PFS) and (B) overall survival (OS) in CCUS.
Multivariable analysis for PFS and OS in patients with CCUS
| Model . | Variable . | HR (95% CI) . | P value . | C index . |
|---|---|---|---|---|
| PFS | ||||
| Clinical | Exposure to therapy | 1.98 (1.10-3.56) | .023 | |
| Lower Hb | 1.11 (0.93-1.33) | .250 | ||
| Lower PLT | 1.01 (1.01-1.01) | .014 | ||
| Higher RDW | 1.19 (1.07-1.31) | <.001 | 0.751 | |
| Higher MCV | 1.01 (0.98-1.05) | .463 | ||
| Modified CHRS | Exposure to therapy | 2.34 (1.35-4.05) | .002 | |
| Higher CHRS Score | 1.38 (1.19-1.60) | <.001 | 0.688 | |
| Genetics only | Exposure to therapy | 2.08 (1.17 3.70) | .013 | 0.632 |
| TP53 mutated | 2.03 (0.84-4.95) | .118 | ||
| SF3B1 mutated | 5.31 (1.60-17.77) | .006 | ||
| OS | ||||
| Clinical | Exposure to therapy | 2.13 (1.10-4.14) | .026 | 0.792 |
| Lower Hb | 1.21 (1.01-1.47) | .042 | ||
| Lower PLT | 1.01 (1.01-1.01) | .015 | ||
| Higher RDW | 1.19 (1.07-1.32) | .001 | ||
| Modified CHRS | Exposure to therapy | 2.28 (1.20-4.31) | .012 | 0.662 |
| Abnormal cyto. | 2.20 (1.17-4.13) | .014 | ||
| Higher CHRS Score | 1.29 (1.10-1.53) | .002 | ||
| Genetics only | Exposure to therapy | 2.02 (1.05-3.85) | .034 | 0.607 |
| TP53 mutated | 2.03 (0.59-6.99) | .264 | ||
| Abnormal cyto. | 2.07 (1.09-3.93) | .006 | ||
| Model . | Variable . | HR (95% CI) . | P value . | C index . |
|---|---|---|---|---|
| PFS | ||||
| Clinical | Exposure to therapy | 1.98 (1.10-3.56) | .023 | |
| Lower Hb | 1.11 (0.93-1.33) | .250 | ||
| Lower PLT | 1.01 (1.01-1.01) | .014 | ||
| Higher RDW | 1.19 (1.07-1.31) | <.001 | 0.751 | |
| Higher MCV | 1.01 (0.98-1.05) | .463 | ||
| Modified CHRS | Exposure to therapy | 2.34 (1.35-4.05) | .002 | |
| Higher CHRS Score | 1.38 (1.19-1.60) | <.001 | 0.688 | |
| Genetics only | Exposure to therapy | 2.08 (1.17 3.70) | .013 | 0.632 |
| TP53 mutated | 2.03 (0.84-4.95) | .118 | ||
| SF3B1 mutated | 5.31 (1.60-17.77) | .006 | ||
| OS | ||||
| Clinical | Exposure to therapy | 2.13 (1.10-4.14) | .026 | 0.792 |
| Lower Hb | 1.21 (1.01-1.47) | .042 | ||
| Lower PLT | 1.01 (1.01-1.01) | .015 | ||
| Higher RDW | 1.19 (1.07-1.32) | .001 | ||
| Modified CHRS | Exposure to therapy | 2.28 (1.20-4.31) | .012 | 0.662 |
| Abnormal cyto. | 2.20 (1.17-4.13) | .014 | ||
| Higher CHRS Score | 1.29 (1.10-1.53) | .002 | ||
| Genetics only | Exposure to therapy | 2.02 (1.05-3.85) | .034 | 0.607 |
| TP53 mutated | 2.03 (0.59-6.99) | .264 | ||
| Abnormal cyto. | 2.07 (1.09-3.93) | .006 | ||
Variables in bold indicate P < 0.05. CHRS, clonal hematopoiesis risk score; cyto., cytogenetics; MCV, mean corpuscular volume.
