TO THE EDITOR:
FLT3 internal tandem duplications (ITD) mutations are found in 25% of patients with acute myeloid leukemia (AML).1 In younger patients, FLT3-ITD is associated with increased relapse and decreased survival.2 However, there are limited data on the prognostic implication of FLT3-ITD+ in older patients (aged ≥60 years) who received treatment, particularly, after the approval of FLT3 inhibitors3,4 and venetoclax (ven).5
We investigated the prevalence and impact of FLT3-ITD+ mutations in a cohort of older patients (aged ≥60 years) with newly diagnosed (ND) AML treated at the Dana-Farber Cancer Institute (DFCI). We included consecutive patients with ND-AML treated with intensive chemotherapy (IC): 7 + 3 or CPX-351 with or without an additional therapeutic agent, or hypomethylating agent (HMA)–based therapy (with or without ven). Groups were defined based on the presence (FLT3-ITD+) or absence (FLT3-ITD−) of a FLT3-ITD mutation based on our next-generation sequencing panel (informatic methods for detecting FLT3-ITDs with a sensitivity of 1% allelic ratio as previously described).6 Patients with FLT3-TKD mutations were considered to be FLT3-ITD−. We excluded patients without diagnostic molecular information and those who received treatments other than IC or HMA. This study was conducted with the approval of the institutional review board at the DFCI and in accordance with the Declaration of Helsinki.
Responses were defined based on the European Leukemia Net 2017 criteria7 and were available for patients treated with IC or HMA + ven. Composite complete response (cCR) was complete response (CR) plus CR with incomplete count recovery (CRi). The verall survival (OS) was assessed using the Kaplan-Meier method, with comparisons using the log-rank test. We used Cox regression analyses to assess the effect of covariates on survival. Because we included allogeneic stem cell transplantation (allo-SCT) as a time-varying covariate, the analyses were performed only among patients who could be considered as allo-SCT candidates based on age (≤75 years) alone. Multivariable model selection was based on Bayesian information criterion using a backward-forward algorithm. A 2-sided P value < .05 was considered statistically significant. Statistics were performed using R version 4.0.4.
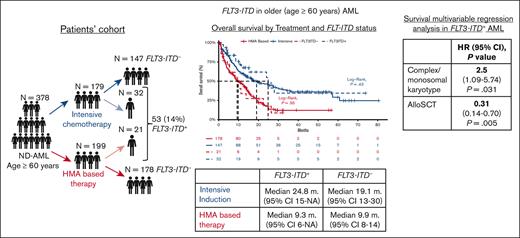
We identified 378 patients aged ≥60 years with ND-AML treated at DFCI between August 2014 and December 2021 (supplemental Figure 1). FLT3-ITD+ mutations were observed in 53 patients (14%), with a median allelic ratio of 28% (interquartile range, 3%-52%). IC was used in 179 (47%) patients, and 199 (53%) were treated with HMA (110 [55%] with and 89 [45%] without venetoclax). Patients’ characteristics are summarized in Table 1.
