The Comparative Study of Children in Haiti and Miami with SCD network supports the creation of 4 sickle cell centers in Haiti, including NBS and pediatric follow-up programs.
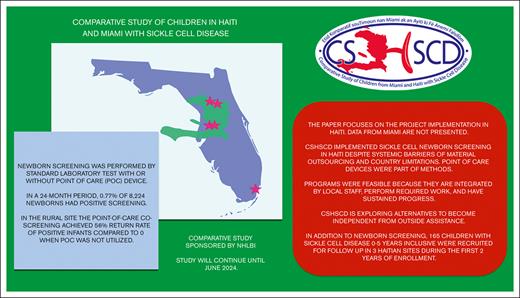
Despite barriers, 61 of 8224 (0.77%) newborns screened tested positive for SCD, and 165 children were enrolled for follow-up over 24 months.
Visual Abstract
One in 120 children are born with sickle cell disease (SCD) in Haiti. However, health care challenges include isolated newborn screening (NBS) activities and lack of transcranial Doppler (TCD) ultrasound to assess stroke risk. The implementation activities of the Comparative Study of Children in Haiti and Miami with Sickle Cell Disease involved both NBS and TCD ultrasound implementations at 4 Haitian clinical sites. We hypothesized that hospital-based newborn SCD screening and follow-up programs would be feasible at Haiti. A traditional NBS laboratory method with dried blood samples was performed at 3 Port-au-Prince sites, and the traditional method plus point-of-care (POC) testing was used at the 2 northern sites. The rate of clinical follow-up for newborns with SCD as the outcome for the NBS intervention was compared with that of the NBS method. The NBS programs identified SCD in 0.77% of 8224 newborns over a 24-month period. In the rural hospital assigned to the combination screening, 56% of newborns identified with POC testing returned for follow-up, compared with 0% when POC was not available (P = .044). Newborns who tested positive for SCD and children aged <6 years with SCD at the clinical sites were eligible for study follow-up. Accrual was successful: 165 participants (mean age, 42 months; 53% males; 93% hemoglobin SS) were recruited and received oral penicillin. TCD ultrasound screening was hampered by poor internet connections and trained staff leaving Haiti, with only 1 active site conducting screening. Despite challenges, the implementation of NBS and sickle cell programs in Haiti is feasible. We are in the process of understanding how to mitigate implementation limitations.
Introduction
Haiti is a low-income country in the Americas. In 2020, the gross domestic product per capita was $1309,1 of which only $13 per capita were allocated to health care.2 Because of poverty and political uncertainty, the United States has received ∼700 000 Haitian immigrants, with the largest diaspora concentrated in the greater Miami area (Miami-Dade, Broward, and Palm Beach Counties), FL, and in Kings County, NY.3
Lack of universal newborn screening (NBS) in Haiti is a barrier to knowing the true disease birth prevalence and connecting children with sickle cell disease (SCD) to care. Other health care limitations include the unavailability of oral penicillin, hydroxyurea, and transcranial Doppler (TCD) ultrasound screening. Recognizing that ∼30% of patients with SCD examined at the University of Miami pediatric and medical sickle cell programs are of Haitian descent, the prevalence of SCD in Haiti is likely high. Data from limited population screening in 1995 showed that 1.1% newborns in the Haitian capital, Port-au-Prince, had SCD.4 In 2010, 2459 newborns were screened at the only children’s hospital in Haiti, St Damien Hospital (SDH), in Tabarre, Port-au-Prince, with dried blood cards processed at Milan, Italy, and with 0.57% of those children being diagnosed with SCD (FS and FSC combined).5 From August 2017 to August 2018, we screened 2159 newborns at Justinien University Hospital (JUH) in Cap Haitien, the second largest city in Haiti, confirming positive results for 1 out of 120 (0.83%), without the need to outsource any specimens.6 In the forementioned pilot study, we validated the lateral flow immunoassay point-of-care (POC) Sickle SCAN (BioMedomics, Morrisville, NC) for NBS against high performance liquid chromatography (HPLC). Our rationale for adding a POC to traditional laboratory testing was that families would know their babies’ status immediately and increase the chances of clinic follow-up, given that finding families in inner cities and rural areas is difficult because of imprecise addresses. In 2019, SDH commenced NBS with isoelectric focusing (IEF) performed on site, receiving samples from Hospital La Paix and Hospital of the University of State of Haiti (HUEH).
The observational study Comparative Study of Children in Haiti and Miami with Sickle Cell Disease (CSHSCD; National Institutes of Health, R01HL149121) aims to (1) implement the expansion of NBS in a coordinated fashion to Haitian sites, testing the intervention of the POC addition to laboratory methods, (2) provide support for clinical follow-up with medications and consultation, (3) implement stroke screening among the sickle cell pediatric population from the 4 participating hospitals in Haiti, and (4) compare clinical outcomes and barriers in a cohort of children from Miami. This article does not present the comparison with the Miami cohort but instead concentrates on the implementation experience in Haiti in the first 2 years of the study. As the primary implementation outcome, we hypothesize that the hospital-based sickle cell program implementation is feasible. To reflect on program feasibility, we discuss expected systemic and patient barriers, such as lack of laboratory materials and an adverse social environment (eg, poverty and lack of knowledge about SCD).
