TO THE EDITOR:
Systemic mastocytosis (SM), first reported in 19491 and defined by the World Health Organization (WHO) classification,2 can be challenging to diagnose. Difficulties arise because the fraction of neoplastic mast cells (MCs) and other hematopoietic cells carrying the acquired activating pathogenic variant KIT p.D816V can be low, necessitating the use of sensitive assays for detection of both KIT p.D816V and aberrant expression of CD25 on neoplastic MCs using flow cytometry (FC) and immunohistochemistry (IHC).3-8 Testing methodology has been improved over time, but challenges remain. We present 15 cases in which resolution of discrepant laboratory results was needed to establish a diagnosis.
We retrospectively identified patients from a MC registry9 who underwent evaluation for SM using 2017 WHO criteria2 and tryptase genotyping for hereditary-α tryptasemia (HαT)10 and met at least 1 of following 4 criteria: (1) ≥2 methods of KIT p.D816V testing had discordant results, (2) basal serum tryptase (BST) values were variably >20 ng/mL, (3) there were discordant results of MC CD25 expression assessed using FC and IHC or among clinical laboratories for IHC alone, or (4) there were <25% spindled MCs or <2 MC aggregates in patients meeting other diagnostic criteria. The protocol submitted to the Walter Reed National Military Medical Center Institutional Review Board was determined to be exempted. The study was conducted according to the declaration of Helsinki. See supplemental Methods for more details.
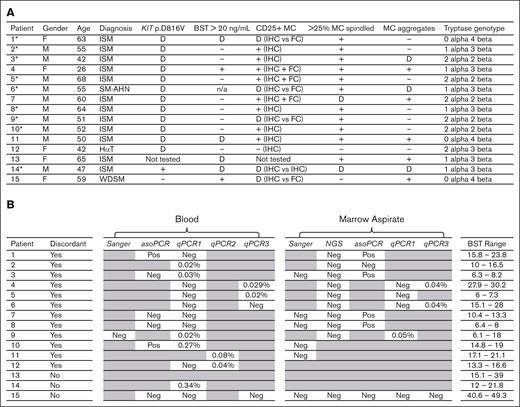
Fifteen of 183 patients from the MC registry had discordant results in ≥1WHO criteria. Five patients were women and 10 were men. Ages ranged from 26 to 68 years. WHO diagnostic results and tryptase genotypes are shown in Figure 1A. Twelve patients had indolent SM, 1 had SM with an associated hematologic neoplasm (SM-AHN polycythemia vera), 1 had HαT, and 1 had well-differentiated SM (WDSM). Of 15 patients, 12 had discordant KIT p.D816V results, 4 had discordant BST results comparing timepoints, 5 had discordant CD25 MC expression comparing IHC to FC or IHC between laboratories, 2 had discordance when assessing MC spindling, and 3 had discordance when assessing MC aggregates. Nine of the 15 patients required repeat testing and a second hematopathology evaluation to be diagnosed with SM.
(A) List of patients showing gender, age, final diagnosis, SM test results, and tryptase genotyping. An asterisk next to a patient number indicates where a discordant result affected the diagnosis. A dash (−) indicates a negative result. A plus (+) refers to a positive result. A (D) indicates a discordant result. Specifically, a discordant KIT p.D816V result occurred when 2 different tests had different results (see Figure 1B). A discordant BST result occurred when at least one timepoint showed a BST ≥20 ng/mL and other timepoints showed BST <20 ng/mL (see Figure 2A). A discordant CD25 result occurred when FC showed CD25 positive MCs but IHC did not; or when IHC initially did not show CD25 expression in 1 clinical lab but did on reanalysis by hematopathologists I.M. and E.P. (see Figure 2B). A discordant result for spindling and aggregates occurred when these criteria were documented in a pathology report as not being met by 1 clinical lab and then as met on reanalysis by authors I.M. and E.P. All 14 patients diagnosed with SM had a normal tryptase genotype. (B) Comparison of 4 PCR-based and 2 sequencing-based clinical methods to detect KIT p.D816V in blood and marrow aspirate. Sanger sequencing and NGS are sequence-based methods. PCR-based assays include asoPCR, qPCR1, qPCR2, and qPCR3. qPCR1-3 correspond to 3 different clinical labs for qPCR. For asoPCR, a “Pos” indicates a positive result in which the mutation was detected. For all methods, a “Neg” indicates a negative result in which the mutation was not detected. The variant allele frequencys are shown for qPCR1-3 methods. Patient 13 had no mutational testing performed and patient 14 had only 1 method performed. BST ranges are shown in ng/mL. Greyed-out cells indicate testing was not performed.
