Key Points
The diagnostic IDH mutation status in AML did not significantly influence outcomes following HSCT.
IDH1 R132 and IDH2 R172 positive MRD at HSCT is associated with inferior outcomes.
Abstract
Somatic mutations in the isocitrate dehydrogenase 1 and 2 genes (IDH1 and IDH2) are common in acute myeloid leukemia (AML). The prognostic impact of the presence of IDH mutations may be influenced by the comutational status, the specific location of the mutation (ie, IDH1 R132, IDH2 R140, and IDH2 R172) at diagnosis, and the dynamics of the mutation burden during disease course. Even though many patients with IDH-mutated AML are consolidated by hematopoietic stem cell transplantation (HSCT), the underlying biology and prognostic consequences remain largely unknown. Here, we present a large analysis of 292 patients with AML who received HSCT in complete remission (CR) or CR with incomplete peripheral recovery (CRi), in which we assessed the IDH mutation status at diagnosis and HSCT as a potential marker for measurable residual disease (MRD). About a quarter of all patients were IDH-mutated at diagnosis. The diagnostic presence of IDH mutations in AML did not have a significant prognostic impact when consolidated with HSCT. However, IDH1 R132 and IDH2 R172 MRD positivity in remission at HSCT associated with an increased risk of relapse, while IDH2 R140 mutations did not. The IDH2 R140 variant allele frequency (VAF) at diagnosis was higher, clustering around 50%, and the mutation clearance at HSCT in morphologic remission was much lower compared with IDH1 R132 and IDH2 R172. In our cohort, IDH2 R140 mutations behaved more like a clonal hematopoiesis-related aberration, while IDH1 R132 and IDH2 R172 harbored AML disease-specific features.
Introduction
Somatic mutations in the isocitrate dehydrogenase 1 and 2 genes (IDH1 and IDH2) are found in about 15% to 20% of patients with acute myeloid leukemia (AML),1,2 with a higher prevalence in those with normal karyotype and/or an additional NPM1 mutation.3IDH mutations lead to neomorphic enzyme activity, resulting in the production of the oncometabolie 2-hydroxyglutarate (2HG), which is associated with DNA and histone hypermethylation, altered gene expression, and blocked differentiation of hematopoietic progenitor cells, as well as inhibited TET2 (Tet methylcytosine dioxygenase 2).4-6
The prognostic significance of mutated IDH in patients with AML remains controversial, with some groups reporting distinct outcomes within specific patient subsets, dependent on the biological and clinical context.1,2,7-10 Additionally, the prognostic impact seems to be influenced by the comutation status and the specific location of the mutation (ie, regarding the hotspot locations IDH1 R132, IDH2 R140, and IDH2 R172). Particularly the IDH2 R172 may be associated with improved outcomes following chemotherapy treatment.9-11 Patients with IDH1 mutation have been reported to have inferior outcomes in the context of an NPM1 mutation lacking an FLT3-ITD.1-3,7IDH mutations have also been suggested to persist in complete remission as clonal hematopoiesis of indeterminate potential (especially the IDH2 R140)12,13 and to associate with older age.3,12,14
Multiple inhibitors targeting mutated IDH are being developed, including ivosidenib (AG-120) and enasidenib (AG-221), which have already been approved by the US Food and Drug Administration (FDA) for patients with IDH1- and IDH2-mutated AML, respectively, allowing a targeted treatment option for these patients.15-17 Furthermore, IDH-mutated AML may have especially favorable outcomes following acazitidine/venetoclax combination treatment.18 Today, allogeneic hematopoietic stem cell transplantation (HSCT) remains the consolidation therapy with the highest chance of sustained remission for most younger and older patients with AML, independent of the underlying biology.19-22 Even though many patients with IDH-mutated AML are consolidated by HSCT and several trials testing IDH inhibitors in a maintenance setting are active, very little data are available on the influence of IDH mutations in the HSCT context. Salhotra and colleagues suggested higher relapse rates of IDH-mutated AML following HSCT; however, the study was limited by low patient numbers, with only 23 patients carrying an IDH mutation.23 Chen and colleagues very recently showed 2-year relapse rates of 31% for IDH1-mutated and 25% for IDH2-mutated AML in 112 patients receiving HSCT.24 But none of these studies analyzed IDH1 R132, IDH2 R172, and IDH2 R140 mutations separately or evaluated the impact of the detection of these mutations in remission at HSCT. Here, we analyzed the impact of mutated IDH at AML diagnosis and in remission at HSCT in a large single-center set of patients with AML receiving consolidating allogeneic HSCT.