Methods
This report was written following the recommendations of the Standards for Reporting Implementation Studies, when applicable.7
The CSHSCD definition of a feasible hospital-based program
The implementation outcome was feasibility. Feasibility is defined by Karsh as the “extent to which a new treatment, or an innovation, can be successfully used or carried out within a given agency or setting.”8 A hospital-based program is feasible if (1) it is formed with local talent; (2) the team works independently, according to protocol (ie, fidelity); and (3) results are obtained from either NBS or patient follow-up. Indicators or measures of feasible data include NBS completion rates following preestablished protocol and participant accrual and retention.9
Ethics
The CSHSCD was approved by the University of Miami institutional review board and by the National Bioethics Committee in Haiti. The Haitian Ministry of Health was also notified of the study. The CSHSCD was not registered at www.clinicaltrials.gov because it is not a clinical trial.
Context of the study in Haiti
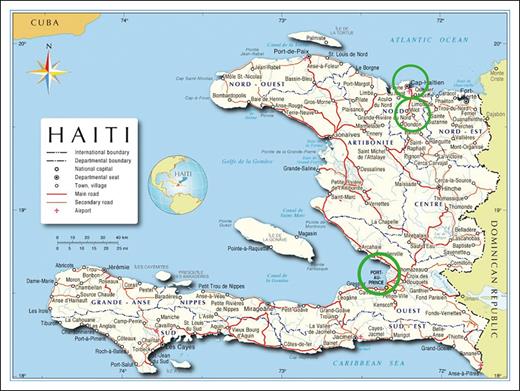
Figure 1 presents the geographic location of the hospitals, their resources, and the clinic populations.
Haiti and the study environment. Port-au-Prince is Haiti’s capital, with 2.9 million inhabitants (2022 data)10 with a poverty level of 87.6% and 30.3% of the population living under the extreme poverty line.11 Social factors include street violence and suboptimal literacy rate for Haiti, in general, with 61.7% literacy for individuals aged ≥15 years and 83% literacy for the subgroup of individuals aged 15 to 24 years.12 SDH, the only pediatric hospital in Haiti, has a 224-bed capacity. The hospital serves 1000 children with SCD, from infancy to age 12 years. The SDH laboratory has a Sebia capillary hemoglobin electrophoresis machine and a Perkin Elmer IEF machine in use for NBS. HUEH does not have capacity for hemoglobinopathy evaluation and outsources samples to SDH. Thirty children with SCD are examined at HUEH. JUH is located at Cap Haitien, Haiti’s second largest city after Port-au-Prince, with 190 000 inhabitants.10 This program has a nurse coordinator and CHW, serving 30 children with SCD. Located at Milot, a rural town of 25 100 inhabitants,13 11 miles south from Cap Haitien, SCH does not have hemoglobinopathy laboratory equipment; >200 children are followed up at SCH. None of the 4 hospitals had a TCD ultrasound machine before the CSHSCD started. Hydroxyurea was purchased from outside vendors (rarely) or supplied by sponsors. Before the CSHSCD, penicillin was administered by intramuscular injection every month as pneumococcal prophylaxis for children aged <5 years.
Haiti and the study environment. Port-au-Prince is Haiti’s capital, with 2.9 million inhabitants (2022 data)10 with a poverty level of 87.6% and 30.3% of the population living under the extreme poverty line.11 Social factors include street violence and suboptimal literacy rate for Haiti, in general, with 61.7% literacy for individuals aged ≥15 years and 83% literacy for the subgroup of individuals aged 15 to 24 years.12 SDH, the only pediatric hospital in Haiti, has a 224-bed capacity. The hospital serves 1000 children with SCD, from infancy to age 12 years. The SDH laboratory has a Sebia capillary hemoglobin electrophoresis machine and a Perkin Elmer IEF machine in use for NBS. HUEH does not have capacity for hemoglobinopathy evaluation and outsources samples to SDH. Thirty children with SCD are examined at HUEH. JUH is located at Cap Haitien, Haiti’s second largest city after Port-au-Prince, with 190 000 inhabitants.10 This program has a nurse coordinator and CHW, serving 30 children with SCD. Located at Milot, a rural town of 25 100 inhabitants,13 11 miles south from Cap Haitien, SCH does not have hemoglobinopathy laboratory equipment; >200 children are followed up at SCH. None of the 4 hospitals had a TCD ultrasound machine before the CSHSCD started. Hydroxyurea was purchased from outside vendors (rarely) or supplied by sponsors. Before the CSHSCD, penicillin was administered by intramuscular injection every month as pneumococcal prophylaxis for children aged <5 years.
