Key Points
SSPE in cancer patients is associated with a high rate of PE recurrence.
Most SSPE events are diagnosed incidentally and do not result in cardiac injury or hemodynamic impairment.
Abstract
In the cancer population, patients diagnosed with venous thromboembolism (VTE) are considered to have a threefold increased risk of mortality compared with those without VTE. With the advent of modern computed tomography (CT), the rate of diagnosis of subsegmental pulmonary embolism (SSPE) has increased, likely as a result of improved visualization of the peripheral pulmonary arteries. The clinical significance of SSPE remains unclear because of the lack of randomized controlled clinical trials. The aim of this study was to identify the incidence and risk factors of recurrent proximal PE within 12 months of diagnosis of SSPE in cancer. We performed a retrospective analysis of 206 adult cancer patients who were diagnosed with SSPE from 2014 to 2016 at the University of Texas MD Anderson Cancer Center. At the time of SSPE diagnosis, the majority had metastatic cancer, 108 patients (53.2%) were undergoing chemotherapy, and 23 patients (11.2%) had a history of VTE. Most patients had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2. Sixty-seven percent of SSPE was discovered incidentally on restaging CT scans, with the majority being a single and isolated event (70.9%). Within 12 months of SSPE diagnosis, 18 patients (8.7%) were found to have a recurrent PE. The patients treated with anticoagulation had a lower rate of PE recurrence (8% vs 13% in those not treated with anticoagulation). Treatment with anticoagulation did not appear to have a significant impact on overall survival (P = .48) when adjusted for ECOG performance status and cancer stage.
Introduction
In the cancer population, patients diagnosed with venous thromboembolism (VTE) are considered to have a threefold increased risk of mortality compared with those without VTE.1 With the advent of modern computed tomography (CT), the rate of diagnosis of SSPE has increased, likely as a result of improved visualization of the peripheral pulmonary arteries. The clinical significance of SSPE remains a subject of clinical research. As shown in the Prospective Investigation of Pulmonary Embolism Diagnosis study, SSPE was found to be most prevalent in patients with low-probability ventilation/perfusion scans, and several outcome studies have shown that patients with low or intermediate ventilation/perfusion scans can be safely managed with serial lower extremity Doppler imaging and no anticoagulation.2-5 Conversely, in a pooled analysis of 926 patients, van der Hulle and colleagues found that there were similar rates of recurrent VTE in patients with SSPE and those with proximal pulmonary embolism (PE).6,7 However, a recent systematic review and meta-analysis showed no increase in recurrence of VTE or death rates in patients with SSPE who were not treated with anticoagulation.8 Approximately one-half of all VTEs in cancer patients are detected incidentally, frequently during restaging imaging.6 With regard to clinical practice, patients with incidental SSPE are often managed in accordance with current guidelines for treatment of symptomatic PE.6,9 Furthermore, surveys have shown that, despite the unclear clinical significance, the majority of physicians chose to anticoagulate cancer patients diagnosed with incidental SSPE.6,10 However, because of the risks associated with anticoagulation therapy, particularly major bleeding, further research is necessary regarding the management of SSPE in the cancer population.
The primary objective of this study was to identify the clinical and radiological factors associated with recurrent SSPE or new proximal PE within 12 months after diagnosis of SSPE in adult cancer patients at the University of Texas MD Anderson Cancer Center.
Methods
A retrospective study, using electronic medical record data, was conducted at the University of Texas MD Anderson Cancer Center from 2014 to 2016. The selection of the time was arbitrary and at the time of institutional review board approval and the study start (2017-2018), the investigators intended to have data for at least 2 years and with 12-month follow-up available for all the patients.
SSPE events were identified using a historical database from the radiology department. SSPE cases were defined and identified as adult patients (≥18 years) with a concurrent cancer diagnosis in our institution and in whom SSPE was found through dedicated CT angiograms of the pulmonary artery or on other contrast enhanced CT of the chest.
Diagnosing SSPE can be challenging, and interobserver variability remains a problem, especially when SSPE is diagnosed on suboptimal scans such as nondedicated contrast-enhanced chest CT. To attenuate the effect of those nuances, all CT chest images were reviewed by a thoracic radiologist with 12 years of subspecialty clinical experience and a number of publications on imaging of PE. This was considered the gold standard in our study for the purpose of case and the primary objective event adjudication.
