Key Points
Substitution of ixazomib for MMF post-HIDT results in reliable engraftment, comparable rates of GVHD and NRM, and favorable OS.
Cytopenias and rashes were common but manageable, resulting in ixazomib dose reductions in most patients.
Abstract
Proteasome inhibition results in extensive immunomodulatory effects that augment natural killer cell cytotoxicity and inhibit aspects of T-cell, B-cell, and dendritic cell function. We performed a phase 2 study that examined the effects of ixazomib for graft-versus-host disease (GVHD) prophylaxis (up to 12 cycles) with posttransplant cyclophosphamide and tacrolimus after standard nonmyeloablative haploidentical donor transplantation (HIDT). Ixazomib was started on day +5 (4 mg on days 1, 8, and 15 of a 28-day cycle), with dose reductions allowed in future cycles for toxicity. All patients received peripheral blood stem cells. Twenty-five patients were enrolled with a median age of 62 years (range, 35-77 years) who had acute leukemia (4), myelodysplastic syndrome (7), non-Hodgkin lymphoma/Hodgkin lymphoma/chronic lymphocytic leukemia (8), and myeloma (6). The hematopoietic cell transplant comorbidity index was ≥3 in 68% of the patients. After a median follow-up of 33.5 months, the cumulative incidence of relapse/progression at 1 year was 24% and 44% at 3 years, which failed to meet the statistically predefined goal of decreasing 1-year risk of relapse. Engraftment occurred in all patients with no secondary graft failure, and 3-year nonrelapse mortality (NRM) was 12%. Cumulative incidence of grade 3 to 4 acute GVHD was 8%, whereas moderate-to-severe chronic GVHD occurred in 19%. Nineteen patients survive with an estimated 1-year overall survival (OS) of 84% and 3-year OS of 74%. Hematologic and cutaneous toxicities were common but manageable. The substitution of ixazomib for mycophenolate mofetil (MMF) post-HIDT results in reliable engraftment, comparable rates of clinically significant GVHD, relapse and NRM, and favorable OS. This trial was registered at www.clinicaltrials.gov as # NCT02169791.
Introduction
In patients without a matched sibling or a readily available matched unrelated donor, haploidentical donor transplantation (HIDT) using posttransplant cyclophosphamide (PTCy), tacrolimus, and mycophenolate mofetil (MMF) has been shown to be an appropriate alternative donor source associated with comparable transplant outcomes.1-3 As originally developed by the team at Johns Hopkins University, a nonmyeloablative (NMA) preparative regimen was used (fludarabine, low-dose total body irradiation [TBI], and cyclophosphamide) with a T-replete bone marrow (BM) graft. This HIDT-PTCy approach allowed for successful transplantation with relatively low rates of graft-versus-host disease (GVHD) and nonrelapse mortality (NRM).4,5 However, relapse rates were relatively high. Peripheral blood stem cell (PBSC) grafts have been used as a means of reducing graft failure and relapse rates after NMA HIDT-PTCy. However, using this approach, graft rejection and relapse remain causes of treatment failure in approximately 10% and 30%, respectively; slightly over half the patients remain alive and disease free 2 years posttransplant.6
The combination of tacrolimus and MMF is the standard GVHD prophylaxis regimen used with PTCy in most HIDT protocols. Alternative immunosuppressive drug combinations may help further reduce the risk of treatment failure. Incorporation of proteasome inhibitors (PIs) into GVHD prevention regimens represents one such strategy, which has generated significant interest. Inhibition of the ubiquitin-proteasome proteolytic pathway results in extensive immunomodulatory effects, inhibiting aspects of T-cell, B-cell, and dendritic cell function while augmenting natural killer (NK) cell cytotoxicity.7,8 Although PIs have direct antimalignancy activity in hematologic malignancies such as myeloma and non-Hodgkin lymphoma,9 they also have the capacity to sensitize cancer cells to T-cell and NK-cell–mediated cytotoxicity.8,10 Murine studies demonstrated reduction of acute GVHD (aGVHD) with retention of graft-versus-malignancy (GVM) effects with posttransplant administration of the PI bortezomib.11 These preclinical results have been confirmed in clinical trials in the context of HLA-mismatched unrelated donor transplantation.12,13
Ixazomib is an orally available PI that potently, reversibly, and selectively inhibits the proteasome. Although ixazomib selectivity and potency are similar to those of bortezomib, the proteasome dissociation half-life for ixazomib is approximately 6 times shorter than that of bortezomib, meaning a wider tissue distribution of the drug.14 Furthermore, the toxicity profile is different from that of bortezomib, with a significantly reduced incidence of peripheral neuropathy. Murine studies have also demonstrated efficacy of ixazomib in GVHD prophylaxis when combined with PTCy.15
We performed a phase 2 study that examined the effects of ixazomib used for GVHD prophylaxis and maintenance (up to 12 cycles), in addition to PTCy and tacrolimus, after a standard NMA HIDT regimen (fludarabine, low-dose TBI, cyclophosphamide) and PBSC transplantation. We hypothesized that substituting ixazomib for MMF would provide acceptable GVHD control while potentially augmenting GVM.