Study team composition
The study team has knowledge in hematology, pediatrics, implementation science, and Haitian culture. O.A.A. is the only pediatric hematologist on the study. The investigators at the 4 Haitian sites are pediatricians with interest in SCD. Our study team includes a Haitian anthropologist (L.H.M.), an implementation scientist (E.C.B.), and a biostatistician (S.M.). Communication was in English because the Haitian principal investigators were bilingual (English and Haitian Creole), using Spanish as a backup language for some of the investigators (N.S.V.D. and E.M.). Furthermore, University of Miami team members (T.H., M.C., and L.H.M.) spoke Haitian Creole, facilitating full communication among all network members.
Study design of the NBS program comparing traditional laboratory method with or without POC
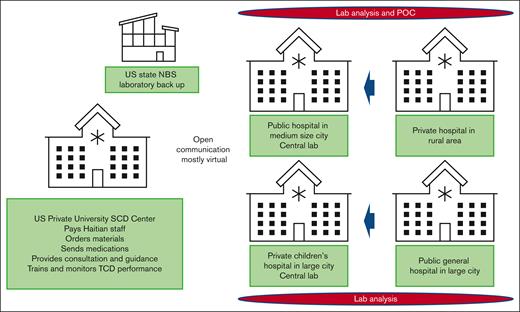
Figure 2 presents the CSHSCD hospital network. The 4 hospitals were assigned according to region to analyze dried blood samples with traditional laboratory IEF (Port-au-Prince sites SDH and HUEH) or to perform the 2-test NBS approach of IEF plus lateral flow immunoassay POC (northern sites, the Sacré Cœur Hospital [SCH] and JUH). NBS nurses were instructed to receive verbal consent from mothers to obtain blood samples from their babies by heel stick. At sites using the 2-test approach, the nurses explained the POC test and notified the results immediately to mothers. SDH processed their samples and those from the HUEH site. JUH was tasked with testing their own samples and samples from SCH. Newborns who had positive screening results for sickle cell hemoglobinopathies (FS, FSC, FSA, and FS-other) or other hemoglobinopathy (not trait) returned to the hospitals for confirmatory testing. Babies with hemoglobin C disease or β-thalassemia were excluded from follow-up in the CSHSCD although they could be followed up as part of standard of care. The traditional laboratory method with POC was compared with the laboratory method without POC to assess the likelihood of follow-up as a health outcome in infants after a positive screen result for SCD.
CSHSCD network. This figure shows the descriptions, location, NBS methods (laboratory analysis alone or with POC), and expected laboratory flow of the different entities. The University of Miami is the US private University SCD Center. The US state NBS laboratory is in Florida. SCH is the private hospital in rural Haiti, which is supposed to send NBS samples to JUH, the public hospital in medium-sized Haitian city, Cap Haitien. The public general hospital in Port-au-Prince (large city) is HUEH that sends NBS samples to the SDH, the private children’s hospital also in Port-au-Prince.
CSHSCD network. This figure shows the descriptions, location, NBS methods (laboratory analysis alone or with POC), and expected laboratory flow of the different entities. The University of Miami is the US private University SCD Center. The US state NBS laboratory is in Florida. SCH is the private hospital in rural Haiti, which is supposed to send NBS samples to JUH, the public hospital in medium-sized Haitian city, Cap Haitien. The public general hospital in Port-au-Prince (large city) is HUEH that sends NBS samples to the SDH, the private children’s hospital also in Port-au-Prince.
NBS personnel
All Haitian sites employed 1 or 2 NBS nurses per site and 1 to 3 community health workers (CHWs) per site. SDH and JUH each employed a laboratory technician dedicated to hemoglobinopathy testing. All staff were trained in person and virtually and paid in total or partially by CSHSCD.
Role of CHWs
CHWs were encouraged to be present when the nurses approached mothers for screening. Because CHWs are traditionally viewed as trustworthy, their presence provided an extra layer of trust for mothers, especially if they had never heard of the NBS activity. CHWs participated in explaining the importance of the testing. In the case of a positive result, CHWs supported families, answered their questions, and assisted with penicillin delivery. CHWs could make house calls if a child had not visited the clinic.
Test methodologies
Laboratory methods
SDH purchased an IEF machine (Perkin Elmer, Waltham, MA) and capillary electrophoresis equipment (Sebia Inc, Norcross, GA) with Pierre Fabre funds. JUH received a Migele IEF machine (Perkin Elmer) through the CSHSCD. The laboratory technician from SDH, who was trained before the CSHSCD, trained the laboratory technician from JUH. The Florida NBS State Laboratory was the backup laboratory to run tests if materials were temporarily unavailable in Haiti or for quality assurance testing, using HPLC to test dried blood samples.
POC methods
The study coordinators, nurses, laboratory technicians, and center investigators were trained on the performance of the POCs. Sickle SCAN training was conducted in person. Gazelle training was conducted virtually for each site headed by the company staff. POCs are shown in Figure 3.
POC devices used to identify or quantify hemoglobins. Sickle SCAN (BioMedomics) detects hemoglobins A, S, and C, and Gazelle Hemoglobin Variant (Hemex Health) detects and quantifies different hemoglobins present in the samples.
POC devices used to identify or quantify hemoglobins. Sickle SCAN (BioMedomics) detects hemoglobins A, S, and C, and Gazelle Hemoglobin Variant (Hemex Health) detects and quantifies different hemoglobins present in the samples.