Cancer status was categorized as “active disease” if patients had measurable disease or were receiving antineoplastic treatment at the time of SSPE diagnosis, and “remission” if they did not have measurable disease and were not in cancer treatment.
Incidental PE was defined as those discovered on CT examinations performed for reasons other than evaluation of possible PE (ie, CT imaging for cancer staging or suspected infection). All available radiology studies were reviewed by an independent chest radiologist, with expertise in cancer patients, for documentation of the study entry criteria and the study primary end point (recurrent SSPE or new proximal PE).
The primary end point was considered within the first 12 months since SSPE diagnosis. CT evidence of right heart strain was recorded as present if the right ventricular to left ventricular diameter ratio was found to be ≥1.11
We excluded patient records from the study if the presentation of SSPE was accompanied by segmental or more proximal thrombotic events in the pulmonary artery, if the review by the independent radiologist ruled out SSPE, or if long-term follow-up data were not available in the electronic medical record.
We collected information related to patient demographics, tumor type, tumor stage, antineoplastic treatment, CT type (CT angiogram vs contrast-enhanced CT), radiological characteristics of SSPE, and the management of SSPE, including anticoagulation therapy. The date of death was also obtained to analyze for survival outcomes. The MD Anderson Cancer Center institutional review board approved the study.
Statistical methods
After compilation, the distribution of each continuous variable was summarized by its median, minimum, and maximum range values. The distribution of each categorical variable was summarized in terms of its frequencies and percentages. χ2 or Kruskal-Wallis tests were used to examine the association between groups of pulmonary embolism recurrence.
The cumulative incidence ratio of the recurrent SSPE or new proximal PE was estimated by Fine-Gray competing risk approach, with death as a competing risk factor. The recurrence-free survival was defined as the time of PE until recurrence or death, and the survival time was censored if the patient was alive at the last follow-up (Figure 1).
Consort diagram for screening and selection of cancer patients with acute subsegmental pulmonary embolism.
Consort diagram for screening and selection of cancer patients with acute subsegmental pulmonary embolism.
The cumulative incidence ratio of recurrent SSPE/new proximal PE was also stratified by the use of anticoagulation vs no anticoagulation, by the CT chest study type (CT angiogram vs contrast-enhanced CT) and censoring of patients when they stopped anticoagulation. Fine-Gray competing risk regression model was used to assess the effect of duration of anticoagulation on SSPE recurrence/new proximal PE.
The overall survival (OS) was evaluated in the study population. Cox regression model was used to evaluate the clinical factor effect on OS where OS was defined from the initial SSPE event until death or last follow-up visit. Under the univariate Cox model, the variables showed potentially significant (ie, P ≤ .1), were used to start the backward model selection. The multivariate Cox model was finalized when the variables reached statistical significance (P ≤ .05). All computations were carried out in SAS, version 9.4.
Results
A total of 206 adult cancer patients diagnosed with SSPE met the inclusion criteria (Figure 1). Of those, the majority had active malignant disease, metastatic cancer, and a good performance status (0-2). The median age at presentation was 62 years (range, 19-90). Most SSPE was discovered incidentally, predominantly in the right lung (Table 1).
Clinical and radiological characteristics of 206 cancer-associated SSPE cases
| Characteristic . | Categories . | N (%) . |
|---|---|---|
| Sex | Female | 87 (42.2) |
| ECOG performance status | 0 | 46 (22.7) |
| 1 | 94 (46.3) | |
| 2 | 33 (16.3) | |
| 3 | 25 (12.3) | |
| 4 | 5 (2.5) | |
| Cancer type* | ||
| Lung | 39 (18.9) | |
| Colorectal | 18 (8.7) | |