Patients and methods
Patients and donors
Twenty-five patients were accrued to this protocol between January 2015 and August 2018. Patients were eligible for inclusion if they were age 18 years or older, had a standard indication for allogeneic transplant, and were appropriate candidates for reduced-intensity conditioning (eg, older age, significant comorbidity, previous autologous transplant). Specific inclusion and exclusion criteria are listed in the supplemental Data. Other inclusion criteria included availability of a 5-8/10 HLA-matched haploidentical family donor in the absence of significant donor-specific antibodies or a positive HLA crossmatch. Exclusion criteria included previous allogeneic transplant, bilirubin ≥2.5 mg/dL, creatinine ≥2 mg/dL, cardiac ejection fraction <40%, pulmonary function <50% predicted, and Karnofsky performance status <70%. Written informed consent was obtained for all patients in accordance with the Declaration of Helsinki. The study was approved by the institutional review board at Northside Hospital.
Treatment protocol
Eligible donors were family members that were HLA allele matched at 5-8/10 loci (HLA-A, -B, -C, -DR, -DQ) with the recipient. Donors were selected to maximize NK alloreactivity (ie, the activating B/x killer cell immunoglobulin-like receptor (KIR) haplotype was favored over the more inhibitory KIR A/A haplotype). Donors were considered ineligible if the recipient possessed high levels of donor-specific HLA antibodies (>3000 mean fluorescent intensity by panel reactive antibody testing) or a positive HLA crossmatch against that specific donor.
Transplant conditioning consisted of fludarabine 30 mg/m2 per day for 5 days (days −6 to −2), cyclophosphamide 14.5 mg/kg per day for 2 days (days −6 and −5), and TBI 200 cGy in 1 fraction on day −1. On day 0, patients received an unmanipulated PBSC allograft with a CD34 dose capped at 5 × 106/kg recipient weight. PTCy was administered on days +3 and +4 at 50 mg/kg per day. Tacrolimus was initiated intravenously on day +5 at 0.03 mg/kg per day to achieve target serum levels of 5 to 15 ng/mL. Tacrolimus was discontinued without taper on day +180 in the absence of GVHD.
Ixazomib was initiated on day +5 at 4 mg orally once per week × 3 doses (28-day cycle) for up to 12 cycles. All patients received cycle 1 at the scheduled dose, but dose adjustments were permitted for future cycles for hematologic and nonhematologic toxicity (see dose modification tables in the supplemental Data).
Antimicrobial prophylaxis was administered according to institutional practice guidelines. Antimicrobial prophylaxis was started on day 0 and included a quinolone antibiotic and acyclovir. Antifungal prophylaxis consisted of micafungin until day +5, and then it was changed to oral therapy with either posaconazole or isavuconazole. Filgrastim 5 µg/kg was given once per day starting on day +5 and continuing until neutrophil engraftment. Quantitative cytomegalovirus (CMV) polymerase chain reaction (PCR) was monitored once per week starting on day +1, and pre-emptive therapy was initiated if viral reactivation was detected (≥400 IU/mL). Letormovir was not used for CMV prophylaxis for any patient during the study.