Patients and treatment characteristics
| . | All, 378 (n %) . | FLT3-ITD+, 53 (n %) . | FLT3-ITD−, 325 (n %) . | P . |
|---|---|---|---|---|
| Age (y, median, range) | 69, (60, 89) | 68 (60, 87) | 70, (60, 89) | .185 |
| Age groups | ||||
| <70 | 189, 50% | 29, 54.7% | 160, 49.2% | .459 |
| ≥70 | 189, 50% | 24, 45.3% | 165, 50.8% | |
| Sex | ||||
| Male | 225, 59.5% | 29, 54.7% | 196, 60.3% | .442 |
| Female | 153, 40.5% | 24, 45.3% | 129, 39.7% | |
| Prior myeloid disease (MDS, MPN, or MDS/MPN overlap) | 85, 22.5% | 6, 11.3% | 79, 24.3 | .036 |
| Prior allo-SCT for MDS and/or MPN | 18, 4.7% | 1, 1.9% | 17, 5.2% | .289 |
| AML type | ||||
| AML-NOS | 291, 76.4% | 45, 84.9% | 244,75.1% | .282 |
| AML-MRC | 71, 18.8% | 6, 11.3% | 65, 20.0% | |
| t-AML | 18, 4.8% | 2, 3.8% | 16, 4.9% | |
| Extramedullary involvement | 13, 3.4% | 2, 3.8% | 11, 3.3% | .250 |
| Cytogenetics | ||||
| CBF | 15, 3.9% | 1, 1.9% | 14, 4.3% | .406 |
| Normal | 127, 33.4% | 31, 58.5% | 96, 29.4% | < .001 |
| Monosomal and/or complex | 105, 27.6% | 4, 7.6% | 99, 30.5% | < .001 |
| Other abnormalities | 91, 23.9% | 11, 20.8% | 80, 24.5% | .557 |
| Missing | 42, 11.1% | 6, 11.3% | 36, 11% | .946 |
| Selected molecular mutations | ||||
| NPM1 | 29, 54.7% | 56, 17.2% | <.001 | |
| IDH1 or IDH2 | 8, 15.9% | 82, 25.2% | .155 | |
| DNMT3A | 16, 30.2% | 80, 24.5% | .48 | |
| ASXL1, BCOR, EZH2, SF3B1, SRSF2, STAG2, U2AF1, ZRSR2 | 18, 34% | 162, 49.7% | .048 | |
| TP53 | 3, 5.7% | 67, 20.6% | .016 | |
| Treatment | .121 | |||
| HMA+ ven. | 110, 29.1% | 12, 22.6% | 98, 30.2% | |
| HMA ± other∗ | 89, 23.5% | 9, 17% | 80, 24.6% | |
| Intensive induction | 179, 47.4% | 32, 60.4% | 147, 45.2% | |
| Intensive therapy specification | ||||
| 7 + 3 | 111, 62% | 14, 43.8% | 97, 66% | |
| 7 + 3 + midostaurin | 20, 11% | 15, 46.9% | 5, 3.4% | |
| 7 + 3 + non-FLT3 inhibitor investigational therapy | 16, 9% | 1, 3.1% | 15, 10.2% | |
| CPX351 | 32, 18% | 2, 6.2% | 30, 20.4% | |
| Allo-SCT after AML diagnosis | 127, 33.6% | 19, 35.9% | 108, 33.2% | .708 |
| . | All, 378 (n %) . | FLT3-ITD+, 53 (n %) . | FLT3-ITD−, 325 (n %) . | P . |
|---|---|---|---|---|
| Age (y, median, range) | 69, (60, 89) | 68 (60, 87) | 70, (60, 89) | .185 |
| Age groups | ||||
| <70 | 189, 50% | 29, 54.7% | 160, 49.2% | .459 |
| ≥70 | 189, 50% | 24, 45.3% | 165, 50.8% | |
| Sex | ||||
| Male | 225, 59.5% | 29, 54.7% | 196, 60.3% | .442 |
| Female | 153, 40.5% | 24, 45.3% | 129, 39.7% | |
| Prior myeloid disease (MDS, MPN, or MDS/MPN overlap) | 85, 22.5% | 6, 11.3% | 79, 24.3 | .036 |