Methods
Patients and treatment
We retrospectively analyzed 292 adult patients with AML with a median age at diagnosis of 63.7 years (range, 21.7-82.3 years) who received allogeneic HSCT at the University of Leipzig between August 2007 and September 2020. For all patients, diagnostic bone marrow (BM) samples were available. For outcome analyses, we restricted our analyses to patients with AML who received an HSCT in complete remission (CR) or CR with incomplete peripheral recovery (CRi) (n = 235). All but 2 patients received age-dependent intensive chemotherapy protocols (under or over 60 years); 2 patients were treated with acazitidine only. One patient received enasidenib as bridging therapy before HSCT, while none of the other patients received an IDH inhibitor. All were consolidated with HSCT in first (n = 152), second (n = 32), or third CR (n = 1) or CRi (n = 50). For detailed information on the conditioning regimens and graft-versus-host disease prophylaxis, please see the supplemental Data. Median follow-up after HSCT for patients alive was 3.9 years. Retrospective analyses were approved by the University of Leipzig Medical Center Institutional Review Board. Written informed consent for participation in these studies was obtained in accordance with the Declaration of Helsinki.
Assessment of IDH mutation status at diagnosis and at HSCT
For IDH1 and IDH2 mutational analyses at diagnosis, we amplified and directly sequenced DNA fragments spanning exons 4 of IDH1 and IDH2, as previously described.3 For patients with adequate material available (n = 125), IDH mutation status was assessed and verified by next-generation sequencing (NGS) as previously described.12 Consequently, for these patients, the BM variant allele frequency (VAF) at diagnosis was available.
At HSCT, we analyzed the IDH mutation status by NGS as previously described (n = 107; n = 28 with mutated IDH).12 Since the NGS analyses were technically restricted to a VAF sensitivity of 3%, we developed more sensitive mutation-specific droplet digital polymerase chain reaction (ddPCR) assays to assess IDH1 R132, IDH2 R140, and IDH2 R172 at HSCT at measurable residual disease (MRD) levels for those patients with adequate material available (n = 44). For further details on the ddPCR assays, please consult the supplemental Data, supplemental Table 1, and supplemental Figure 1. Although limited by the number of patients with detectable IDH mutations at HSCT by NGS, the IDH mutation burden correlated well when we compared the results derived from NGS or ddPCR techniques (see also supplemental Data; supplemental Figure 2).
Cytogenetics, flow cytometry, and additional molecular markers
Cytogenetic analyses were performed centrally in our institution using standard banding techniques. In cases where no metaphases could be obtained, fluorescence in-situ hybridization was used to screen for recurrent abnormalities [ie, del5/5q, del7/7q, trisomy 8, inv(3), abn11q23, t(8;21), inv(16), and t(15;17)]. At diagnosis, the presence of internal tandem duplication in the FLT3 gene (FLT3-ITD), mutations in the FLT3 tyrosine kinase domain (FLT3-TKD), and in the NPM1 and CEBPA genes were determined as previously described.25
A panel of AML-associated genes was analyzed using the NGS platform MiSeq (Illumina) for 125 patients with available material at diagnosis.12 Patients were grouped according to the European LeukemiaNet (ELN) 2017 classification into 3 risk groups.20,25
For 205 patients with material available, the BM CD34+/CD38− cell burden at diagnosis, as well as additional flow markers, were determined as previously described,26 and the results are presented in the supplemental Data.