Definitions and study end points
Neutrophil engraftment was defined as the first of 3 days of an absolute neutrophil count (ANC) of >0.5 × 109/L after transplantation. Platelet engraftment was defined as a platelet count of >20 × 109/L without transfusion for the 7 preceding days. aGVHD was scored on the basis of the modified Keystone criteria.16 Chronic GVHD (cGVHD) diagnosis and grading were based on the National Institutes of Health consensus criteria.17 Progression-free survival (PFS) was defined as survival posttransplant without evidence of relapse/progression. Patients were considered to have died as a result of NRM if there was no evidence of disease relapse/progression before death.
For acute leukemia, relapse was defined as (1) reappearance of leukemia blast cells in the PB, (2) >5% blasts in the bone marrow (BM) not attributable to another cause (eg, BM regeneration), (3) development of extramedullary leukemia or leukemic cells in the cerebral spinal fluid, or (4) reappearance of cytogenetic abnormalities present before transplantation. For myelodysplastic syndrome (MDS), relapse/progression was defined by the International Working Group response criteria in myelodysplasia.18 For non-Hodgkin lymphoma/Hodgkin lymphoma, relapse/progression was defined by the Lugano criteria.19 For myeloma, relapse/progression was defined by the International Myeloma Working Group uniform response criteria.20 For chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), relapse/progression was defined by the International Workshop Group on CLL response criteria.21
The major study end point was the 1-year cumulative incidence of relapse/progression. Secondary study end points were overall survival (OS), PFS, cumulative incidence of ANC/platelet engraftment, graft failure, aGVHD and cGVHD, relapse/progression, and NRM. Additional end points were lineage-specific donor chimerism; T-, NK-, and B-lymphocyte immune reconstitution; and drug-related toxicities attributable to ixazomib.
Statistical methods
The study was powered to show a reduction in the expected 1-year risk of relapse or progression posttransplant. Experience from the literature at the time of study design suggested that after an NMA HIDT using PTCy, the risk of relapse was expected to be ∼50% at 1 year posttransplant.4 It was hypothesized that by using this protocol, this rate would be at most 25%. Thus, we statistically formalized this study by testing the null hypothesis that p, the 1-year PFS rate would be ≤0.25 vs the alternative hypothesis that p would be ≥0.5. The sample size calculation of 25 patients allowed for a 90% power with an α = 0.05, using the formula for a 1-sample binomial (2-sided) test of a proportion. Stopping rules were established to halt study accrual in the setting of higher-than-expected graft failure (>20%), NRM (>20%), or grade 3 to 4 aGVHD (>15%).
Probabilities of OS and PFS were estimated by using the Kaplan-Meier product-limit method.22 The cumulative incidences of NRM, relapse, aGVHD, and cGVHD were computed to take into account the presence of competing risks.23 NRM and relapse are 2 competing risks. Death was considered a competing risk for GVHD end points. A log-rank test was used to compare survival probabilities between 2 subgroups. We used box plots to summarize immune cell count variables. Statistical analysis was performed using SAS version 9.3 software (SAS Institute).
Results
Patient characteristics
A total of 25 patients with a median age of 62 years (range, 35-77 years) who had hematologic malignancies received a transplant on this study (lymphoma or CLL [8], MDS [7], myeloma [6], and acute leukemia [4]). Patient characteristics are listed in Table 1. The hematopoietic cell transplantation–specific comorbidity index (HCT-CI was low (0-2) in 8 patients (32%), and the disease risk index (DRI) was low in 4 patients (16%). Donors were 5/10 matched for 22 recipients (88%) who received a transplant. KIR haplotype was B/x in 18 donors (72%).