Sickle SCAN
Sickle SCAN (BioMedomics) is a POC method that uses lateral flow immunoassay technology, providing results in 5 minutes.14 After obtaining a few blood drops (5 μL), the sample is hemolyzed in a buffer solution. Five drops are placed in the device well and allowed to migrate. Antibody bands for hemoglobins A, S, and C capture the hemoglobins that are present in the blood, with levels of detection of 1%, 2.4%, and 3.4%, respectively.15,16 A control is present in the device to determine whether it is functional. We previously validated Sickle SCAN for NBS in Haiti, obtaining 97% specificity and 90% sensitivity.6 The northern sites used this POC along with the dried blood samples for the CSHSCD NBS implementation.
Gazelle Hemoglobin Variant
Gazelle Hemoglobin Variant (Hemex Health, Portland, OR) is a miniaturized HemeChip cellulose acetate paper–based microchip -hemoglobin electrophoresis that provides results in 8 minutes.17 Gazelle Hemoglobin Variant has 99% sensitivity and 99% specificity to detect SCD and thalassemia,18 and has recently been validated for newborns.19 The machine stores the information, and results can be printed. We used this POC method for baseline hemoglobinopathy evaluation for confirmatory testing and for the follow-up of children when verifying SCD genotype or when monitoring for hydroxyurea effect. The CSHSCD provided Gazelle Hemoglobin Variant HemeChip to all Haitian sites.
Follow-up of children with SCD
Consent was obtained in writing from parents in Haiti by the center investigator or site study coordinator using the approved Haitian Creole informed consent version. There was no alteration in the clinical care while on the study, except (1) TCD ultrasound screening for children with sickle cell anemia was not available unless they were on the study and (2) oral penicillin and hydroxyurea availability was increased while participating. If oral penicillin was not available from other sources, children would receive monthly intramuscular penicillin injections, which is Haiti’s standard practice for pneumococcal prophylaxis. Immunizations were administered according to the childhood schedule and were not interrupted, even if the child did not participate in the CSHSCD. Nevertheless, the study provided oral penicillin and paid for pneumococcal conjugate and polysaccharide immunizations if necessary. Children with hemoglobin SS and Sß0 thalassemia were eligible to receive hydroxyurea according to clinical parameters of severity (eg, frequent pain crisis and severe anemia) or were offered hydroxyurea at an age of ∼1 year. Growth parameters, including height, weight, and midupper arm circumference, were monitored to assess nutritional status.20 At entry a hemoglobinopathy evaluation was required, which was provided by either a laboratory method or by Gazelle Hemoglobin Variant POC.
TCD ultrasound performance
Children with hemoglobin SS and Sß0 thalassemia, aged ≥2 years were eligible to be screened with TCD ultrasound as part of standard of care. Before the CSHSCD, there were no TCD ultrasound machines in Haiti. Eight staff traveled from Haiti to Miami for a 4-day training course, during which 2 TCD ultrasound sonographers instructed them on the theorical and practical aspects of TCD ultrasound testing. We paid attention to training fidelity because the training was provided in 3 different sessions to different staff members. Providers at each site were trained and received 1 DWL Doppler Box X1 portable machine (Compumedics DWL, Singen, Germany) to take back to Haiti. After a staff member returned to work in Haiti, the plan was to use the monitoring system, Zeriscope, which is a patented web-based mobile technology designed to observe via camera how the sonographer is performing; the monitor view included how the probe was positioned and the Doppler results.
Follow-up enrollment ended on 31 March 2023, with participants being followed up until March 2024.