| Breast | 18 (8.7) | |
| Hematologic | 17 (8.3) | |
| Urologic | 17 (8.3) | |
| Gynecologic | 15 (7.3) | |
| Sarcoma | 15 (7.3) | |
| Esophageal | 11 (5.3) | |
| Pancreatic | 9 (4.4) | |
| Gastric | 6 (2.9) | |
| Other | 41 (19.9) | |
| AJCC stage† | 1 | 9 (4.7) |
| 2 | 14 (7.4) | |
| 3 | 28 (14.7) | |
| 4 | 139 (73.2) | |
| Cancer status | Active disease | 17 7(85.9) |
| Remission within 2 y | 17 (8.3) | |
| >2 y since remission | 12 (5.8) | |
| Central nervous tumor or metastasis | 21 (10.2) | |
| Lung or pleural metastasis | 101 (49) | |
| Liver metastasis | 43 (20.9) | |
| Active chemotherapy | 108 (53.2) | |
| Endocrine therapy | 11 (5.4) | |
| Antiangiogenic therapy | 21 (10.3) | |
| Major surgery within 30 d before SSPE | 25 (12.) | |
| History of venous thrombosis | 23 (11.2) | |
| Other indication for long-term anticoagulation | 30 (14.6) | |
| Concurrent antiplatelet therapy | 35 (17) | |
| Obesity | 68 (33) | |
| Concurrent use of erythropoietin agents | 1 (0.5) | |
| Type of CT study | CT chest PE protocol | 68 (33) |
| Contrast-enhanced CT | 138 (67) | |
| Single SSPE | 139 (70.9) | |
| Right pulmonary artery location | 132 (64.1) | |
| RV/LV ratio >1 | 11 (5.3) |
| Characteristic . | Categories . | N (%) . |
|---|---|---|
| Sex | Female | 87 (42.2) |
| ECOG performance status | 0 | 46 (22.7) |
| 1 | 94 (46.3) | |
| 2 | 33 (16.3) | |
| 3 | 25 (12.3) | |
| 4 | 5 (2.5) | |
| Cancer type* | ||
| Lung | 39 (18.9) | |
| Colorectal | 18 (8.7) | |
| Breast | 18 (8.7) | |
| Hematologic | 17 (8.3) | |
| Urologic | 17 (8.3) | |
| Gynecologic | 15 (7.3) | |
| Sarcoma | 15 (7.3) | |
| Esophageal | 11 (5.3) | |
| Pancreatic | 9 (4.4) | |
| Gastric | 6 (2.9) | |
| Other | 41 (19.9) | |
| AJCC stage† | 1 | 9 (4.7) |
| 2 | 14 (7.4) | |
| 3 | 28 (14.7) | |
| 4 | 139 (73.2) | |
| Cancer status | Active disease | 17 7(85.9) |
| Remission within 2 y | 17 (8.3) | |
| >2 y since remission | 12 (5.8) | |
| Central nervous tumor or metastasis | 21 (10.2) | |
| Lung or pleural metastasis | 101 (49) | |
| Liver metastasis | 43 (20.9) | |
| Active chemotherapy | 108 (53.2) | |
| Endocrine therapy | 11 (5.4) | |
| Antiangiogenic therapy | 21 (10.3) | |
| Major surgery within 30 d before SSPE | 25 (12.) | |
| History of venous thrombosis | 23 (11.2) | |
| Other indication for long-term anticoagulation | 30 (14.6) | |
| Concurrent antiplatelet therapy | 35 (17) | |
| Obesity | 68 (33) | |
| Concurrent use of erythropoietin agents | 1 (0.5) | |
| Type of CT study | CT chest PE protocol | 68 (33) |
| Contrast-enhanced CT | 138 (67) | |
| Single SSPE | 139 (70.9) | |
| Right pulmonary artery location | 132 (64.1) | |
| RV/LV ratio >1 | 11 (5.3) |
AJCC, American Joint Committee on Cancer; ECOG, Eastern Cooperative Oncology Group; RV/LV, right ventricular/left ventricular; SSPE, subsegmental pulmonary embolism.
Patients had multiple concurrent malignancies.
Calculated based on the number of patients (N = 190) classified using AJCC system, when available and applicable.
Although most of the cases did not have a prior indication or history of anticoagulation for venous thrombosis, some had other indications for anticoagulation (eg, atrial fibrillation, history of venous thrombosis) and developed SSPE while on long-term anticoagulation therapy (Table 1). After diagnosis of SSPE, 151 patients naïve to anticoagulation were started on it, 9 patients were managed with inferior vena cava interruption device only (6 because of active clinical relevant bleeding, 2 because of recent neurosurgery procedures, and 1 because of thrombocytopenia) and 15 patients did not receive any therapy (1 case from active clinical relevant bleeding; in 1 case, a dedicated CT pulmonary angiogram was recommended but not done, 2 cases were deemed clinically not relevant by the evaluating physician, 1 patient transitioned to hospice and declined antithrombotic care, and 10 cases did not have a clear reason for the clinical decision) .
Low-molecular-weight heparins (LMWH) were the most common treatment (123, 59.7%), followed by direct oral anticoagulants (DOACs) (20, 9.7%). Of those treated with LMWH, 10 patients transitioned to DOACs during follow-up.