Patient characteristics (n = 25)
| Characteristic . | Status . | No. . | Median . | Range . |
|---|---|---|---|---|
| Sex | ||||
| Male | 18 | |||
| Female | 7 | |||
| Age, y | ||||
| Patient | 62 | 35-77 | ||
| Donor | 38 | 20-69 | ||
| Donor relationship | ||||
| Child | 18 | |||
| Sibling | 7 | |||
| Recipient seropositive CMV status | 16 | |||
| HLA match | ||||
| 5/10 | 22 | |||
| 6/10 | 1 | |||
| 7/10 | 2 | |||
| HCT-CI | ||||
| 0-2 | 8 | |||
| ≥3 | 17 | |||
| Karnofsky performance status <90 | 20 | |||
| Female donor/male recipient | 8 | |||
| Donor KIR haplotype | ||||
| A/A | 7 | |||
| B/X | 18 | |||
| Disease | ||||
| Ph+ acute lymphoblastic leukemia | CR1 | 2 | ||
| Acute myelogenous leukemia-MK | CR1 | 1 | ||
| Acute myelogenous leukemia | CR2 | 1 | ||
| Myelodysplastic syndrome | ||||
| EB2 | 3 | |||
| EB1 | 2 | |||
| RCMD | 2 | |||
| Chronic lymphocytic leukemia | Rel-sens | 2 | ||
| Chronic lymphocytic leukemia | Rel-res | 1 | ||
| Hodgkin lymphoma | Rel-sens | 2 | ||
| MCL | PIF-sens | 1 | ||
| MCL | Rel-sens | 1 | ||
| DLBCL | Rel-sens | 1 | ||
| MM-HR | Rel-sens | 3 | ||
| MM-SR | Rel-sens | 3 | ||
| Disease risk index | ||||
| Low | 4 | |||
| Intermediate | 16 | |||
| High | 5 | |||
| Previous autologous SCT | 9 | |||
| CD34 dose, × 106/kg | 5.01 | 3.23-5.09 | ||
| CD3 dose, × 108/kg | 1.58 | 0.27-5.33 |
| Characteristic . | Status . | No. . | Median . | Range . |
|---|---|---|---|---|
| Sex | ||||
| Male | 18 | |||
| Female | 7 | |||
| Age, y | ||||
| Patient | 62 | 35-77 | ||
| Donor | 38 | 20-69 | ||
| Donor relationship | ||||
| Child | 18 | |||
| Sibling | 7 | |||
| Recipient seropositive CMV status | 16 | |||
| HLA match | ||||
| 5/10 | 22 | |||
| 6/10 | 1 | |||
| 7/10 | 2 | |||
| HCT-CI | ||||
| 0-2 | 8 | |||
| ≥3 | 17 | |||
| Karnofsky performance status <90 | 20 | |||
| Female donor/male recipient | 8 | |||
| Donor KIR haplotype | ||||
| A/A | 7 | |||
| B/X | 18 | |||
| Disease | ||||
| Ph+ acute lymphoblastic leukemia | CR1 | 2 | ||
| Acute myelogenous leukemia-MK | CR1 | 1 | ||
| Acute myelogenous leukemia | CR2 | 1 | ||
| Myelodysplastic syndrome | ||||
| EB2 | 3 | |||
| EB1 | 2 | |||
| RCMD | 2 | |||
| Chronic lymphocytic leukemia | Rel-sens | 2 | ||
| Chronic lymphocytic leukemia | Rel-res | 1 | ||
| Hodgkin lymphoma | Rel-sens | 2 | ||
| MCL | PIF-sens | 1 | ||
| MCL | Rel-sens | 1 | ||
| DLBCL | Rel-sens | 1 | ||
| MM-HR | Rel-sens | 3 | ||
| MM-SR | Rel-sens | 3 | ||
| Disease risk index | ||||
| Low | 4 | |||
| Intermediate | 16 | |||
| High | 5 | |||
| Previous autologous SCT | 9 | |||
| CD34 dose, × 106/kg | 5.01 | 3.23-5.09 | ||
| CD3 dose, × 108/kg | 1.58 | 0.27-5.33 |
CR1, first complete response; CR2, second complete response; DLBCL, diffuse large B-cell lymphoma; EB1, excess blasts-1; EB2, excess blasts-2; HR, high risk; MCL, mantle cell lymphoma; MK, monosomal karyotype; MM, multiple myeloma; Ph+, Philadelphia chromosome positive; PIF, primary inducion failure; RCMD, refractory cytopenia with multilineage dysplasia; rel-res, chemotherapy-resistant relapse; rel-sens, chemotherapy-sensitive relapse; SCT, stem cell transplantation; SR, standard risk.