Implementation strategies and interventions
These methods are summarized in Table 1, in line with the Exploration, Preparation, Implementation, and Sustainment implementation framework.21
CHSCD implementation strategies and interventions tested
| Phase . | Strategy . | Intervention tested . | Time frame . |
|---|---|---|---|
| Preparation | Interviews with mothers regarding best way of conducting NBS; learning collaborative; protocol with manual of operations, French version; and informed consents, Haitian Creole | Working together as the CSHSCD network | 2016 2019 to present 2019 to 2020 2019 to present |
| Implementation | Reciprocal feedback between Miami and Haiti involving center investigators and study coordinators; backup for NBS laboratory; use of POC devices; encouraging active participation of study coordinators in discussions; availability of people who speak Haitian Creole in discussion; training and virtual monitoring of TCD ultrasound performance; data comparison; and barrier assessment | Diagnostic, NBS Diagnostic, Sickle SCAN Diagnostic, TCD ultrasound screening REDCap database Parent surveys Staff focus groups | Monthly ongoing 2020 to present 2020 to present 2020 to present 2018 to present 2021 to present 2019 to present 2019 to present 2022 |
| Sustainment | Discussion about how to achieve independence; meeting with Minister of Health designee; updates to Minister of Health seeking policy change for NBS and medication availability; and meetings with 2 pharmacy companies to bring oral penicillin and hydroxyurea to Haiti | Public health, policy Therapeutic, assuring a continuous supply of oral penicillin and hydroxyurea | 2022 to present 2018 2018, 2019, 2021, and 2022 2022 and 2023 |
| Phase . | Strategy . | Intervention tested . | Time frame . |
|---|---|---|---|
| Preparation | Interviews with mothers regarding best way of conducting NBS; learning collaborative; protocol with manual of operations, French version; and informed consents, Haitian Creole | Working together as the CSHSCD network | 2016 2019 to present 2019 to 2020 2019 to present |
| Implementation | Reciprocal feedback between Miami and Haiti involving center investigators and study coordinators; backup for NBS laboratory; use of POC devices; encouraging active participation of study coordinators in discussions; availability of people who speak Haitian Creole in discussion; training and virtual monitoring of TCD ultrasound performance; data comparison; and barrier assessment | Diagnostic, NBS Diagnostic, Sickle SCAN Diagnostic, TCD ultrasound screening REDCap database Parent surveys Staff focus groups | Monthly ongoing 2020 to present 2020 to present 2020 to present 2018 to present 2021 to present 2019 to present 2019 to present 2022 |
| Sustainment | Discussion about how to achieve independence; meeting with Minister of Health designee; updates to Minister of Health seeking policy change for NBS and medication availability; and meetings with 2 pharmacy companies to bring oral penicillin and hydroxyurea to Haiti | Public health, policy Therapeutic, assuring a continuous supply of oral penicillin and hydroxyurea | 2022 to present 2018 2018, 2019, 2021, and 2022 2022 and 2023 |
Data collection and analysis
The University of Miami developed a REDCap database, and each institution designee(s) entered data into various forms. All newborns and children on the study were assigned a code with the initials of each site and numbers according to screening sequence or active follow-up enrollment. The biostatistician (S.M.) from the University of Miami, Department of Public Health Sciences, conducted the data analysis for this report. All authors had access to the data.
The NBS form included data on age, sex, screening dates, type of test(s) used (POC, IEF, and/or HPLC), and screening results for each test (FA, FSC, FSA, FS, FAC, FC, FS-other, or other). The final medical diagnosis was based on a combination of the POC, IEF, and/or HPLC test results, depending on the type of test that was available. The counts and frequencies of the combinations of testing types were computed using statistical analysis software. The incidence of SCD (sickle cell anemia or hemoglobin SS, hemoglobin SC, and sickle β+-thalassemia) was calculated from the screening frequencies as per the site location. Clinic follow-up for newborns with positive results was also captured and compared with the NBS method. For the children follow-up, which includes newly diagnosed babies, frequencies were calculated for accrual, age at enrollment, sex, percentage of children with hemoglobin SS, and percentage of those receiving hydroxyurea treatment. Accrual, retention, NBS activities, and TCD ultrasound screening were considered for the feasibility evaluation.
All statistical computations were performed and output was generated using SAS Software version 9.4 (The SAS Institute, Cary, NC).
Results
Program infrastructure
Recruitment of staff was completed within the first 6 months. During monthly meetings with the University of Miami staff, study investigators and coordinators were trained in person and virtually to conduct the study. The manual of operations was written in French for better dissemination to all staff.
NBS
Between 25 May 2020 and 24 May 2022, the Haitian sites screened 8224 newborns for hemoglobinopathies. Table 2 shows the number of screenings performed and test results. During the study period, 736 patients (8.95%) were identified to have sickle cell trait, and 61 newborns (0.77%) were identified with SCD (FS, FSC, or FSA). The incidence of SCD was 0.74% at Port-au-Prince and 0.85% in the northern cities of Cap Haitien and Milot. There were 5757 newborns who were screened with IEF alone and 1395 newborns who were screened with either IEF or HPLC and POC. HPLC was performed when the samples were sent to the Florida NBS Laboratory. The remaining samples were screened by either POC alone (n = 162), HPLC alone (n = 791), HPLC and IEF (n = 117), or HPLC, IEF, and POC (n = 2). The results of 266 newborns screened (3.23%) could not be determined by laboratory test. This occurred because of long sample storage of >60 days before running samples. The POC was not performed for 38% of eligible infants because of delays in shipment deliveries, affecting implementation fidelity.
NBS results
| Hemoglobin genotypes . | n (%) . |
|---|---|
| FA (normal pattern) | 6914 (84.07) |
| FA + Barts hemoglobin | 2 (0.02) |
| FAS (sickle cell trait) | 736 (8.95) |
| FAC (hemoglobin C trait) | 226 (2.75) |
| FS (sickle cell anemia) | 44 (0.54) |
| FSC (sickle cell and hemoglobin C) | 13 (0.16) |
| FSA (sickle cell β+-thalassemia) | 4 (0.05) |
| FC (hemoglobin C disease) | 8 (0.10) |
| FAG (hemoglobin G trait) | 1 (0.01) |
| F only (β thalassemia major) | 3 (0.04) |
| Other | 7 (0.09) |
| Not determined | 266 (3.23) |
| Total | 8224 (100) |
| Hemoglobin genotypes . | n (%) . |
|---|---|
| FA (normal pattern) | 6914 (84.07) |
| FA + Barts hemoglobin | 2 (0.02) |
| FAS (sickle cell trait) | 736 (8.95) |
| FAC (hemoglobin C trait) | 226 (2.75) |
| FS (sickle cell anemia) | 44 (0.54) |
| FSC (sickle cell and hemoglobin C) | 13 (0.16) |
| FSA (sickle cell β+-thalassemia) | 4 (0.05) |
| FC (hemoglobin C disease) | 8 (0.10) |
| FAG (hemoglobin G trait) | 1 (0.01) |
| F only (β thalassemia major) | 3 (0.04) |
| Other | 7 (0.09) |
| Not determined | 266 (3.23) |
| Total | 8224 (100) |
Results of screening data obtained from May 2020 to May 2022.