| Prior allo-SCT for MDS and/or MPN | 18, 4.7% | 1, 1.9% | 17, 5.2% | .289 |
| AML type | ||||
| AML-NOS | 291, 76.4% | 45, 84.9% | 244,75.1% | .282 |
| AML-MRC | 71, 18.8% | 6, 11.3% | 65, 20.0% | |
| t-AML | 18, 4.8% | 2, 3.8% | 16, 4.9% | |
| Extramedullary involvement | 13, 3.4% | 2, 3.8% | 11, 3.3% | .250 |
| Cytogenetics | ||||
| CBF | 15, 3.9% | 1, 1.9% | 14, 4.3% | .406 |
| Normal | 127, 33.4% | 31, 58.5% | 96, 29.4% | < .001 |
| Monosomal and/or complex | 105, 27.6% | 4, 7.6% | 99, 30.5% | < .001 |
| Other abnormalities | 91, 23.9% | 11, 20.8% | 80, 24.5% | .557 |
| Missing | 42, 11.1% | 6, 11.3% | 36, 11% | .946 |
| Selected molecular mutations | ||||
| NPM1 | 29, 54.7% | 56, 17.2% | <.001 | |
| IDH1 or IDH2 | 8, 15.9% | 82, 25.2% | .155 | |
| DNMT3A | 16, 30.2% | 80, 24.5% | .48 | |
| ASXL1, BCOR, EZH2, SF3B1, SRSF2, STAG2, U2AF1, ZRSR2 | 18, 34% | 162, 49.7% | .048 | |
| TP53 | 3, 5.7% | 67, 20.6% | .016 | |
| Treatment | .121 | |||
| HMA+ ven. | 110, 29.1% | 12, 22.6% | 98, 30.2% | |
| HMA ± other∗ | 89, 23.5% | 9, 17% | 80, 24.6% | |
| Intensive induction | 179, 47.4% | 32, 60.4% | 147, 45.2% | |
| Intensive therapy specification | ||||
| 7 + 3 | 111, 62% | 14, 43.8% | 97, 66% | |
| 7 + 3 + midostaurin | 20, 11% | 15, 46.9% | 5, 3.4% | |
| 7 + 3 + non-FLT3 inhibitor investigational therapy | 16, 9% | 1, 3.1% | 15, 10.2% | |
| CPX351 | 32, 18% | 2, 6.2% | 30, 20.4% | |
| Allo-SCT after AML diagnosis | 127, 33.6% | 19, 35.9% | 108, 33.2% | .708 |
AMML, acute myelomonocytic/monoblastic leukemia; CNS, central nervous system; CBF, core binding factor; MDS, myelodysplastic syndrome; MPN, myeloproliferative neoplasm; MRC, myelodysplastic related changes; NOS, not otherwise specified; TR, therapy related.
Two patients in the FLT3-ITD+ group received HMA + sorafenib, whereas all other patients with FLT3-ITD+ received HMA monotherapy.
The rates of CR and cCR were similar in patients with FLT3-ITD+ vs FLT3-ITD− treated with IC (69% vs 66%; P = .764 and 72% vs 66%; P = .521, respectively) and with HMA + ven therapy (25% vs 30%; P = .741 and 58% vs 46%; P = .416, respectively). The 30-day and 60-day mortality rates were also comparable in FLT3-ITD+ vs FLT3-ITD− in patients treated with IC (30-day mortality, 0% vs 5% [P = .208]; 60-day mortality, 3% vs 10% [P = .236]) and HMA-based therapy (5% vs 6% [P = .796]; 14% vs 16% [P = .813], respectively). Allo-SCT was performed in 127 patients (33.6%), with similar rates between patients with FLT3-ITD+ and FLT3-ITD− treated with IC (53% vs 58%, respectively; P = .627) or HMA-based therapy (9.5% vs 12.9%; P = .657).