Engraftment and chimerism
Sustained donor engraftment occurred in all patients with no late graft failure; median time to neutrophil recovery was 16 days (range, 13-27 days) and to platelet recovery was 29 days (range, 16-47 days). The median day +30 PB donor CD3 chimerism was 100% (range, 90%-100%) and CD33 chimerism was 100% (range, 93%-100%).
Infections, GVHD, and duration of immunosuppression
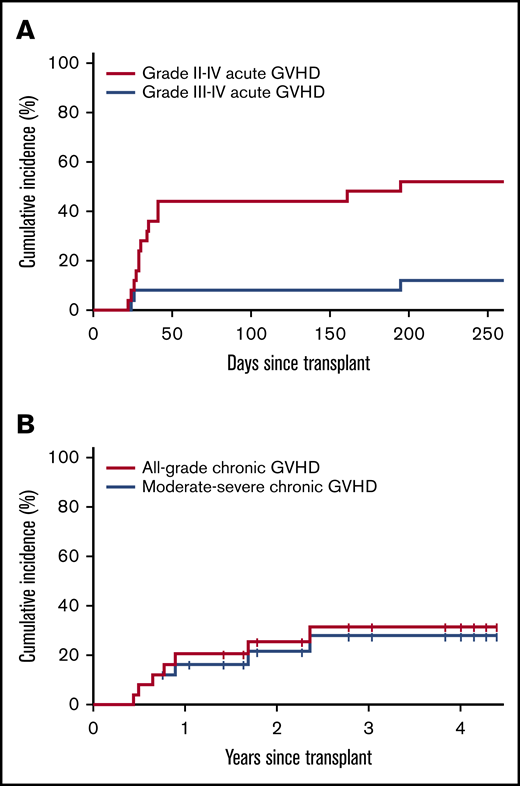
CMV reactivation (PCR ≥400 IU/mL) was observed in 14 transplant recipients (56%) (88% of CMV-seropositive recipients). No CMV-related disease or mortality was observed. BK virus-associated cystitis occurred in 5 patients with only 1 having symptoms deemed severe enough to require hospitalization for bladder irrigation and/or analgesic support. There were no cases of Epstein-Barr virus–associated lymphoproliferative disease. The cumulative incidence of grade 2 to 4 aGVHD was 48%; the cumulative incidence of grade 3 to 4 aGVHD was 8% by day +180 (Figure 1A). aGVHD occurred at a median of 28.5 days (range, 22-191 days) posttransplant. Two cases of late aGVHD were noted at day +160 and day +191. The cumulative incidence of all-grade and moderate-to-severe cGVHD at 2 years was 25% and 19%, respectively (Figure 1B). Severity grade was mild (1), moderate (3), or severe (3); 4 cases of cGVHD (3 severe and 1 moderate) occurred after interventions for posttransplant relapse. A comparison of cumulative incidences of aGVHD and cGVHD with a contemporaneous cohort of patients from our institution who received NMA PBSC HIDT-PTCy with standard tacrolimus/MMF GVHD prophylaxis is provided in supplemental Table 1. All systemic immunosuppression was discontinued in 64% of patients by 1 year and in 81% by 2 years posttransplant.
Estimated cumulative incidence of GVHD. (A) Grade 2-4 and 3-4 aGVHD. (B) All-grade and moderate-severe cGVHD.
Estimated cumulative incidence of GVHD. (A) Grade 2-4 and 3-4 aGVHD. (B) All-grade and moderate-severe cGVHD.