In SDH, a private children’s hospital located at Port-au-Prince, 26 of 42 (62%) children who tested positive assessed by IEF alone returned for follow-up, with 38% of the children who tested positive not followed up despite attempts to notify their families. Two children died perinatally of causes not related to SCD. At JUH, located in the medium-sized city of Cap Haitien, only 4 children were diagnosed with SCD; 2 had HPLC-based screening only, and 2 received 2-test screening; 1 child (50%) in each subgroup returned for follow-up, resulting in no difference when comparing the use of the 2-test approach with using POC (P = 1.0). At SCH, located in a rural area, only 33% of children with positive testing returned for follow-up. Nine of 15 infants who tested positive had testing that included POC; whereas the remaining 6 infants had only the laboratory test performed because the site had run out of POC. POC screening resulted in the successful follow-up of 5 of 9 (56%) children who tested positive compared with 0 of 6 children who tested positive returning to follow-up when the POC was not available (P = .044).
Use of POC devices and availability of laboratory materials
After initial training, the nurses and laboratory technicians performed the POCs proficiently. Gazelle Hemoglobin Variant machines were replaced several times and updated to a more sensitive model that could detect hemoglobin A and S concentrations as low as 4% with 100% accuracy, when compared with the previous model with a lower level of detection of 10% for both hemoglobins.22 Regarding laboratory material availability, we faced prolonged interruptions in the IEF laboratory performance at JUH because of power outages damaging the machine, and delays in delivering hemoglobin gels from outside of the country. Therefore, Florida NBS Laboratory analyzed dried blood samples from the 2 northern sites when such interruptions occurred. The laboratory work from SDH continued without interruption.
Children follow-up
Except for the HUEH site, all sites had children with SCD who were followed up at their outpatient clinics and recruited children for study participation. During the 24-month study period, the families of 165 children consented to participate in follow-up, including 7 children who were identified by NBS. Table 3 shows accrual per site according to the percentage of the clinical population served. The mean age of the children enrolled in follow-up was 42 months, 53% were male, and 93% had hemoglobin SS. Of those children with hemoglobin SS, 13% were receiving hydroxyurea at the time of enrollment.
Accrual from the eligible population (children less than 6 years of age)
| Sites . | Clinical load before the CSHSCD . | Identified by NBS . | Total . | Recruited participants, n (%) . |
|---|---|---|---|---|
| HUEH | 20 | 0 | 20 | 0 (0) |
| JUH | 20 | 4 | 24 | 26 (108)∗ |
| SCH | 47 | 15 | 62 | 48 (77.4) |
| SDH | 300 | 42 | 342 | 91 (26.6) |
| Total | 387 | 61 | 448 | 165 (36.8) |
| Sites . | Clinical load before the CSHSCD . | Identified by NBS . | Total . | Recruited participants, n (%) . |
|---|---|---|---|---|
| HUEH | 20 | 0 | 20 | 0 (0) |
| JUH | 20 | 4 | 24 | 26 (108)∗ |
| SCH | 47 | 15 | 62 | 48 (77.4) |
| SDH | 300 | 42 | 342 | 91 (26.6) |
| Total | 387 | 61 | 448 | 165 (36.8) |
The eligible population comprised children aged <6 years.
JUH has >100% enrollment because this site acquired 2 more patients who were eligible but were not part of the original clinical load and who were not screened by NBS.
TCD ultrasound screening
SCH was the only site that screened children using TCD ultrasound. Monitoring was conducted via WhatsApp, with successful implementation. None of the 6 children screened had a conditional or abnormal result. The other 2 sites did not feel confident implementing TCD ultrasound because the designated person required more training or emigrated from Haiti. However, the SCH sonographer is currently training at least 1 staff member from JUH.
Chronology of systemic barriers
The chronology of implementation barriers is presented in Table 4. Medication outsourcing of penicillin and hydroxyurea did not present problems because we sent these items from Miami. Internet service was poor, delaying data entry and making Zeriscope monitoring of TCD ultrasounds impossible. However, we did not see significant problems or delays in communication with the investigators when using Zoom and WhatsApp.