(A) List of patients showing gender, age, final diagnosis, SM test results, and tryptase genotyping. An asterisk next to a patient number indicates where a discordant result affected the diagnosis. A dash (−) indicates a negative result. A plus (+) refers to a positive result. A (D) indicates a discordant result. Specifically, a discordant KIT p.D816V result occurred when 2 different tests had different results (see Figure 1B). A discordant BST result occurred when at least one timepoint showed a BST ≥20 ng/mL and other timepoints showed BST <20 ng/mL (see Figure 2A). A discordant CD25 result occurred when FC showed CD25 positive MCs but IHC did not; or when IHC initially did not show CD25 expression in 1 clinical lab but did on reanalysis by hematopathologists I.M. and E.P. (see Figure 2B). A discordant result for spindling and aggregates occurred when these criteria were documented in a pathology report as not being met by 1 clinical lab and then as met on reanalysis by authors I.M. and E.P. All 14 patients diagnosed with SM had a normal tryptase genotype. (B) Comparison of 4 PCR-based and 2 sequencing-based clinical methods to detect KIT p.D816V in blood and marrow aspirate. Sanger sequencing and NGS are sequence-based methods. PCR-based assays include asoPCR, qPCR1, qPCR2, and qPCR3. qPCR1-3 correspond to 3 different clinical labs for qPCR. For asoPCR, a “Pos” indicates a positive result in which the mutation was detected. For all methods, a “Neg” indicates a negative result in which the mutation was not detected. The variant allele frequencys are shown for qPCR1-3 methods. Patient 13 had no mutational testing performed and patient 14 had only 1 method performed. BST ranges are shown in ng/mL. Greyed-out cells indicate testing was not performed.
Thirteen of 15 patients were positive for the KIT p.D816V mutation; 12 of the 13 patients had discordance among different tests (Figure 1B). Sequencing methods including Sanger sequencing in 7 patients and next generation sequencing (NGS) in 10 patients did not detect KIT p.D816V. In contrast, polymerase chain reaction (PCR) methods including qualitative allele–specific oligonucleotide PCR (asoPCR) from one laboratory and quantitative PCR (qPCR) from 3 additional laboratories (qPCR1, qPCR2, and qPCR3) detected KIT p.D816V but 7 patients had discordance among PCR results performed using different methods. Six of 7 patients with discordant PCR results had SM with a variant allele frequency near the lower limit of detection. Patient 1 had KIT p.D816V detected using asoPCR of blood and marrow samples but not using qPCR1 of blood sample 134 days later, patient 2 had KIT p.D816V detected using qPCR1 of blood sample 392 days after testing negative via asoPCR of marrow sample, patient 3 had KIT p.D816V detected using qPCR1 of blood sample 576 days after testing negative via asoPCR of blood sample, patient 4 had KIT p.D816V detected using qPCR3 but not using qPCR1 of blood or marrow samples from the samples collected on the same day, patient 5 had KIT p.D816V detected via qPCR3 of blood sample but not via qPCR1 of blood or marrow samples from the samples collected on the same day, and patient 6 had KIT p.D816V detected using qPCR3 but not using qPCR1 of blood or marrow samples from the samples collected on the same day. There was no evidence of SM disease progression or remission in any patient which might account for the discordant test results. These results demonstrate challenges in detecting KIT p.D816V when the fraction of cells with the mutation approaches the assay sensitivity limit. Notably, patient 12 failed to meet the 2017 WHO criteria for SM when reviewed by hematopathologists I.M. and E.P. Patient 12 also had no history of anaphylaxis and had a diagnosis of HαT. This patient had KIT p.D816V originally detected using qPCR2 but not via repeat qPCR1 of blood sample 58 days later, suggesting a false positive original qPCR2 result.