Analysis for MRD markers at HSCT (NPM1 mutation burden, as well as BAALC and MN1 expression) for patients with material available (n = 51, n = 145, n = 147, respectively) were evaluated as previously described.27-29
Definition of clinical endpoints and statistical analyses
For event-free survival (EFS) and overall survival (OS), survival estimates were calculated using the Kaplan-Meier method, and groups were compared with the log-rank test. Cumulative incidence of relapse (CIR) was calculated considering the competing risk nonrelapse mortality (NRM) using the Fine and Gray model.30 Associations with baseline clinical, demographic, and molecular features were compared using the Kruskal-Wallis test and Fisher’s exact test for continuous and categorical variables, respectively. Statistical analyses were performed using the R statistical software platform (version 4.0.3).31 For further details, see the supplemental Data.
Results
Diagnostic IDH1 and IDH2 mutations
In the analyzed cohort of 292 patients, we observed 70 (24.0%) patients with IDH mutation; 11.4% harbored an IDH1 mutation, all of which were R132 substitutions; 14.0% had an IDH2 mutation, with 8.9% of the patients harboring R140 substitutions, and 5.1% of the patients with R172 substitutions. IDH1 and IDH2 mutations were mutually exclusive in our cohort, except for 1 patient that had an IDH1 R132H and an IDH2 R140Q mutation.
Clinical and biological characteristics associated with IDH mutations are shown in Table 1. Patients with IDH mutation had higher BM blasts at diagnosis and were more frequently DNMT3A-mutated, while a monosomal karyotype, RUNX1 mutations, and TP53 mutations were found in lower frequencies. Associated immunphenotypical characteristics are shown in supplemental Table 2 and supplemental Figure 3. The CD34+/CD38− cell burden was not different with respect to the IDH mutation status (data not shown).
Clinical and biological characteristics according to the IDH mutation status
| Characteristics . | IDH wt (n = 222) . | IDH-mutated (n = 70) . | P . |
|---|---|---|---|
| Age (yr) at diagnosis median (range) | 63.1 (21.7-82.3) | 64.9 (31.2-78.2) | .23 |
| Sex, n (%) female | 116 (52) | 39 (56) | .68 |
| Hb, g/dL median (range) | 8.9 (4-15.68) | 8.2 (4.3-13.4) | .09 |
| Platelets, 109/L median (range) | 65 (1-501) | 66 (2-950) | .22 |
| WBC, 109/L median (range) | 6.3 (0.7-366) | 3.9 (0.8-385) | .96 |
| Blasts BM, % median (range) | 50 (3-95) | 61.5 (16-95) | .02 |
| Blasts PB, % median (range) | 20 (0-97) | 32 (1-97) | .15 |
| ELN2017, n (%) | |||
| Favorable | 47 (23) | 16 (27) | .33 |
| Intermediate | 69 (34) | 25 (42) | |
| Adverse | 85 (42) | 19 (32) | |
| Monosomal KT, n (%) present | 30 (14) | 2 (3.1) | .01 |
| NPM1, n (%) mutated | 51 (23) | 17 (25) | .87 |
| FLT3-ITD, n (%) present | 39 (18) | 13 (19) | .86 |
| CEBPA, n (%) mutated | 28 (14) | 8 (12) | .84 |
| ASXL1, n (%) mutated | 11 (12) | 6 (15) | .78 |
| RUNX1, n (%) mutated | 17 (20) | 1 (2.6) | .01 |
| TP53, n (%) mutated | 15 (17) | 0 (0) | .01 |
| DNMT3A, n (%) mutated | 19 (22) | 19 (48) | .01 |
| Characteristics . | IDH wt (n = 222) . | IDH-mutated (n = 70) . | P . |
|---|---|---|---|
