TO THE EDITOR:
Mutations in the WT1 transcription factor (WT1) gene are found in ∼5% to 12% of adults with acute myeloid leukemia (AML) and are generally associated with a poor prognosis.1-8 Several studies have also reported frequent co-occurrence of FLT3-ITD in patients with mutated WT1, ranging from 23% to 75%.1,6,9,10 In some,1,10 but not all,5-7 analyses, patients with both WT1 mutations and FLT3-ITD had lower complete remission rates, higher relapse rates, and inferior overall survival than patients with mutated WT1, but no FLT3-ITD.
Although WT1 mutations are common in adult AML, there are limited data describing whether they are initiating or late mutations in AML pathogenesis. Furthermore, the clonal architecture and complexity of this AML subtype are not well described. The utility of single-cell DNA sequencing has provided important biologic insights into other subtypes of AML, particularly with respect to drug resistance.11,12 In this study, we performed bulk targeted DNA sequencing of 96 newly diagnosed patients with AML with WT1 mutations, treated in Cancer and Leukemia Group B (CALGB) therapeutic trials, followed by single-cell DNA sequencing in a subset of patients to provide higher resolution and insight into both the initiating and clonal framework of mutations co-occurring with WT1. CALGB is now part of the Alliance for Clinical Trials in Oncology (Alliance).
We examined 96 adult patients with de novo AML and WT1 mutations, treated in CALGB/Alliance trials, for whom tissue bank material (pretreatment marrow and blood samples) was available and underwent next-generation sequencing at The Ohio State University, as previously described.13 Patients with acute promyelocytic leukemia and those who died within 30 days of treatment were excluded from analysis. The patients provided written informed consent to participate in the treatment trials and companion protocols CALGB 8461 (cytogenetic studies; www.clinicaltrials.gov as #NCT00048958), CALGB 9665 (leukemia tissue bank; www.clinicaltrials.gov as #NCT00899223), and/or CALGB 20202 (molecular studies; www.clinicaltrials.gov as #NCT00900224). The treatment protocols were in accordance with the Declaration of Helsinki and approved by the institutional review boards at each center.
For next-generation sequencing, mononuclear cells were enriched by Ficoll-Hypaque gradient centrifugation and cryopreserved until use. For bulk targeted sequencing, genomic DNA was extracted using the DNeasy Blood and Tissue Kit (QIAGEN, Hilden, Germany). The mutational status of 81 protein-coding genes was determined by targeted amplicon sequencing using the MiSeq platform (Illumina, San Diego, CA). Sequenced reads were aligned to the hg19 genome and analyzed as previously described.14 A variant allele fraction (VAF) cutoff of 0.10 was used to define the WT1 mutated data set. A WT1 variant of .30 or greater was defined as a dominant mutation based on the Beat AML trial definition that has been previously reported.15 A VAF cutoff of 0.02 was set for reporting co-occurring mutations; low VAF variants frequently mutated in AML were manually reviewed for inclusion. Single nucleotide polymorphisms with no known pathogenic associations were not reported as mutations. Visual inspection of all variants was performed using the Integrative Genomics Viewer version 2.8 (Broad Institute).
For single-cell DNA sequencing, the MissionBio (San Francisco, CA) AML panel was utilized. Total cells were diluted to a concentration of 3500 cells per μL in Cell Buffer and the Tapestri single-cell DNA sequencing user-guide was followed. The final normalized libraries were sequenced using 2 × 300 bp paired-end multiplexed runs on the Illumina MiSeq platform. Tapestri analysis pipeline was utilized to process and analyze the sequencing fastq files to generate .loom files. LOOM files were then visualized in Tapestri Insight to call variants and visualize clones.
For statistical analysis, baseline characteristics were compared between the groups of patients with AML using Fisher exact test for categorical variables and the Wilcoxon rank-sum test for continuous variables.16 For time-to-event analyses, we calculated survival estimates using the Kaplan-Meier method and compared the groups using the log-rank test.16 Statistical analyses were performed using the Alliance Statistics and Data Center. The results analyzed are available in the database as of 20 July 2022. The median follow-up for patients still alive was 7.6 years.