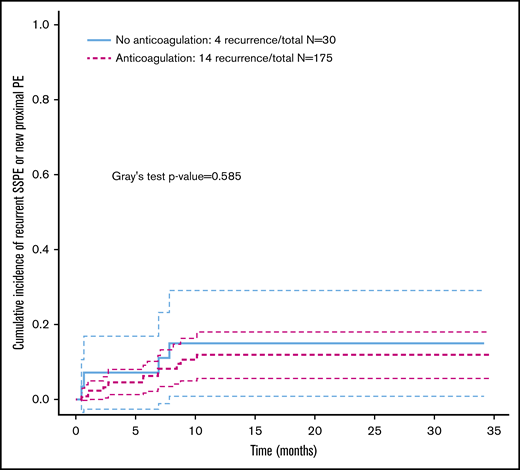
Within 12 months of SSPE diagnosis, 18 patients (8.7%) were found to have recurrent SSPE or new proximal PE. Of the clinical and radiological factors analyzed, none was found to have a significant association with recurrence of PE (Table 2). The rate of recurrent events was lower in those receiving anticoagulation vs those who did not receive anticoagulation (8% vs 13%, respectively); there were 14 PE recurrent events in the patients receiving anticoagulation (Figure 2). The patients under therapeutic anticoagulation had a lower cumulative incidence of PE recurrence, although the P value is nonsignificant (P = .585).
Univariate analysis for clinical, radiological characteristics and pulmonary embolism recurrence outcome in SSPE cases
| Characteristic . | Categories . | Recurrence: no (n = 188), n (%) . | Recurrence: yes (n = 18), n (%) . | Fisher's exact test P (2-tailed) . |
|---|---|---|---|---|
| Sex | Female | 82 (43.6) | 5 (27.8) | .2212 |
| Male | 106 (56.4) | 13 (72.2) | ||
| ECOG performance status | 0 | 43 (23.2) | 3 (16.7) | .8621 |
| 1 | 84 (45.4) | 10 (55.6) | ||
| 2 | 31 (16.8) | 2 (11.1) | ||
| 3 | 22 (11.9) | 3 (16.6) | ||
| 4 | 5 (2.7) | 0 | ||
| Cancer type | Lung | 33 (17.6) | 6 (33.3) | .3750 |
| Colorectal | 17 (9.0) | 1 (5.6) | ||
| Breast | 17 (9.0) | 1 (5.6) | ||
| Hematologic | 14 (7.4) | 3 (16.7) | ||
| Urologic | 13 (6.9) | 4 (22.2) | ||
| Gynecologic | 15 (8.0) | 0 | ||
| Sarcoma | 14 (7.4) | 1 (5.6) | ||
| Esophageal | 11 (5.9) | 0 | ||
| Pancreatic | 8 (4.3) | 1 (5.6) | ||
| Gastric | 6 (3.2) | 0 | ||
| Other | 38 (2.0) | 3 (16.7) | ||
| AJCC stage | 1 | 8 (4.6) | 1 (6.3) | .5152 |
| 2 | 12 (6.9) | 2 (12.5) | ||
| 3 | 27 (15.5) | 1 (6.3) | ||
| 4 | 127 (73.0) | 12 (75.0) | ||
| Cancer status | Active disease | 162 (86.2) | 15 (83.3) | .8613 |
| Remission within 2 y | 15 (8.0) | 2 (11.1) | ||
| >2 y since remission | 11 (5.9) | 1 (5.6) | ||
| Central nervous tumor or metastasis | Yes | 19 (10.1) | 2 (11.1) | 1.000 |
| No | 169 (89.9) | 16 (88.9) | ||
| Lung or pleural metastasis | Yes | 92 (48.9) | 9 (50.0) | 1.000 |
| No | 96 (51.1) | 9 (50.0) | ||
| Liver metastasis | Yes | 39 (20.7) | 4 (22.2) | 1.000 |
| No | 149 (79.3) | 14 (77.8) | ||
| Active chemotherapy | Yes | 100 (53.2) | 8 (44.4) | .4675 |
| No | 85 (45.2) | 10 (55.6) | ||
| Endocrine therapy | Yes | 10 (5.3) | 1 (5.6) | 1.000 |
| No | 175 (93.1) | 17 (94.4) | ||
| Antiangiogenic therapy | Yes | 18 (9.6) | 3 (16.7) | .4079 |
| No | 167 (88.8) | 15 (83.3) | ||
| Major surgery within 30 d before SSPE | Yes | 22 (11.7) | 3 (16.7) | .4641 |
| No | 166 (88.3) | 15 (83.3) | ||
| History of venous thrombosis | Yes | 20 (10.6) | 3 (16.7) | .4318 |