NRM, relapse, disease-free survival, and OS
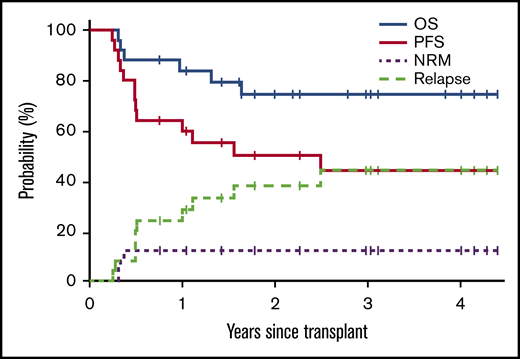
Median follow-up for surviving patients is 33.5 months (range, 9.2-52.8 months). OS, PFS, NRM, and relapse or progression curves are presented in Figure 2. Estimated OS at 1, 2, and 3 years is 84%, 74%, and 74%, respectively; corresponding rates of PFS were 64%, 50%, and 44%. The cumulative incidence of relapse/progression was 24%, 38%, and 44% at 1, 2, and 3 years posttransplant, respectively. Posttransplant relapse/progression occurred in 10 patients. Of these, 3 patients died at 2, 9, and 14 months after relapse. Seven patients remain alive and in complete response (CR) as of the data cutoff with a median follow-up for surviving patients of 16 months (range, 8-46 months). Supplemental Table 2 details postrelapse treatment and outcome for these patients. The cumulative incidence of NRM was 12% at 1, 2, and 3 years posttransplant. Three-year NRM was similar in patients with high (HCT-CI ≥3) and low comorbidity burden (12% vs 13%).
Survival estimates and estimated cumulative incidence of OS, PFS, relapse/progression, and NRM.
Survival estimates and estimated cumulative incidence of OS, PFS, relapse/progression, and NRM.
In patients with low or intermediate DRI, 1- and 3-year OS rates were 90% and 79% compared with 53% and 53% for patients with high DRI. In patients younger than age 63 years (median), 1- and 3-year OS rates were 92% and 83% compared with 74% and 65% for patients age 63 years or older. In patients with high (HCT-CI ≥3) and low comorbidity burden, 3-year OS rates were 88% and 50%, respectively. Lower survival in patients with low HCT-CI was a result of higher relapse risk (54% vs 38%), possibly because of increased percentages of high DRI (37.5% vs 12%) and older median age (68 vs 62 years) in this cohort.
OS was higher in patients whose donor possessed the KIR B/x vs A/A haplotype, with 1- and 3-year OS rates of 94% and 88% vs 69% and 62%, respectively (P = .007). This difference in OS was the result of a significant increase in NRM in patients whose donor possessed the inhibitory KIR A/A vs B/x haplotype (3-year NRM rates of 43% vs 0%, respectively). The higher NRM in patients with a KIR A/A haplotype was the result of increased infectious mortality (disseminated adenovirus, viral pneumonia, fungal pneumonia).
Ixazomib administration and tolerability
All patients received cycle 1 of ixazomib. Only 9 patients received cycle 2 at full dose. Both hematologic and cutaneous toxicities at least possibly related to ixazomib were common, resulting in dose reductions and cycle delays in most patients. The median number of total ixazomib cycles received was 3 (range, 1-12 cycles), with only 1 patient receiving all 12 planned cycles. Nine patients received only 1 cycle. More granular data regarding ixazomib dosing is presented in supplemental Figure 1.
Rash, at least possibly related to ixazomib, was particularly frequent occurring in 19 patients (76%) at median day +32 (range, 17-139 days) after transplant, although distinguishing cutaneous ixazomib-associated drug toxicity with skin-only GVHD was challenging. Reasons for early ixazomib discontinuation included rash (9), cytopenias (7), relapse/progression (4), GVHD (2), diarrhea (1), and arthralgias (1).
Immune reconstitution
Posttransplant immune recovery (absolute lymphocyte count [ALC], T cell, CD4+ T cell, CD8+ T cell, B cell and NK cell) is shown in supplemental Figure 2. The kinetics of immune recovery differs by cell type with recovery of NK cells by day +60, CD8 T cells and B cells by 6 months, with normal CD4+ T cells taking a year or more to recover.