Analysis of previous therapy
In t-CCUS, the median time from the first cytotoxic therapy to diagnosis was 3.2 years (range, 1 month-38 years). The median time from exposure to topoisomerase inhibitor to diagnosis of CCUS was significantly shorter than for other types of therapy (1.8 years vs 5.1 years, P = .006). Nine patients had exposure to topoisomerase inhibitor (8 with lymphoma and 1 with ovarian cancer). Of 9 patients exposed to topoisomerase, 3 (33%) patients harbored TP53mut CCUS. Exposure to topoisomerase inhibitors but not other classes of therapies was associated with inferior PFS (HR, 3.37; CI, 1.34-8.49; P = .010) and OS (HR, 2.89; CI, 1.07-7.83; P = .037; supplemental Table 8). Finally, when stratified by therapy as topoisomerase vs nontopoisomerase class, topoisomerase II therapies were associated with shorter PFS and OS (supplemental Table 9).
Findings specific to patients who experienced progression or death
Of 151 patients, 56 (37%) experienced either progression to MN or death (event). This was compared with 95 patients who remained event-free, agnostic of the CCUS or t-CCUS groups (supplemental Table 10). There were no differences in the sex distribution, smoking status, or median age between the “event” and the “event-free” groups. Those experiencing an event presented with a significantly lower Hb (9.9 vs 11.5, P = .001), higher mean corpuscular volume (102.2 vs 98, P = .042), lower platelet count (92 vs 129, P = .001), and higher RDW (14.3 vs 17.1, P = .001) at diagnosis. No differences were found in bone marrow cellularity, median bone marrow blasts, proportion of cases with abnormal cytogenetics, or number of mutations on NGS. Both the patients who experienced an event and the event-free cohort were similarly treated, primarily with surveillance (75% vs 74%). Interestingly, there was a trend toward a higher VAF in the event-free cohort than in the event cohort (38% vs 43, P = .088).
Discussion
Our aim was to evaluate whether the risk of progression and/or death in CCUS are different based on prior exposure to DNA-damaging therapies. Comparing a cohort of consecutive patients with CCUS with the largest published cohort of t-CCUS, we show that prior DNA-damaging therapies was indeed an independent risk-factor for inferior PFS and OS.
Clinical and demographic characteristics were generally comparable between the 2 cohorts, except patients with t-CCUS were more likely to present with hypocellular bone marrow aspirates/biopsy along with more bone marrow blasts while remaining <5%. Intuitively, one would ascribe hypocellular marrow states with higher degrees of cytopenias in t-CCUS compared with CCUS, however, blood parameters were comparable between the 2 cohorts.
Previously, we showed that within 2 years of diagnosis, 17% patients with CCUS experienced progression to MNs, with transfusion requirement and survival being comparable with that of lower-risk MDS.15 In this study, we curated our database, classifying patients with CCUS who had received prior cytotoxic therapy as t-CCUS and found that this subgroup of patients had significantly worse PFS and OS in comparison with those with de novo CCUS. In contrast, patients with t-CCUS had better survival outcomes in comparison with those with t-MN, regardless of the t-MN phenotype at diagnosis.11 Despite the relative similarity in presentation and management, previous exposure to DNA-damaging agents for patients with CCUS was independently associated with inferior PFS and OS, suggesting that prior cytotoxic therapies assign a distinct subentity within the larger CCUS umbrella.
We created clinically useful risk-prediction models using data available at the outset and after comprehensive evaluation is completed. In all the models, inclusion of prior cytotoxic therapy as a variable remained independently associated with PFS and OS, highlighting the value of clinical history taking.