| No | 168 (89.4) | 15 (83.3) | ||
| Other indication for long-term anticoagulation | Yes | 28 (14.9) | 2 (11.1) | 1.000 |
| No | 160 (85.1) | 16 (88.9) | ||
| Concurrent antiplatelet therapy | Yes | 33 (17.6) | 2 (11.1) | .7438 |
| No | 155 (82.4) | 16 (88.9) | ||
| SSPE treatment | Other | 27 (14.4) | 4 (22.2) | .4860 |
| Anticoagulation | 161 (85.6) | 14 (77.8) | ||
| Obesity | Yes | 63 (33.5) | 5 (27.8) | .7946 |
| No | 125 (66.5) | 13 (72.2) | ||
| Concurrent use of erythropoietin agents | Yes | 1 (0.5) | 0 | 1.000 |
| No | 187 (99.5) | 18 (100) | ||
| Type of CT study | CT chest PE protocol | 61 (32.4) | 7 (38.9) | .6051 |
| CT chest with contrast | 127 (67.6) | 11 (61.1) | ||
| SSPE, no/ | 1 | 125 (66.5) | 14 (77.8) | .5432 |
| 2 | 20 (10.6) | 2 (11.1) | ||
| 3 | 33 (17.6) | 1 (5.6) | ||
| 4 | 1 (0.5) | 0 | ||
| SSPE location | Right sided | 117 (62.2) | 15 (83.3) | .2419 |
| Left sided | 32 (17.0) | 1 (5.6) | ||
| Bilateral | 39 (20.7) | 2 (11.1) | ||
| RV/LV ratio > 1 | Yes | 11 (5.9) | 0 | .6038 |
| No | 177 (94.1) | 18 (100) |
| Characteristic . | Categories . | Recurrence: no (n = 188), n (%) . | Recurrence: yes (n = 18), n (%) . | Fisher's exact test P (2-tailed) . |
|---|---|---|---|---|
| Sex | Female | 82 (43.6) | 5 (27.8) | .2212 |
| Male | 106 (56.4) | 13 (72.2) | ||
| ECOG performance status | 0 | 43 (23.2) | 3 (16.7) | .8621 |
| 1 | 84 (45.4) | 10 (55.6) | ||
| 2 | 31 (16.8) | 2 (11.1) | ||
| 3 | 22 (11.9) | 3 (16.6) | ||
| 4 | 5 (2.7) | 0 | ||
| Cancer type | Lung | 33 (17.6) | 6 (33.3) | .3750 |
| Colorectal | 17 (9.0) | 1 (5.6) | ||
| Breast | 17 (9.0) | 1 (5.6) | ||
| Hematologic | 14 (7.4) | 3 (16.7) | ||
| Urologic | 13 (6.9) | 4 (22.2) | ||
| Gynecologic | 15 (8.0) | 0 | ||
| Sarcoma | 14 (7.4) | 1 (5.6) | ||
| Esophageal | 11 (5.9) | 0 | ||
| Pancreatic | 8 (4.3) | 1 (5.6) | ||
| Gastric | 6 (3.2) | 0 | ||
| Other | 38 (2.0) | 3 (16.7) | ||
| AJCC stage | 1 | 8 (4.6) | 1 (6.3) | .5152 |
| 2 | 12 (6.9) | 2 (12.5) | ||
| 3 | 27 (15.5) | 1 (6.3) | ||
| 4 | 127 (73.0) | 12 (75.0) | ||
| Cancer status | Active disease | 162 (86.2) | 15 (83.3) | .8613 |
| Remission within 2 y | 15 (8.0) | 2 (11.1) | ||
| >2 y since remission | 11 (5.9) | 1 (5.6) | ||
| Central nervous tumor or metastasis | Yes | 19 (10.1) | 2 (11.1) | 1.000 |
| No | 169 (89.9) | 16 (88.9) | ||
| Lung or pleural metastasis | Yes | 92 (48.9) | 9 (50.0) | 1.000 |
| No | 96 (51.1) | 9 (50.0) | ||
| Liver metastasis | Yes | 39 (20.7) | 4 (22.2) | 1.000 |
| No | 149 (79.3) | 14 (77.8) | ||
| Active chemotherapy | Yes | 100 (53.2) | 8 (44.4) | .4675 |
| No | 85 (45.2) | 10 (55.6) | ||
| Endocrine therapy | Yes | 10 (5.3) | 1 (5.6) | 1.000 |