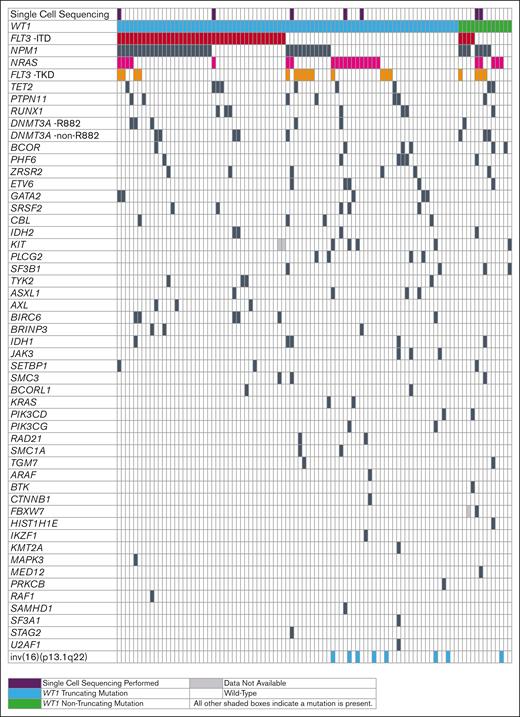
Of the 1675 adults with de novo AML sequenced, 96 (5.7%) were identified to have WT1 mutations. Most of the WT1 mutations were localized in exons 7 and 9 (WT1 isoform D: NM_024426.6) and 83 (86.5%) patients had truncating mutations. There was a wide range of VAFs for WT1 mutations among patients, ranging from 0.109 to 0.843, with 63 (72%) patients having a VAF >0.30. The baseline clinical characteristics of patients are listed in supplemental Table 1. Seventy-nine (82%) patients with WT1-mutated AML, were aged <60 years. Consistent with previous reports,1,6,9,10 the most common co-occurring mutation was FLT3-ITD (Figure 1), which was detected in 47% (45/96) of the patients with mutated WT1 (WT1mut/FLT3-ITD). There were few baseline clinical differences between patients with WT1mut/FLT3-ITD and those with mutated WT1 without FLT3-ITD (WT1mut/no FLT3-ITD; supplemental Table 1). However, patients with WT1mut/FLT3-ITD were more commonly classified into the intermediate-risk group (80% vs 18%) according to the 2022 European LeukemiaNet criteria,17 with only 5% having a favorable-risk disease, as opposed to 58% of patients with WT1mut/no FLT3-ITD (P < .001). Compared with patients with AML with WT1mut/no FLT3-ITD, those with WT1mut/FLT3-ITD had inferior disease-free survival (median, 0.6 vs 1.2 years; P < .001), overall survival (median, 0.7 vs 1.5 years; P < .001) and event-free survival (0.2 vs 0.8 years; P < .001), but no significant difference in complete remission rates (53% vs 69%; P = .14) (supplemental Figure 1; supplemental Table 2).
Oncoprint of co-occurring mutations observed in WT1-mutated adult patients with de novo AML. Patients in whom single-cell DNA sequencing was performed are marked in purple. Patients with truncating WT1 mutations are shown in blue and patients with nontruncating mutations are shown in green. Patients carrying FLT3-ITD are marked in red, those with NRAS mutations magenta, those with FLT3-TKD orange, and those with mutations in the remaining genes in black. White denotes wild-type genes or the absence of FLT3-ITD or FLT3-TKD; light gray indicates unknown mutational gene status.
Oncoprint of co-occurring mutations observed in WT1-mutated adult patients with de novo AML. Patients in whom single-cell DNA sequencing was performed are marked in purple. Patients with truncating WT1 mutations are shown in blue and patients with nontruncating mutations are shown in green. Patients carrying FLT3-ITD are marked in red, those with NRAS mutations magenta, those with FLT3-TKD orange, and those with mutations in the remaining genes in black. White denotes wild-type genes or the absence of FLT3-ITD or FLT3-TKD; light gray indicates unknown mutational gene status.
Next, we analyzed the co-occurring mutations in patients with WT1mut/FLT3I-ITD and those with WT1mut/no FLT3-ITD (Figure 1). Mutations in NPM1 were the most common, occurring in 58% of patients with WT1mut/FLT3-ITD (N = 26/45). Notably, diminished signaling pathways activate mutations in RAS and FLT3-TKD. In contrast, patients with WT1mut/no FLT3-ITD had significantly more frequent mutations in NRAS (37% vs 11%; P = .004) and FLT3-TKD (25% vs 11%; P = .11) compared with patients with WT1mut/FLT3-ITD, and fewer mutations in NPM1 (29% vs 58%; P = .007).