| Age (yr) at diagnosis median (range) | 63.1 (21.7-82.3) | 64.9 (31.2-78.2) | .23 |
| Sex, n (%) female | 116 (52) | 39 (56) | .68 |
| Hb, g/dL median (range) | 8.9 (4-15.68) | 8.2 (4.3-13.4) | .09 |
| Platelets, 109/L median (range) | 65 (1-501) | 66 (2-950) | .22 |
| WBC, 109/L median (range) | 6.3 (0.7-366) | 3.9 (0.8-385) | .96 |
| Blasts BM, % median (range) | 50 (3-95) | 61.5 (16-95) | .02 |
| Blasts PB, % median (range) | 20 (0-97) | 32 (1-97) | .15 |
| ELN2017, n (%) | |||
| Favorable | 47 (23) | 16 (27) | .33 |
| Intermediate | 69 (34) | 25 (42) | |
| Adverse | 85 (42) | 19 (32) | |
| Monosomal KT, n (%) present | 30 (14) | 2 (3.1) | .01 |
| NPM1, n (%) mutated | 51 (23) | 17 (25) | .87 |
| FLT3-ITD, n (%) present | 39 (18) | 13 (19) | .86 |
| CEBPA, n (%) mutated | 28 (14) | 8 (12) | .84 |
| ASXL1, n (%) mutated | 11 (12) | 6 (15) | .78 |
| RUNX1, n (%) mutated | 17 (20) | 1 (2.6) | .01 |
| TP53, n (%) mutated | 15 (17) | 0 (0) | .01 |
| DNMT3A, n (%) mutated | 19 (22) | 19 (48) | .01 |
BM, bone marrow; ELN2017, European LeukemiaNet 2017; Hb, hemoglobin; KT, karyotype; PB, peripheral blood; WBC, white blood cells; wt, wild-type.
For patients with IDH1 mutations (see supplemental Table 3), we observed a lower white blood cell count at diagnosis. In patients with IDH2-mutated AML (see supplemental Tables 4 and 5), no cooccurring of monosomal karyotypes, RUNX1 mutations, or TP53 were found, but DNMT3A mutations were observed more frequently. Comutations at diagnosis for patients with available data are displayed in Figure 1A.
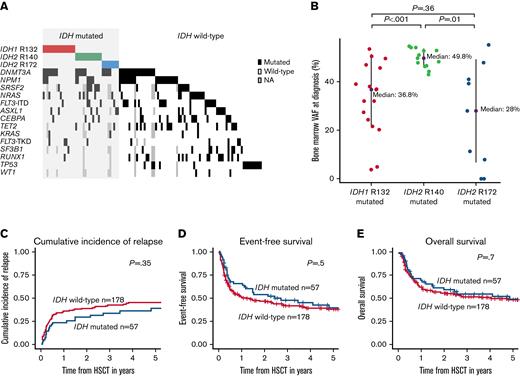
Characteristics and outcomes according to IDH mutation status at diagnosis. (A) Comutations in patients with IDH mutation (ie, IDH1 R132, IDH2 R140, IDH2 R172) and wild-type at diagnosis (n = 125) consolidated by HSCT. (B) BM VAF at diagnosis according to IDH mutation status. (C) CIR, (D) EFS, and (E) OS of HSCT-consolidated patients with AML according to IDH mutation status.
Characteristics and outcomes according to IDH mutation status at diagnosis. (A) Comutations in patients with IDH mutation (ie, IDH1 R132, IDH2 R140, IDH2 R172) and wild-type at diagnosis (n = 125) consolidated by HSCT. (B) BM VAF at diagnosis according to IDH mutation status. (C) CIR, (D) EFS, and (E) OS of HSCT-consolidated patients with AML according to IDH mutation status.
In general, patients with IDH-mutation did not differ significantly regarding their age in this set (Table 1); however, patients with IDH2 and especially IDH2 R140 mutation had the highest median age at diagnosis (patients with IDH wild type: 63.1 years; patients with IDH1 mutation: 63.5 years; patients with IDH2 R172 mutation: 64.5 years; and patients with IDH2 R140 mutation: 66.5 years). Noteworthy, while 40% of IDH2 R140 also harbored NPM1 mutations, none of the IDH2 R172 were NPM1-mutated (Figure 1A; supplemental Table 5) (P = .02), an observation in line with previous publications.9,10
For the diagnostic BM VAFs associated with the 3 IDH mutation types, we observed a mutation-specific pattern. While IDH2 R140 mutations clustered around a median VAF of about 50%, the VAFs for IDH1 R132 and IDH2 R172 mutations at diagnosis were significantly lower with a wider distribution (Figure 1B).