| No | 175 (93.1) | 17 (94.4) | ||
| Antiangiogenic therapy | Yes | 18 (9.6) | 3 (16.7) | .4079 |
| No | 167 (88.8) | 15 (83.3) | ||
| Major surgery within 30 d before SSPE | Yes | 22 (11.7) | 3 (16.7) | .4641 |
| No | 166 (88.3) | 15 (83.3) | ||
| History of venous thrombosis | Yes | 20 (10.6) | 3 (16.7) | .4318 |
| No | 168 (89.4) | 15 (83.3) | ||
| Other indication for long-term anticoagulation | Yes | 28 (14.9) | 2 (11.1) | 1.000 |
| No | 160 (85.1) | 16 (88.9) | ||
| Concurrent antiplatelet therapy | Yes | 33 (17.6) | 2 (11.1) | .7438 |
| No | 155 (82.4) | 16 (88.9) | ||
| SSPE treatment | Other | 27 (14.4) | 4 (22.2) | .4860 |
| Anticoagulation | 161 (85.6) | 14 (77.8) | ||
| Obesity | Yes | 63 (33.5) | 5 (27.8) | .7946 |
| No | 125 (66.5) | 13 (72.2) | ||
| Concurrent use of erythropoietin agents | Yes | 1 (0.5) | 0 | 1.000 |
| No | 187 (99.5) | 18 (100) | ||
| Type of CT study | CT chest PE protocol | 61 (32.4) | 7 (38.9) | .6051 |
| CT chest with contrast | 127 (67.6) | 11 (61.1) | ||
| SSPE, no/ | 1 | 125 (66.5) | 14 (77.8) | .5432 |
| 2 | 20 (10.6) | 2 (11.1) | ||
| 3 | 33 (17.6) | 1 (5.6) | ||
| 4 | 1 (0.5) | 0 | ||
| SSPE location | Right sided | 117 (62.2) | 15 (83.3) | .2419 |
| Left sided | 32 (17.0) | 1 (5.6) | ||
| Bilateral | 39 (20.7) | 2 (11.1) | ||
| RV/LV ratio > 1 | Yes | 11 (5.9) | 0 | .6038 |
| No | 177 (94.1) | 18 (100) |
Cumulative incidence of pulmonary embolism recurrence stratified by treatment strategy: anticoagulation vs no anticoagulation. SSPE, subsegmental pulmonary embolism.
Cumulative incidence of pulmonary embolism recurrence stratified by treatment strategy: anticoagulation vs no anticoagulation. SSPE, subsegmental pulmonary embolism.
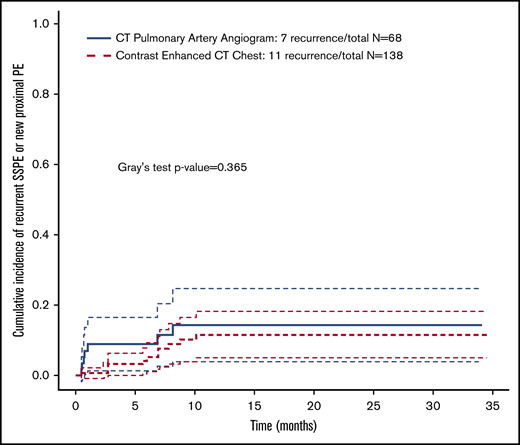
Additionally, we analyzed the cumulative PE recurrence by the diagnostic study used (CT angiogram of the chest PE protocol vs other incidentally detected events by chest CT with contrast). There was not a statistically significant difference among the CT chest modalities (Figure 3).
Cumulative incidence of pulmonary embolism recurrence stratified by CT study type.
Cumulative incidence of pulmonary embolism recurrence stratified by CT study type.
We also captured other venous thrombotic complications that occurred in the patients with recurrent SSPE or new proximal PE. We observed 5 additional events distributed in 5 patients. Two of those events were incidentally found (inferior vena cava thrombosis) and 3 events were accompanied by symptoms (right subclavian deep vein thrombosis in 2 patients and 1 left femoropopliteal deep vein thrombosis). All of those events occurred in patients who had received anticoagulation for the index SSPE.