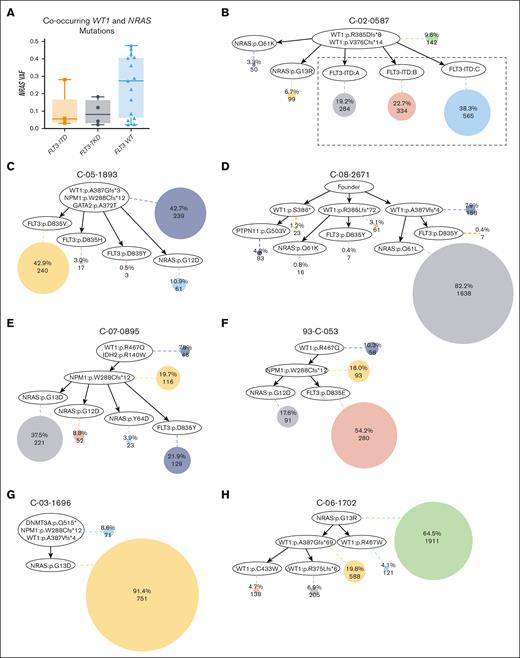
Given the difference in NRAS mutation frequencies between patients with WT1mut/FLT3-ITD and WT1mut/no FLT3-ITD, we compared the VAF of NRAS mutations in the presence and absence of FLT3 mutations. There was a trend for a higher VAF of NRAS mutations in patients with mutated WT1 with concurrent NRAS mutations but no FLT3 mutations (Figure 2A), suggesting that this may be due to WT1 being the founding mutation with 2 or more distinct subclones. To further delineate this observation, we performed single-cell DNA sequencing on 7 samples with co-occurring WT1 and NRAS mutations, with or without FLT3 mutations (Figures 2B-H). Mean WT1 VAF was comparable in the 7 samples with single-cell DNA sequencing and the entire data set of 96 samples with bulk DNA sequencing (0.38 vs 0.36). WT1 mutations were observed in the founder clones of 6 of the 7 samples. In 1 sample, an NRAS mutation was observed in a prominent founder clone, representing 64.5% of all leukemia cells (Figure 2H). In all other cases, FLT3 (ITD or TKD) or NRAS mutations arose in mutually exclusive subclones from a WT1 mutated founder clone; 3 samples had a greater percentage of cells in subclones with FLT3 mutations than NRAS mutations (mean, 60.0% vs 12.3%) (Figure 2B-C,F), whereas 2 samples had a greater percentage of cells with NRAS mutations (mean, 66.5% vs 11.5%) (Figure 2D-E). In 4 samples with co-occurring NPM1 mutations, NPM1 cooccurred with WT1 in the founder clone in 2 samples (Figure 2C,G) or was acquired before FLT3-TKD or NRAS mutations in the other 2 (Figure 2E-F).
Single-cell DNA sequencing of samples with co-occurring WT1 and NRAS mutations. (A) Box plot of the NRAS VAF in WT1 and NRAS comutated patients with FLT3-ITD, FLT3-TKD, and patients with FLT3 wild-type. (B-H) Inferred clone relationships derived from tapestri single-cell DNA sequencing. Each oval represents a mutational event. Each shaded circle represents a subclone with accumulated mutational events along its path from the top of the tree. The size of each shaded circle is proportional to the size of the clone population. The first number within each colored circle is the percentage of each clone among the total number of tumor cells included in the single-cell data analysis for that sample and the second number is the absolute number of cells included in that subclone. (B) FLT3-ITD and NRAS mutations arose in mutually exclusive subclones of a WT1 mutated founder clone. The 3 detected FLT3-ITDs may represent a single subclone because of difficulties in mapping the FLT3-ITD reads. A TET2 mutation observed by bulk sequencing was not detected by single-cell sequencing. (C-F) FLT3-TKD and RAS pathway mutations arise in mutually exclusive subclones from a WT1 mutated founder clone. Sample C-05-1893: FLT3-ITD mutation was observed by bulk sequencing but was not detected by single-cell sequencing; sample C-08-2671: FLT3-TKD and PTPN11 mutations were not detected by bulk sequencing. (G-H) Patients with non-FLT3-mutation. In 1 sample, the prominent subclone harbors an NRAS mutation; in the other sample, NRAS was in the prominent founder clone, followed by the accumulation of WT1 mutations in the subclones. Sample C-03-1696: NPM1 was detected with low coverage and DNMT3A mutations were not covered by bulk sequencing.