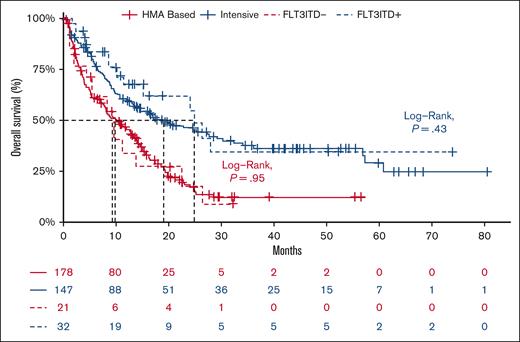
The median OS of the entire cohort was 13.6 months (95% confidence interval [95% CI], 11-16) and did not differ between patients with FLT3-ITD+ and those with FLT3-ITD− (median OS, 15.4 months [95% CI, 11-28] vs 13.1 [95% CI, 11-16]; P = .32). When stratified based on treatment, the median OS was similar between patients with FLT3-ITD+ and those with FLT3-ITD− who received IC at 24.8 months (95% CI, 15 to not available [NA]) vs at 19.1 months (95% CI, 13-30; P = .43) and HMA-based therapy at 9.3 months (95% CI, 6 to NA) vs at 9.9 months (95% CI, 8-14; P = .95; Figure 1), including in the subgroup treated with HMA + ven (FLT3-ITD+ median OS, 11.2 months [95% CI, 9 to NA] vs FLT3-ITD−, 12.5 months [95% CI, 8-16]; P = .92). The similar OS between patients with FLT3-ITD+ and FLT3-ITD− remained consistent in sensitivity analyses in which patients with TP53 mutant were excluded in both the IC group (median OS, 24.8 months [95% CI, 15-NA] vs 20.0 months [95% CI, 14-57]; P = .6) and HMA-based group (median OS, 9.7 months [95% CI, 6-NA] vs 12.5 months [95% CI, 8-15]; P = .8), respectively (supplemental Figure 2).
Overall survival between FLT3-ITD+ AML and FLT3-ITD–AML stratified based on treatment-intensive chemotherapy (blue) or HMA-based therapy (red).
Overall survival between FLT3-ITD+ AML and FLT3-ITD–AML stratified based on treatment-intensive chemotherapy (blue) or HMA-based therapy (red).
In a multivariable OS analysis in patients aged ≤75 years who were treated with IC (n = 158), the FLT3-ITD status did not affect the OS (hazard ratio [HR], 0.98; 95% CI, 0.51-1.88; P = .958). Consolidation with allo-SCT was associated with an improved OS (HR, 0.39; 95% CI, 0.27-0.56; P < .001), whereas a monosomal and/or complex karyotype (HR, 2.20; 95% CI, 1.3-3.71; P = .003), prior myeloid neoplasm (HR, 1.94; 95% CI, 1.24-3.02; P = .003), and TP53 mutation (HR, 2.1; CI 95%, 1.14-3.86; P = .017) were associated with a worse OS (supplemental Table 1). In a multivariable analysis of patients aged ≤75 years who were treated with HMA-based therapy (n = 93), the presence of FTL3-ITD mutation was not associated with changes in the OS (HR, 1.16; 95% CI, 0.49-2.76; P = .738). Allo-SCT was associated with improved OS (HR, 0.15; 95% CI, 0.06-0.35; P < 0. 001) whereas TP53 mutation was associated with worse survival (HR, 2.75; 95% CI, 1.1-6.88; P = .03; supplemental Table 2). In a multivariable subgroup analysis in patients with FLT3-ITD+ aged ≤75 years (n = 43), an allo-SCT was associated with improved survival (HR, 0.31; 95% CI, 0.14-0.70; P = .005), whereas a monosomal and/or complex karyotype was associated with worse survival (HR, 2.5; 95% CI, 1.09-5.74; P = .031).
As FLT3-ITD mutations usually occur in de novo AML,8,9 the low FLT3-ITD+ rate (14%) seen in our study is partly due to older age of patients, associated with higher rates of prior myeloid malignancy, chemotherapy exposure, and high-risk genetic features10,11 Although Papaemmanuil observed that 22% of patients with AML harbored an FLT3-ITD mutation, only 11% were aged >60 years1; in a post hoc analysis of FLT3 significance in the VIALE-A cohort, the incidence was similar (11%) to that in our cohort.12 In addition, the cCR rate of 47% observed in our cohort of patients who were treated with HMA+ ven reflects the aforementioned high-risk features as well as the real-world nature of our population, consistent with similar studies.13,14
The absence of a FLT3-ITD prognostic effect may be because of the higher incidence of adverse prognostic factors in our older FTL3-ITD− cohort and/or nonhematologic comorbidities in older adults, which may be more relevant than FLT3-ITD.15,16 Furthermore, the use of FLT3 inhibitor therapy might have improved the outcome in the FLT3-ITD+ group treated with IC, which was historically worse in the premidostaurin era according to some17 but not other studies.18,19 In a pooled analysis from the VIALE-A and phase 1b studies of patients treated with HMA + ven, FLT3-ITD was associated with a modest benefit from ven in combination with HMA (median OS,12.1 months).20 Another post hoc analysis of this population showed high cCR (63.3%) rates in patients with FLT3-ITD+, but comparable OS was observed between patients with FLT3-mutated and those with FLT3-WT (median OS, 12.5 months vs 14.7 months).12
The prognostic value of allo-SCT was consistent in both the IC and HMA-based therapy groups as well as in the subset of patients with FLT3-ITD+ AML. Our finding regarding the beneficial effect of allo-SCT is comparable with that in previously reported studies21 and similar to the effect in younger patients enrolled in the RATIFY trial.3,22
Our study has several limitations, such as the retrospective nature of our cohort that may lead to selection bias, and significant patient heterogeneity that exists in real-world analyses. Each of these limitations were addressed via subgroup and regression analyses, although small patient numbers in subsets was partially limiting.