Clinically relevant bleeding complications were documented in 26 patients (12.6%). According to the International Society on Thrombosis and Haemostasis bleeding classification criteria,12 11 patients (5.3%) presented major bleeding and 15 patients (7.3%) had nonmajor events. All events occurred in patients that received anticoagulation treatment.
In regard to the location, cutaneous/subcutaneous and gastrointestinal bleeding accounted for 7 events each, followed by 4 events in the urinary tract, 3 events of intracranial bleeding, and 2 cases of oral/nasal mucosal bleeding. The remaining of the events occurred in other locations.
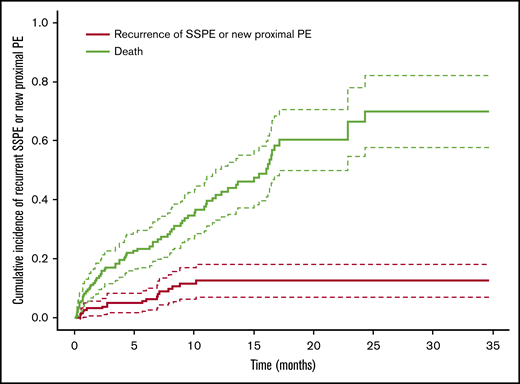
We examined the OS of the patients after the diagnosis of SSPE and the time-to-event for recurrent PE using death as a competing risk factor. A cumulative incidence of recurrence was estimated by Fine-Gray competing risk approach, with death as a competing risk factor. The recurrence-free survival was defined as the time of SSPE until PE recurrence or death, and the survival time was censored if the patient was alive at the last follow-up time (Figure 4).
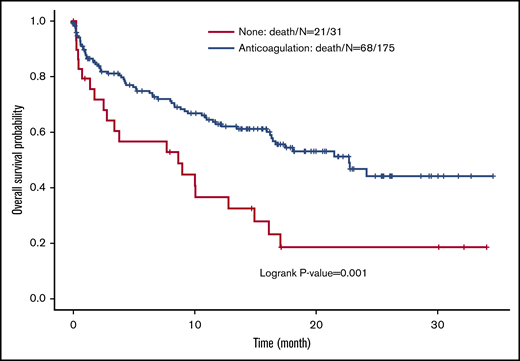
We found a survival benefit for those patients who received anticoagulation (P = .001) (Figure 5). We also found that the Eastern Cooperative Oncology Group performance status and the cancer staging were the main determinants of OS. When adjusted for those variables, the use of anticoagulation for SSPE did not seem to have an effect on OS (Figure 5; Table 3).
Kaplan-Meier estimates for overall survival (y) stratified by the use of anticoagulation in the treatment of cancer patients with acute subsegmental pulmonary embolism.
Kaplan-Meier estimates for overall survival (y) stratified by the use of anticoagulation in the treatment of cancer patients with acute subsegmental pulmonary embolism.
Univariate and multivariate analysis of overall survival in cancer patients with acute subsegmental pulmonary embolism
| Variable . | Univariate analysis . | Multivariate analysis . | |||||||
|---|---|---|---|---|---|---|---|---|---|
| P . | HR . | 95% HR confidence limits . | P . | HR . | 95% HR confidence limits . | ||||
| Age | .575 | 1.00 | 0.99 | 1.02 | — | — | — | — | |
| Sex (male vs female) | .420 | 0.84 | 0.55 | 1.28 | — | — | — | — | |
| Use of anticoagulation | .001 | 0.44 | 0.27 | 0.72 | .4799 | 0.818 | 0.469 | 1.428 | |
| ECOG (3-4 vs 0-2) | <.0001 | 6.15 | 3.74 | 10.12 | <.0001 | 6.345 | 3.614 | 11.139 | |
| AJCC stage (4 vs 1-3) | <.0001 | 4.80 | 2.31 | 9.97 | <.0001 | 5.031 | 2.386 | 10.604 | |
| Variable . | Univariate analysis . | Multivariate analysis . | |||||||
|---|---|---|---|---|---|---|---|---|---|
| P . | HR . | 95% HR confidence limits . | P . | HR . | 95% HR confidence limits . | ||||
| Age | .575 | 1.00 | 0.99 | 1.02 | — | — | — | — | |
| Sex (male vs female) | .420 | 0.84 | 0.55 | 1.28 | — | — | — | — | |
| Use of anticoagulation | .001 | 0.44 | 0.27 | 0.72 | .4799 | 0.818 | 0.469 | 1.428 | |
| ECOG (3-4 vs 0-2) | <.0001 | 6.15 | 3.74 | 10.12 | <.0001 | 6.345 | 3.614 | 11.139 | |
| AJCC stage (4 vs 1-3) | <.0001 | 4.80 | 2.31 | 9.97 | <.0001 | 5.031 | 2.386 | 10.604 | |
Discussion
VTE represents a major cause of morbidity in cancer patients.13 Both incidentally found and symptomatic cancer related-VTE have a high risk of recurrence as shown in recent prospective cohort studies.14,15 In our study, the majority of SSPE diagnoses occurred incidentally on CT scans performed for reasons other than to evaluate for PE. There are no data from randomized controlled prospective studies to guide whether these patients should receive anticoagulation.