Single-cell DNA sequencing of samples with co-occurring WT1 and NRAS mutations. (A) Box plot of the NRAS VAF in WT1 and NRAS comutated patients with FLT3-ITD, FLT3-TKD, and patients with FLT3 wild-type. (B-H) Inferred clone relationships derived from tapestri single-cell DNA sequencing. Each oval represents a mutational event. Each shaded circle represents a subclone with accumulated mutational events along its path from the top of the tree. The size of each shaded circle is proportional to the size of the clone population. The first number within each colored circle is the percentage of each clone among the total number of tumor cells included in the single-cell data analysis for that sample and the second number is the absolute number of cells included in that subclone. (B) FLT3-ITD and NRAS mutations arose in mutually exclusive subclones of a WT1 mutated founder clone. The 3 detected FLT3-ITDs may represent a single subclone because of difficulties in mapping the FLT3-ITD reads. A TET2 mutation observed by bulk sequencing was not detected by single-cell sequencing. (C-F) FLT3-TKD and RAS pathway mutations arise in mutually exclusive subclones from a WT1 mutated founder clone. Sample C-05-1893: FLT3-ITD mutation was observed by bulk sequencing but was not detected by single-cell sequencing; sample C-08-2671: FLT3-TKD and PTPN11 mutations were not detected by bulk sequencing. (G-H) Patients with non-FLT3-mutation. In 1 sample, the prominent subclone harbors an NRAS mutation; in the other sample, NRAS was in the prominent founder clone, followed by the accumulation of WT1 mutations in the subclones. Sample C-03-1696: NPM1 was detected with low coverage and DNMT3A mutations were not covered by bulk sequencing.
To our knowledge, these data represent the largest series of adults with WT1-mutated AML, and, for the first time, we describe clonal architecture and the combination of mutations co-occurring in single cells. This study did not use oligonucleotide-conjugated antibody technology because it was unavailable at the time of this analysis. Future studies should focus on the application of this added feature to confirm that WT1 mutations do not exist in lymphoid cells as well. Based on the current data, we can conclude that WT1 loss-of-function mutations can serve as an initiating gene for development of AML. Our data provide a deeper understanding of the genomic complexity and biology of WT1-mutated AML and may help in the future development of novel therapeutic strategies.
Acknowledgments: The authors are grateful to the patients who consented to participate in the clinical trials and the families who supported them, Donna Bucci and the CALGB/Alliance Leukemia Tissue Bank at The Ohio State University Comprehensive Cancer Center, Columbus, Ohio, for sample processing and storage services, and Lisa J. Sterling for data management.
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award numbers U10CA180821, U10CA180882, and U24CA196171, (to the Alliance for Clinical Trials in Oncology), UG1CA189824, UG1CA233327, UG1CA233331, UG1CA233338, UG1CA233339, P30CA016058, and R35 CA198183 (J.C.B.), the Four Winds Foundation, Harry F. Mangurian Foundation, the Coleman Leukemia Research Foundation (J.C.B.), and The Ohio State University Comprehensive Cancer Center Pelotonia Foundation (A.-K.E., J.S.B., S.D.B., and J.C.B.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health (https://acknowledgments.alliancefound.org).
Contribution: B.B., J.K., S.J.O., D.R.B., D.N., K.M., A.-K.E., S.D.B., and J.C.B contributed to the design and analysis of the study and the writing of the manuscript; S.J.O., D.R.B., S.F., C.C.O., and E.K.H. performed laboratory-based research; J.S.B. performed data processing; J.K. and D.N. performed statistical analysis; J.E.K., G.U., W.S., B.L.P., and J.C.B. were involved directly or indirectly in the care of patients and/or sample procurement; and all authors read and agreed to the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Bhavana Bhatnagar, West Virginia University Cancer Institute, Wheeling Hospital, 1 Medical Park, Wheeling, WV 26003; e-mail: bhavana.bhatnagar1@hsc.wvu.edu; and John C. Byrd, University of Cincinnati College of Medicine, Room 6065, Medical Science Building, 231 Albert Sabin Way, Cincinnati, OH 45267-0551; e-mail: byrd2jc@ucmail.uc.edu.
References
Author notes
∗S.D.B. and J.C.B. contributed equally to this study.
The data will be made available through the Alliance for Clinical Trials in Oncology and on request from the corresponding author, Bhavana Bhatnagar (bhavana.bhatnagar1@hsc.wvu.edu).
The full-text version of this article contains a data supplement.