Although the underlying rationale remains unclear, the mechanisms of the differential survival rates are likely multifactorial. First, patients with t-CCUS were enriched with TP53mut compared with de novo CCUS, which are traditionally considered to be higher risk for leukemic progression and survival. Moreover, we recently showed that TP53mut and TP53wt CCUS had comparable PFS and OS.16 Second, having encountered that an additional malignancy and receiving therapy may lead to more pronounced cytopenia and inability or preference for not receiving CCUS-directed therapy. However, such patterns did not emerge as the degree of cytopenia was comparable between the 2 cohorts. Moreover, within the scope of retrospective analysis, management strategies were not different between CCUS and t-CCUS. Finally, progression of the primary disease may have led to increased mortality in t-CCUS compared with CCUS (39.1% vs 24.8%), although the difference was not statistically significant. A larger, preferably prospective study may help answer these questions.
Our retrospective analysis has notable limitations. First, although ours is the largest study comparing outcomes of CCUS with t-CCUS, relatively small numbers limited the number of variables that could be analyzed simultaneously. This is in contrast with CH in which the association and impact of cytotoxic therapies on the genetic landscape and clonal evolution is well characterized.3,4,17 CCUS diagnosis was made clinically as opposed to uniform sequencing of a large cohort at a uniform timing in larger CH studies.4,5,18 Finally, the risk of leukemic transformation is much higher and the latency much shorter after CCUS than CH. Second, highlighted earlier, the management after CCUS diagnosis was at providers’ discretion and may have been biased by factors beyond our consideration. In addition, the lack of consensus in MDS-defining cytogenetics and diagnostic criteria may have also affected management decisions. With regard to the newer classifications by ICC, it is likely that 8 patients from of our cohort may represent the entity now recognized as clonal cytopenia and monocytosis of undetermined significance. This may inherently bias the cohort toward higher risk of progression to MDS/myeloproliferative neoplasm, especially to chronic myelomonocytic leukemia.12 Finally, progressive versions of the NGS panels used over the accrual period included additional genes and higher sensitivity, which may contribute to underrepresentation of some of the emerging variants. A notable example is PPM1D, 1 of the most common therapy-emergent variants, was not included in our earlier clinical NGS panel and is likely underrepresented in the study.
Acknowledging these limitations, we concluded that t-CCUS has distinct morphological and genetic features compared with CCUS and had a higher risk of progression to MN and death. Given the overall higher risk of progression and death, more stringent monitoring may be considered for patients with t-CCUS. Whether earlier intervention should be considered for patients with t-CCUS who do not technically have a hematologic malignancy remains to be determined and is a topic for further investigation.
Acknowledgments
The authors are grateful to the patients and their families.
M.V.S. was supported by Bridget Kiely Clinician Career Development in Transplant Research and a K2R Pipeline Award at Mayo Clinic, Rochester, MN.
Authorship
Contribution: M.L., A.B., S.N.S., M.G., and M.V.S. collected data; M.L. and M.V.S. performed statistical analysis and wrote the first draft of the manuscript; A.A.-K., H.A., J.F., C.A.Y., A.M., K.M., A.T., M.M.P., and M.V.S. recruited patients and edited the manuscript; D.C. performed independent rereview of pathology and edited the manuscript; M.M.P. and M.V.S. conceived the study; and all authors approved the final draft of the manuscript.
Conflict-of-interest disclosure: A.A.-K. reports research funding from Novartis and received research support to institution from Astex. M.M.P. reports membership on an entity's board of directors or advisory committees of Stemline Therapeutics, and reports research funding from Kura Oncology. M.V.S. reports research funding to institution from Astellas, AbbVie, Celgene, and Marker Therapeutics. The remaining authors declare no competing financial interests.
Correspondence: Mrinal Patnaik, Department of Hematology, Mayo Clinic, 200 1st St SW, Rochester, MN 55905; email: patnaik.mrinal@mayo.edu; and Mithun Vinod Shah, Department of Hematology, Mayo Clinic, 200 1st St SW, Rochester, MN 55905; email: shah.mithun@mayo.edu.
References
Author notes
Presented, in part, at the 2023 Hybrid Congress of the European Hematology Association, Frankfurt, Germany, 8 June 2023.
Data are available on request from authors, Mrinal Patnaik (patnaik.mrinal@mayo.edu) and Marissa Li (li.marissa@mayo.edu).
The full-text version of this article contains a data supplement.