The current consensus guidelines recommend anticoagulation treatment of cancer patients with SSPE, even if incidentally detected.16-18 Our patients were typically treated with anticoagulation, and LMWH was the medication of choice as recommended by those same guidelines. Some patients received DOACs because they had begun to be introduced in the medical practice for cancer patients. Recent randomized clinical trials for the treatment of cancer-associated VTE have confirmed the efficacy of those agents in the prevention of recurrent VTE.19-21 An important observation is that in our cohort the incidence of recurrent PE was similar to the rates seen in the recent cancer-VTE anticoagulation trials. In our cohort, the rate of recurrent events was lower in those receiving anticoagulation vs those who did not receive anticoagulation (8% vs 13%, respectively); however, that difference did not reach statistical significance (P = .368). Altogether, our observations further support the indication for anticoagulation in cancer-related SSPE.
In regard to the impact of VTE recurrence on the overall survival after SSPE, it seems that advanced cancer and poor performance status are important competing risks and substantial contributors to survival outcomes. Similar observations have been identified in all large randomized cancer-VTE trials.19,21-24 None of those studies has shown a survival advantage for the use of anticoagulation in cancer patients. Whether patients with early or locally advanced cancer and good performance status would have a survival benefit from treating SSPE with anticoagulation remains an unanswered question. Additionally, there are relatively few patients with multiple SSPE or CT evidence of right ventricular strain in our cohort. We did not observe associations of these imaging features with clinical outcomes of our patients.
The limitations of our study are the biases possibly introduced because of the retrospective nature of our cohort study. First, the use of a historical radiology database for initial identification of cases of SSPE may have introduced discrepancy and errors in the case reporting. We decided to mitigate those potential errors by reviewing all radiology studies considered in the present study and under the scrutiny of an expert radiologist specializing in thoracic and pulmonary artery disease. Although we believe that this strategy may have attenuated the chance of overdiagnosis, we cannot determine if other eligible cases may have not been included given the strategy used to initially identify SSPE cases.
Second, there were 40 patients initially identified and confirmed to have SSPE who were not included because of missing follow-up data. Other baseline characteristics for those patients were not fully available. Therefore, selection bias may have been introduced in our cohort. Nevertheless, our findings for SSPE recurrence are not different when compared with those of prospective cohorts of incidental PE.14 We presume the effect of the selection by only the cases with complete information may have not had a substantial impact on the overall results.
We did a comprehensive review and collection of several clinical, tumor-related, and radiological features, but cannot rule out that there are other factors contributing to the outcomes related to SSPE. It is also possible that the true rate of recurrent PE is underestimated because each patient had a different opportunity to have repeat imaging during follow up (eg, different restaging protocols according to the plan of follow-up for the primary malignancy).
In conclusion, the management of SSPE in cancer patients remains a clinical challenge because of the lack of randomized controlled clinical trials examining these cases. The current evidence, including ours, supports the use of anticoagulation treatment of SSPE in the oncology setting, whether they are symptomatic or incidentally found.
Send data sharing requests via e-mail to the corresponding author.
Authorship
Contribution: C.M.R.-H. and M.Y. designed the study, collected and analyzed the data, and approved the final manuscript document; R.K. and C.C.W. collected and analyzed the data and approved the final manuscript document; and W.Q. analyzed the data and approved the final manuscript document.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Cristhiam M. Rojas-Hernandez, Section of Benign Hematology, Division of Internal Medicine, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Suite 1464 Houston, TX 77030; e-mail: cmrojas@mdanderson.org.