With respect to the outcome of patients with AML following HSCT, we observed no difference according to the IDH mutation status at diagnosis (Figure 1C-E). This also held up when we analyzed the IDH1 R132 mutations and IDH2 R140 and IDH2 R172 mutations separately (supplemental Figures 4 and 5). Also, in multivariable analyses, the diagnostic IDH mutation status did not significantly associate with outcomes (supplemental Table 6).
IDH mutation status and ELN2017 risk stratification
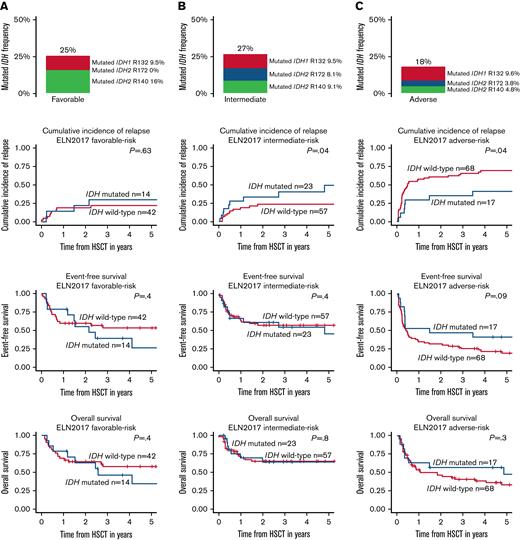
We found the distribution of IDH mutations differed according to the ELN2017 risk classification (Figure 2). While the frequency of IDH1 mutations was similar in all 3 groups (ELN2017 favorable risk: 9.5%; intermediate risk: 9.6%; adverse risk: 9.6%), the frequency of IDH2 R140 decreased from favorable risk (16%) to intermediate risk (9.6%) to adverse risk (4.8%). IDH2 R172 mutations were most frequent in the intermediate-risk group (8.5%) and absent in the ELN2017 favorable-risk group.
IDH mutation (ie, IDH1 R132, IDH2 R140, IDH2 R172) frequency, CIR, EFS, and OS of HSCT-consolidated patients with AML according to the ELN2017 risk groups. (A) ELN2017 favorable, (B) ELN2017 intermediate, and (C) ELN2017 adverse.
IDH mutation (ie, IDH1 R132, IDH2 R140, IDH2 R172) frequency, CIR, EFS, and OS of HSCT-consolidated patients with AML according to the ELN2017 risk groups. (A) ELN2017 favorable, (B) ELN2017 intermediate, and (C) ELN2017 adverse.
With respect to outcomes following HSCT consolidation in the ELN2017 favorable-risk group, IDH mutation status did not impact outcomes (Figure 2A). In the ELN2017 intermediate-risk group, patients with IDH mutation had an increased relapse rate compared with patients with IDH wild-type, which did not translate into shorter EFS or OS (Figure 2B). Within the ELN2017 adverse-risk group, patients with IDH mutation had a lower CIR and, by trend, longer OS and EFS (Figure 2C). However, IDH mutations are also associated with lower frequencies of the particularly adverse-risk factors monosomal karyotypes and TP53 mutations. When these characteristics were excluded in a subanalysis (supplemental Figure 6), with the caveat of low patient numbers, a trend for a lower CIR was observed (P = .07), while there was no impact of the IDH mutation status on EFS and OS. Noteworthy, patients with IDH-mutated AML grouped by the ELN2017 risk classification did not have significantly different outcomes (supplemental Table 7).
Persistence of IDH mutations at HSCT
Forty-six patients with IDH mutation had material for IDH mutation status at HSCT available. Two had NGS but no ddPCR data available, and 17 had no NGS but ddPCR data available. In general, for 44 patients with IDH1 or IDH2 mutations, material for ddPCR-based MRD detection was available, of which 33 (75%) had detectable IDH MRD.