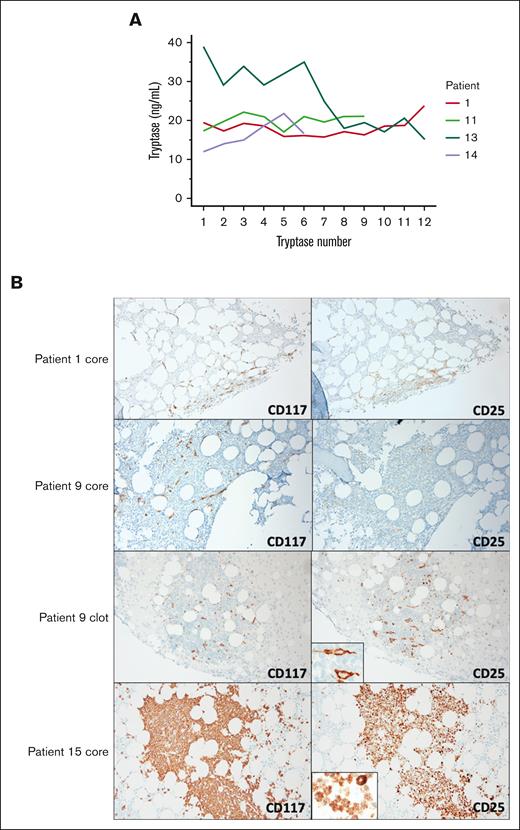
Eight patients had BST values consistently <20 ng/mL and 2 patients had values consistently >20 ng/mL. Five patients had BST values that over time were neither consistently above nor below 20 ng/mL (Figure 2A). A tryptase level >20 ng/mL did not contribute to the diagnosis of SM in any of the 14 patients because all met other minor WHO diagnostic criteria regardless of their BST values. These data support prior reports that sampling multiple timepoints may be needed to assess the dynamic BST range.9,11 However, in all 14 SM cases, BST did not provide value within the diagnostic framework.
(A) BST values over time in 4 patients with indolent SM. Patients 1, 11, 13, and 14 are shown in whom some BST values are above and others fall below 20 ng/mL. Patient 6 is not shown because tryptase is not a WHO minor criterion in those diagnosed with SM-AHN. There was no acute allergic reaction, renal injury, or cytoreductive treatment used at the time of BST measurement at any timepoint. (B) CD117 and CD25 IHC are shown for patients 1 and 9 with indolent SM as well as patient 15 with WDSM. Original CD25 IHC core biopsy slides for patients 1 and 9 were negative (not shown). Patients 1 had new core slides cut and stained for CD117 and CD25 IHC at a second pathology lab and results showed colocalization of CD25 expression with CD117-positive cells. Patient 6 (not shown) had results similar to that of patient 1. Patient 9 had new core and clot slides cut and stained for CD117 and CD25 IHC at a second pathology lab and results showed colocalization of CD25 expression with CD117-positive cells (see insert) in the clot but not the core. The discordance between the core and the clot CD25 IHC for patient 9 suggested that the core biopsy decalcification procedure contributed to a false-negative result. Patient 15 with WDSM had large aggregates of round MCs. CD25 colocalized to CD117-positive cells in a cytoplasmic rather than plasma membrane distribution (see insert).
(A) BST values over time in 4 patients with indolent SM. Patients 1, 11, 13, and 14 are shown in whom some BST values are above and others fall below 20 ng/mL. Patient 6 is not shown because tryptase is not a WHO minor criterion in those diagnosed with SM-AHN. There was no acute allergic reaction, renal injury, or cytoreductive treatment used at the time of BST measurement at any timepoint. (B) CD117 and CD25 IHC are shown for patients 1 and 9 with indolent SM as well as patient 15 with WDSM. Original CD25 IHC core biopsy slides for patients 1 and 9 were negative (not shown). Patients 1 had new core slides cut and stained for CD117 and CD25 IHC at a second pathology lab and results showed colocalization of CD25 expression with CD117-positive cells. Patient 6 (not shown) had results similar to that of patient 1. Patient 9 had new core and clot slides cut and stained for CD117 and CD25 IHC at a second pathology lab and results showed colocalization of CD25 expression with CD117-positive cells (see insert) in the clot but not the core. The discordance between the core and the clot CD25 IHC for patient 9 suggested that the core biopsy decalcification procedure contributed to a false-negative result. Patient 15 with WDSM had large aggregates of round MCs. CD25 colocalized to CD117-positive cells in a cytoplasmic rather than plasma membrane distribution (see insert).