Systemic implementation barriers
| Year . | Barriers . | Resolution . |
|---|---|---|
| 2019-2020 | Employer strike action at JUH and HUEH The COVID-19 pandemic slowed down clinic attendance in both Haiti and Miami. | Both resolved. |
| 2020-2021 | Electrical outage damaged IEF machine at JUH. | Machine replaced. Surge protector purchased and installed. |
| 2021-2022 | Gang violence at Port-au-Prince; Haitian president was assassinated (7 July 2021); ongoing delays in deliveries of IEF gels to Cap Haitien (not to Port-au-Prince); and emigration of 2 study staff from Haiti resulting in extra training needed for TCD ultrasound sonographers. | Ongoing street violence. Outsourcing of dried blood cards to Florida after brief trials sending them to SDH (there were delays in deliveries, making the samples uninterpretable). 1 site remains without a person responsible for performing TCD ultrasound. |
| 2022-2023 | Strike action at JUH and HUEH; gang violence at Port-au-Prince; and ongoing delays with gel deliveries to Cap Haitien. | Ongoing strikes. Searching for partnerships in the Dominican Republic to shorten the time for gel delivery. |
| Year . | Barriers . | Resolution . |
|---|---|---|
| 2019-2020 | Employer strike action at JUH and HUEH The COVID-19 pandemic slowed down clinic attendance in both Haiti and Miami. | Both resolved. |
| 2020-2021 | Electrical outage damaged IEF machine at JUH. | Machine replaced. Surge protector purchased and installed. |
| 2021-2022 | Gang violence at Port-au-Prince; Haitian president was assassinated (7 July 2021); ongoing delays in deliveries of IEF gels to Cap Haitien (not to Port-au-Prince); and emigration of 2 study staff from Haiti resulting in extra training needed for TCD ultrasound sonographers. | Ongoing street violence. Outsourcing of dried blood cards to Florida after brief trials sending them to SDH (there were delays in deliveries, making the samples uninterpretable). 1 site remains without a person responsible for performing TCD ultrasound. |
| 2022-2023 | Strike action at JUH and HUEH; gang violence at Port-au-Prince; and ongoing delays with gel deliveries to Cap Haitien. | Ongoing strikes. Searching for partnerships in the Dominican Republic to shorten the time for gel delivery. |
Discussion
The CSHSCD implements NBS as one of the most important activities. The incidence of SCD among Haitian newborns is higher than in the United States, where 1 in 365 (0.27%) African American children screen positive for SCD.23 In this report, 0.77% of screened Haitian children tested positive for SCD, with a slight increase of this incidence in the northern region (0.85%) compared with in the capital Port-au-Prince (0.74%). This is in accordance with previously reported higher incidence of SCD in Cap Haitien (1.48%, with confirmation in 0.83%)6 but is higher than reported previously (0.58%) for SDH a decade earlier.5 Therefore, the SCD load among Haitian newborns is significant.
To institute large-scale NBS screening in Haiti, the government may have to fund this effort, or international agencies could assist in funding. In the case of HIV,24 the US President’s Emergency Plan for AIDS Relief25 along with the Haitian government were instrumental in initiating a national HIV case-surveillance known as Suivi Actif Longitudinal du VIH en Haiti, which provided a national network of centers for testing, counseling, prevention of mother-to-child transmission, and therapy.26 Currently, there are no government funds for SCD, although SCD was recognized as a public health priority by Haiti’s Minister of Health in 2022. Despite challenges and limitations, hospital-based NBS was feasible, as demonstrated herein. The NBS nurses did well in obtaining blood samples from heel sticks and performing POC. These are dedicated nurses who are paid by the CSHSCD. For the program to be sustainable beyond grant funding, maternity nurses would have to continue this task, unless the hospitals are capable of continuing paying dedicated NBS staff.
Availability of several POC devices were instrumental for hemoglobinopathy identification, because we anticipated difficulties in obtaining materials and conducting laboratory testing in a low-resource setting. We observed 62% follow-up with the IEF alone in Port-au-Prince within the setting of a children’s hospital. Because we had not added POC to that site, we do not know whether that could have increased the return rate of children. However, the dual screening technique resulted in 56% of children who screened positive being brought back for follow-up when compared with none being brought back when only IEF was used at the rural site, SCH. Although we did not find a difference in follow-up rate at JUH when using the POC, our previous study showed shorter time for follow-up (P = .02) and shorter time for confirmatory test at that site (P = .001) when POC was implemented.6 The POC allowed the staff to educate families of children with positive screening results at the time of birth, possibly increasing adherence. Having a backup result with POCs is another advantage as in the case when dried blood samples could not be processed (eg, screening cards were not available) or were processed too late.
The northern sites faced implementation challenges with laboratory testing: electricity was unstable and laboratory materials took a long time to reach those sites. Costs were prohibitive for them, should they have to pay for such materials. As an alternative, the Florida NBS Laboratory has served as the reference laboratory. It is typically faster to mail samples to Florida than to mail them internally to Port-au-Prince.
We believe that the most reasonable step toward sustainability is the generalization of POCs at all sites, especially at those having problems receiving laboratory materials. Paying for POC tools at the current price will result in $160 USD a week ($8320 USD a year) to screen 40 children at each hospital, which is currently the average neonate load per week.