The patients with MRD-positive ddPCR-based IDH mutation were differently distributed (IDH1 R132: 64.7%; IDH2 R140: 94.1%; IDH2 R172: 44%). No clear association of the IDH VAF at HSCT and relapse or an ELN2017 risk group could be observed (Figure 3A). However, the VAFs at HSCT were much lower in IDH1 R132 (0.17%) and IDH2 R172 (0.20%) compared with IDH2 R140 (11.6%) (Figure 3A,B).
Characteristics and outcomes according to IDH mutation status at HSCT. (A) ddPCR-based MRD at HSCT in patients with IDH-mutated (ie, IDH1 R132, IDH2 R140, IDH2 R172) AML. (B) Pre-HSCT MRD VAF according to IDH mutation status. (C) CIR, (D) EFS, and (E) OS of HSCT-consolidated patients with AML according to optimized IDH MRD status (IDH1 R132 or IDH2 R172 MRD-positive n = 5; IDH2 R140 MRD-positive n = 11; IDH MRD-negative n = 28).
Characteristics and outcomes according to IDH mutation status at HSCT. (A) ddPCR-based MRD at HSCT in patients with IDH-mutated (ie, IDH1 R132, IDH2 R140, IDH2 R172) AML. (B) Pre-HSCT MRD VAF according to IDH mutation status. (C) CIR, (D) EFS, and (E) OS of HSCT-consolidated patients with AML according to optimized IDH MRD status (IDH1 R132 or IDH2 R172 MRD-positive n = 5; IDH2 R140 MRD-positive n = 11; IDH MRD-negative n = 28).
Using an optimized mutation-specific cutoff (determined as described in the supplemental Data [supplemental Figure 8]), we observed a strong relapse association of IDH1 R132 positivity or IDH2 R172 positivity, while IDH2 R140 MRD positivity associated with an optically elevated but not significantly different CIR curve (Figure 3C). Patients that were MRD-positive for IDH1 R132 or IDH2 R172 mutations also had a shorter, though not significant, EFS and OS (Figure 3D,E). Of the 5 patients with either IDH1 R132 (n = 2) or IDH2 R172 (n = 3) positivity, none harbored an NPM1 mutation, and only 1 patient harbored a DNMT3A mutation. In 3 of the 5 patients, the relapse risk could not be predicted by an elevated BAALC or MN1 expression at the time of HSCT. Of the 11 patients who were IDH2 R140 MRD-positive, 8 harbored an NPM1 mutation, of whom 1 was positive for NPM1 mutation-based MRD at HSCT and was also the only patient suffering relapse after HSCT.
Discussion
In AML, the prognostic impact of IDH mutations in patients consolidated with chemotherapy remains ambiguous, with differing results in some larger retrospective studies.1-3 After consolidation with HSCT, the prognostic impact is even less clear. In the here analyzed set of HSCT-consolidated patients with AML, the presence of an IDH mutation at diagnosis did not significantly impact CIR, EFS, or OS following HSCT. This also held up when we analyzed IDH1 R132, IDH2 R140, and IDH2 R172 mutations separately. Patient relapse rates at 2 years of IDH1- and IDH2-mutated AML following HSCT were 31% and 29%, respectively, and thus similar to those reported by Chen and colleagues.24
In general, the observed clinical and biological associations (eg, a high diagnostic BM blast count, a high frequency of DNMT3A comutations, and low frequencies of TP53 mutations in patients with IDH mutation) were similar to previously published data.10,24,32
One of the most commonly applied prognostic systems in AML is the ELN2017 risk classification, with 3 risk groups that have been shown to harbor prognostic information, also in patients with AML consolidated with HSCT.25 When we analyzed the impact of IDH mutations within these groups, we observed no distinct outcomes within the ELN2017 favorable-risk group. Relapse rates were higher in intermediate-risk patients with IDH-mutated ELN2017, but this did not translate into EFS or OS differences. In the ELN2017 adverse-risk group, relapse rates were lower, and by trend EFS and OS were longer in patients with IDH mutation. Interestingly, Eisfeld and colleagues described an improved outcome for younger patients with IDH2-mutated AML with ELN2017 adverse risk consolidated with chemotherapy.33 However, in our set, patients with IDH mutations had a low frequency of monosomal karyotype and lacked TP53 mutations, both of which have an especially dismal prognosis even in the ELN2017 adverse-risk group,25 an association that may result from a selection effect since we only included transplanted patients. When we excluded these subgroups from our analysis, no significant difference for CIR, EFS, or OS was observed with respect to the IDH mutation status. In fact, patients with IDH mutation grouped by the ELN2017 risk classification did not have different outcomes (supplemental Data). Thus, at least following HSCT consolidation, IDH-mutated AML may represent a prognostic entity not fully captured by the current ELN2017 risk classification.