Patients 1, 6, 9, 14, and 15 had discordant results for MC CD25 immunostaining when compared among different laboratories (Figures 1A and 2B). Tissue was reanalyzed by hematopathologists I.M. and E.P. Patients 1, 6, 9, and 14 had detectable KIT p.D816V, but negative CD25 MC expression, raising concern for errors based on reported concordance of KIT p.D816V with positive MC CD25 expression.12 In patients 1, 6, and 9, FC showed CD25 expression, but IHC did not. In patients 1 and 6, CD25 IHC on the core was positive on restaining in a different laboratory. In patient 9, CD25 IHC on the marrow clot but not on biopsy core was positive on restaining suggesting that the decalcification procedure contributed to a false-negative result. CD25 IHC was positive in the original and restained slides for patient 14. Finally, in patient 15 diagnosed with KIT p.D816V-negative WDSM, FC did not show surface MC CD25 expression but CD25 was positive as shown using IHC in a cytoplasmic distribution. Overall, KIT p.D816V-positive SM with negative CD25 results were shown to be erroneous, and true FC and IHC discordance in WDSM could be explained by subcellular distribution.
Pathology reports of patients 7, 14, and 15 noted absence of MC spindling. Patient 15 had WDSM with round, mature appearing MCs. After second hematopathology re-examination, patients 7 and 14 showed >25% spindled MCs in both original and recut marrow sections. In addition, 3 of 10 patients without originally reported MC aggregates were found to have them on re-examination.
We presented several pitfalls in SM WHO criteria interpretation. First, sensitivity and specificity affect detection of KIT p.D816V. We found that NGS and Sanger sequencing lacked sensitivity compared with PCR methods in detection of KIT p.D816V3,13,14 in the cases of SM we presented. Relative to this issue, the recently published International Consensus Classification of myeloid neoplasms and acute leukemias revised SM criteria recommends high-sensitivity PCR testing to avoid false-negative results.15 Considering PCR, we further found 6 patients with SM with discordant PCR results with variant allele frequencies near the lower limit of detection. Therefore, these findings support this need for high-sensitivity PCR for improvement in KIT p.D816V detection to both aid in diagnosis and predict treatment responses to KIT p.D816V selective tyrosine kinase inhibitors.16 Until new methods are developed and available, multiple timepoints and qPCR methods should be assessed in blood and marrow samples if the initial measurement is negative and there is a high pretest probability of disease.9,17,18 Interestingly, one patient with no history of anaphylaxis who tested positive for HαT with BST values <20 ng/mL, and no histopathological evidence of SM, tested positive for KIT p.D816V by 1 qPCR test and negative by another. This may well be the first false positive result reported for any KIT p.D816V PCR-based assay and shows that KIT p.D816V positive cases should always be re-evaluated for the presence of other WHO minor criteria, such as CD25 MC expression and spindling.12 Second, we illustrate that BST variability9,11 can affect whether the minor criterion is met. However, BST values added no value to SM diagnostic criteria in cases with detectable KIT p.D816V because SM occurred at BST values as low as 6 ng/mL when other minor criteria were present (MC CD25 expression and spindling).12 Third, we showed that preanalytical IHC errors accounted for 3 cases of CD25 discordance and analytical errors accounted for 1 case of CD25, 2 cases of MC spindling, and 3 cases of MC aggregate discordance, supporting the benefit of centralized expert bone marrow sample evaluation.19 We also showed true discordance between FC and IHC in a patient with WDSM reflecting CD25 cellular localization. In summary, SM evaluations would benefit from improved and standardized KIT p.D816V assays, potential elimination of BST from SM diagnostic criteria, and consultations at a reference center where there is hematopathology expertise.
Acknowledgment: This research was funded in whole or in part by the Division of Intramural Research of the National Institute of Allergy and Infectious Diseases, National Institutes of Health. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, Uniformed Services University, or Department of Defense; nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
Contribution: N.B. and I.M. designed the study. Hematopathologists I.M. and E.P. evaluated bone marrow biopsy tissue; N.B., I.M., X.S., J.M., J.L., E.P., and D.R. generated the study data; N.B. wrote the manuscript with input from I.M., D.M., X.S., J.M., J.L., J.M., E.P., D.R.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Nathan A. Boggs, WRNMMC, 4954 North Palmer Rd, Building 19, Room 4112, Bethesda, MD 20889; e-mail: nathan.boggs@usuhs.edu.
References
Author notes
For data sharing, contact the corresponding author: nathan.boggs@usuhs.edu
The full-text version of this article contains a data supplement.