Despite NBS efforts, Haitian investigators found that the most important reason for the lack of follow-up for infants who tested positive was a fear of gang violence. Some families decided not to proceed with follow-up because their children were doing well and did not manifest any problem. Typically, if the majority of patients who return for follow-up are patients with more severe disease, a bias may occur when measuring clinical outcomes.
Managing children with SCD in Haiti
The sites accrued 36.8% of their clinical population. The reported attrition was low at 2.4%. However, the number may be much higher because some families were not returning because of street violence. The site staff performed very well in following treatment protocols, with 100% penicillin prophylaxis achieved, unless families could not be located after NBS. The main factor that contributed to challenges in care was the lack of availability of oral penicillin (although intramuscular was available) and hydroxyurea, unless the CSHSCD or other sponsors provided these. To address sustainability, we are in the process of assessing the potential for a local pharmaceutical company to import these medications or to buy them from a pharmaceutical company in the Dominican Republic, which is in the process of manufacturing them.
In summary, the CSHSCD faced several barriers, some of which we are addressing and others that might be out of our control. A significant barrier was the NBS follow-up rate. Lack of follow-up or an inability to locate families also occurs in the United States. Follow-up for the 3 sites ranged between 33% and 62%. The POC made a statistical difference in follow-up at the rural site. One attempt to improve follow-up was by employing CHWs, who have been effective in maternal-child health preventive interventions27 and are respected and trusted in Haiti.28 Even if CHWs were employed by the CSHSCD, some families could not be reached or refused to have their children followed up. Poverty, lack of resources, gang violence, and distrust in the SCD diagnosis all may play a role in the lack of follow-up. Improvement in other areas of care, such as enhancing TCD ultrasound screening and a wider use of hydroxyurea, will take longer to achieve, requiring training workers, educating families about the importance of stroke risk screening, and using hydroxyurea as the main source of primary stroke prevention to offset a very limited blood transfusion supply in Haiti.
Despite these barriers, our results show that sickle cell follow-up and NBS programs are feasible in Haiti. Local staff were engaged in the study. Infants were screened at birth and enrolled in the study. The work of the SDH’s independent laboratory was sustainable. Having POCs for screening at the rural hospital achieved better follow-up rates. The CSHSCD confirmed a high incidence of births screening positive for SCD. To be more effective in NBS follow-up, we propose to evaluate whether all newborns might benefit from screening with the double method. Focus groups were conducted to elucidate barriers and determine ways to overcome them. Results from the focus groups will be reported elsewhere. Although enablers and barriers have been examined in other low-resource populations,29 the barriers encountered in Haiti might be unique and are currently under review for a future attempt to enhance implementation.
Acknowledgments
The authors are indebted to the study nurse coordinators Paulonne Bien-Aimé, Mimose Voltaire, Woodgina Mondesir, and Gurvitch Julien. Other staff contributors were Gina St. Eloi, Jeef Alix, Michelia Pierre, Ruth Lundy, Anne Stephanie Joseph, and Osly Vilceus (at SDH); Regine Victorin, Pascal Blaise, Judelyne Chery, Floye Denis, Nirva Fils-Aimé, and Liline Garcon (at SCH); Lourdes D. Belony, Carline Joseph, Jennie C. Désir, and Ulrick Liberus (at JUH); Thelesta Pavelus, Ladie-Wisnise Damas, and Marlène Petit-Frère (at HUEH). The authors are indebted to Jose Romano, Nelly Campo, and Iszet Campo-Bustillo from the University of Miami and Robert Adams from the Medical University of South Carolina for conducting the TCD ultrasound training. The authors especially thank families who agreed to NBS and to participating in the clinical follow-up of their children as part of this study.
The authors acknowledge the National Institutes of Health and Fondation Pierre Fabre for their economic support and the Florida NBS Laboratory for their generous contribution in analyzing NBS samples for quality assurance and as backup for the northern Haitian sites.
Authorship
Contribution: O.A.A. was responsible for study design and overall supervision, and manuscript writing; N.S.V.D. was responsible for data entry and implementation at SDH; M.P.H. was an SCH investigator; R.S.F. and E.M. served as JUH center investigators; R.E. served as HUEH center investigator; E.L. served as SDH center investigator; T.H. and M.C. were responsible for University of Miami study coordination and overall study support coordination; S.M. was responsible for data analysis; E.C.B. was responsible for implementation review and counseling; L.H.M. was responsible for cultural counseling; and all authors have contributed to the manuscript with review and comments and approved the content.
Conflict-of-interest disclosure: O.A.A. has participated in advisory boards for Novartis and Global Blood Therapeutics. The remaining authors declare no competing financial interests.
Correspondence: Ofelia A. Alvarez, Mailman Center for Child Development, University of Miami Miller School Medicine, 1601 NW 12th Ave, Room 5048, Miami, FL 33136; email: oalvarez2@med.miami.edu.
References
Author notes
Data are available on request from the corresponding author, Ofelia A. Alvarez (oalvarez2@med.miami.edu).
The full-text version of this article contains a data supplement.