Recently, a positive IDH mutation status in patients with AML has been linked to cardiovascular complications during cytotoxic treatment.34 While we lack detailed cardiovascular information on the here analyzed patients, we did not observe a different NRM according to the IDH mutation status in the patients undergoing HSCT (supplemental Figure 9).
Some previous studies have indicated that IDH1 R132, IDH2 R140, and IDH2 R172 mutations might cause diverse AML phenotypes.3,11 Despite their common pathomechanism involving 2HG production and similar outcomes when detected at AML diagnosis following HSCT, the 3 hotspot mutations may mediate different disease biologies, leading to clinical and therapeutical consequences.
Here we observed different features of the 3 hotspot mutations, including an ELN2017 risk group-dependent distribution, differential comutations, immunophenotypical differences, and VAF patterns (Table 2). A considerable property of the IDH2 R140 vs the IDH1 R132 or IDH2 R172 mutation is the longitudinal mutation behavior. For the IDH2 R140 at diagnosis, we observed clustering of the VAF around 50%, much higher and tighter compared with the other 2 mutation hotspots. This distinct pattern (although lower) was also reflected in the patients in remission. All but 1 patient did not clear the IDH2 R140 mutation before HSCT, with a median VAF at HSCT of still 11.6%. Furthermore, the prognostic impact of detectable IDH2 R140 at HSCT was limited compared with the other 2 mutations. Subsequently, it appears that IDH2 R140 may have more properties of a clonal hematopoiesis-related mutation, while IDH1 R132 and IDH2 R172 may be more AML disease-specific. Noteworthy, in a second small set of patients with IDH-mutated, transplanted AML with the material in remission before HSCT was available, similar results could be obtained (see supplemental Data). However, our sample size is limited, and these properties should be validated in further longitudinal studies but may be considered in future clinical trials.
Clinical and biological features of IDH mutation types
| IDH1 R132 . | IDH2 R140 . | IDH2 R172 . |
|---|---|---|
| Equally distributed over ELN2017 risk groups | Most common in the ELN2017 favorable group | Most common in the ELN2017 intermediate group |
| Lower and more spread diagnostic VAF | Diagnostic VAF clustering around 50% | Lower and more spread diagnostic VAF |
| Low clearance rate in remission | ||
| Limited prognostic impact as MRD in transplanted patients with AML | ||
| High NPM1 comutation rate | Low NPM1 comutation rate | |
| Low CD34 surface expression | High CD34 surface expression | |
| High CD33 surface expression | ||
| High CD117 surface expression |
| IDH1 R132 . | IDH2 R140 . | IDH2 R172 . |
|---|---|---|
| Equally distributed over ELN2017 risk groups | Most common in the ELN2017 favorable group | Most common in the ELN2017 intermediate group |
| Lower and more spread diagnostic VAF | Diagnostic VAF clustering around 50% | Lower and more spread diagnostic VAF |
| Low clearance rate in remission | ||
| Limited prognostic impact as MRD in transplanted patients with AML | ||
| High NPM1 comutation rate | Low NPM1 comutation rate | |
| Low CD34 surface expression | High CD34 surface expression | |
| High CD33 surface expression | ||
| High CD117 surface expression |
ELN2017, European LeukemiaNet 2017; MRD, minimal residual disease; VAF, variant allele frequency.
In remission at HSCT, IDH mutations persisted in high frequencies (75%), although at a low median VAF level of 1.2%. However, the frequency of persistence and the median persisting VAF level was dependent on the IDH mutation type, and especially the presence of IDH1 R132 and IDH2 R172 at HSCT was associated with worse outcomes. Thus, clinically, the elimination of persisting IDH mutations (especially of IDH1 R132 and IDH2 R172) before HSCT could be an important milestone toward a cure for these patients.