In summary, our findings suggests that among older patients, the presence of FLT3-ITD+ may not be of major prognostic value and that allo-SCT should be pursued, when possible, irrespective of initial therapy in older patients with AML, including in those with FLT3-ITD+.
Acknowledgments: This work was supported by the Dana-Farber Cancer Institute Hematologic Malignancy Data Repository and financially supported by the Lunder Leukemia Research Fund.
Contribution: S.S., R.C.L., and R.M.S. designed the research; S.S., E.C.C., and H.K.T. performed data extraction; S.S., G.F., D.S.N., and H.K.T. analyzed the data; S.S. and R.M.S. wrote the initial draft of the manuscript; M.W., E.S.W., J.S.G., M.R.L., D.S.N., D.J.D., and R.C.L. reviewed the manuscript and contributed to its final version; and all authors reviewed the final version of the manuscript and agreed for submission.
Conflict-of-interest disclosure: E.S.W. consults for Curis, Pfizer, and Takeda. J.S.G. consults and serves on the scientific ad boards for AbbVie, Genentech, Astellas, Servier, and Bristol Myers Squibb (BMS), and receives institutional funds from Prelude, Pfizer, Genentech, and AbbVie. M.R.L. reports research funding from Novartis and AbbVie, and has served on the advisory board of Pfizer. M.S. consulted for Boston Consulting; served on the advisory board for Novartis, Kymera, Sierra Oncology, and GlaxoSmithKline (GSK); and participated in graduate medical education activity for Novartis, Curis Oncology, Haymarket Media, and Clinical Care Options. D.S.N. has stock ownership in Madrigal Pharmaceuticals. D.J.D. has served as a consultant for Amgen, Autolos, Agios, Blueprint, Forty-Seven, Gilead, Incyte, Jazz, Novartis, Pfizer, Servier, and Takeda, and has received research funding from AbbVie, Glycomimetics, Novartis, and Blueprint Pharmaceuticals. R.C.L. reports consulting fees from bluebird bio, Takeda Pharmaceuticals, Qiagen, Nuprobe, Jazz Pharmaceuticals, and Thermo Fisher. R.M.S. reports consulting fees from AbbVie, AbbVie/Genetech, Actinium, Amgen, Aptevo, Aprea, Arog, AvenCell, BergenBio, BMS, Boston Pharmaceuticals, Cellularity, CTI Pharma, Epizyme, Foghorn Therapeutics, Gemoab, GSK, Innate, Janssen, Jazz, Kura Oncology, Novartis, Onconova, Rigel, Syntrix, Syros, and Takeda. The remaining authors declare no competing financial interests.
Correspondence: Richard M. Stone, Dana-Farber Cancer Institute, 450 Brookline Ave, Boston, MA 02215; e-mail: richard_stone@dfci.harvard.edu.
References
Author notes
The data that support the findings of this study are available on request from the corresponding author, Richard M. Stone (richard_stone@dfci.harvard.edu).
The data are not publicly available because of privacy or ethical restrictions.
The full-text version of this article contains a data supplement.