The data also indicate that more sensitive methods of MRD detection (such as error-corrected NGS or the here used ddPCR) compared with traditionally applied NGS analyses with a higher limit of detection (eg, 5% to 2% VAF) may be more clinically informative and relevant.
Recently, the FDA approved ivosidenib and enasidenib for the treatment of adult patients with relapsed or refractory AML with mutated IDH1 or IDH2, respectively. Both drugs showed single-agent activity with overall response rates of around 40%, with a median response duration of about 6 months in phase 1 studies.16,17 However, a phase 3 trial testing enasidenib vs conventional regimens in relapsed or refractory AML failed to meet its primary endpoint of OS. The combination of ivosidenib and azacytidine in a phase 1 trial was well tolerated in newly diagnosed AML ineligible for intensive therapy with promising results.35 Ivosidenib or enasidenib was also tested in combination with intensive chemotherapy in newly diagnosed patients with IDH-mutated AML.36 The combination was well tolerated with encouraging initial clinical activity. Given these trial results, the role of IDH inhibitors within AML treatment concepts has still to be defined today. Randomized, controlled trials are needed to compare response rates for IDH inhibitors in combination with intensive and nonintensive chemotherapies. Certainly, these trials should incorporate IDH mutation MRD monitoring.
In the context of consolidating HSCT in IDH-mutated AML, it will be interesting to see if deeper remissions (defined by IDH MRD VAF at HSCT) by novel induction concepts and/or trials testing IDH inhibitors as maintenance treatment (eg, after HSCT) will improve outcomes of patients with IDH-mutated AML in the future.
For now, in conclusion, we showed here that while the diagnostic presence of IDH mutations in AML did not have a strong prognostic impact following HSCT consolidation, the presence, especially of IDH1 R132 and IDH2 R172 mutations at higher MRD levels in remission, associated with an increased risk of relapse following HSCT.
Acknowledgments
The authors thank Janet Bogardt, Annette Jilo, Dagmar Cron, Ines Kovacs, Kathrin Wildenberger, Scarlett Schwabe, Manuela Quandt, Claudia Diener, Christine Günther, Daniela Bretschneider, Evelin Hennig, and Christel Müller for their assistance.
This work was supported by the Deutsche José-Carreras-Stiftung (#04R/2016 and #PS15/05 [J.G.]), Verein zusammen gegen den Krebs e.V., Deutsche Gesellschaft für Innere Medizin (M.J.), an intramural scholarship by the University of Leipzig (L.B.), and Ein Herz für Kinder e.V. M.B. is funded by the German Cancer Aid as a fellow of the Mildred Scheel Early Career Center.
Authorship
Contribution: M.B. and S.S. contributed to the design and analysis of this study and the writing of the manuscript; J.G., L.B., M.J., L.K.S., M.B., D. Backhaus, D. Brauer, and K.G. carried out the laboratory-based research; M.J., J.K., and S.S. performed statistical analyses; A.S.M., G.-N.F., V.V., D.N., U.P., A.-K.E., and S.S. provided administrative support; and all authors agreed on the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Marius Bill, Medical Clinic and Policlinic I, University Hospital of the Technische Universität Dresden, Fetscherstrasse 74, 01307 Dresden, Germany; e-mail: marius.bill@uniklinikum-dresden.de; and Madlen Jentzsch, Medical Clinic and Policlinic 1, Hematology Cellular Therapy and Hemostaseology, University of Leipzig Medical Center, Leipzig, Liebigstraße 22, Haus 7, 04103 Leipzig, Germany; e-mail: madlen.jentzsch@medizin.uni-leipzig.de.
References
Author notes
Original data are available by e-mail request to the corresponding authors, Marius Bill (marius.bill@uniklinikum-dresden.de) and Madlen Jentzsch (madlen.jentzsch@medizin.uni-leipzig.de).
The full-text version of this article contains a data supplement.
Sebastian Schwind died on 17 December 2022.