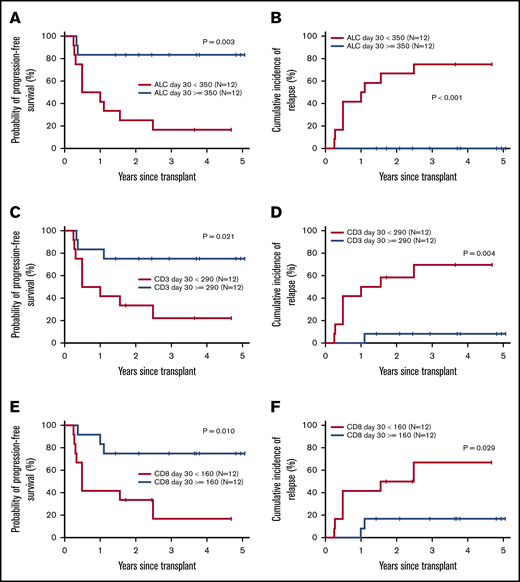
PFS and cumulative incidence of relapse/progression was directly correlated with more rapid immune reconstitution of ALC, CD3+, and CD8+ T cells (Figure 3). Patients with day +30 ALC above the median (≥350/μL) had an estimated 3-year PFS of 83% compared with 17% for patients with low day +30 ALC. Similarly, patients with higher day +30 CD3+ and CD8+ T-cell counts had 3-year PFS rates of 75% and 75%, respectively, compared with 22% and 17%, respectively, in patients with lower day +30 CD3+ and CD8+ counts. This improvement in disease-free survival was the result of a lower 3-year relapse risk in patients with more rapid ALC, CD3+, and CD8+ T-cell recovery (0% vs 75%, 8% vs 69%, and 17% vs 67%, respectively).
Effect of posttransplant lymphocyte recovery on PFS and relapse/progression. Estimates of probability of PFS according to day +30 ALC (A), CD3 T-cell count (C), and CD8+ cytotoxic T-cell count (E). Cumulative incidence of relapse/progression according to day +30 ALC (B), CD3 T-cell count (D), and CD8+ cytotoxic T-cell count (F).
Effect of posttransplant lymphocyte recovery on PFS and relapse/progression. Estimates of probability of PFS according to day +30 ALC (A), CD3 T-cell count (C), and CD8+ cytotoxic T-cell count (E). Cumulative incidence of relapse/progression according to day +30 ALC (B), CD3 T-cell count (D), and CD8+ cytotoxic T-cell count (F).
Discussion
In this study, we report on a prospective trial designed to test the feasibility of substituting the oral PI ixazomib for MMF for posttransplant GVHD prophylaxis after standard Baltimore NMA conditioning for HIDT-PTCy using PBSCs as the source for stem cells. We hypothesized that this might promote improved engraftment, maintain the low GVHD and NRM rates inherent in this regimen, and augment NK cell–mediated cytotoxicity, which is believed to be important in HIDT-associated GVM effects.24-26 Although the study failed to meet our statistically predefined goal of decreasing relapse risk, it did show durable engraftment in all patients, which was unexpected, given the historical experience of 10% graft failure with NMA HIDT-PTCy and a PBSC graft. Furthermore, NRM was low (12% at 3 years posttransplant) confirming the safety of this regimen. Rates of severe aGVHD and moderate-to-severe cGVHD were comparable when compared with a contemporaneous control group at our institution or to Center for International Blood and Marrow Transplant Research (CIBMTR) registry patients receiving HIDT-PTCy using PBSCs.6 In spite of the significant risk of relapse (44% at 3 years posttransplant), OS was favorable (74% at 3 years posttransplant). T-cell and NK-cell immune reconstitution was similar to that described after conventional NMA HIDT-PTCy,27 whereas B-cell immune reconstitution was slower as might be predicted by the effects of proteasome inhibition on B-cell numbers and function.28,29 Faster immune reconstitution of lymphocytes and CD8+ T cells correlated with decreased relapse and improved PFS in our study.
Notably, there were significant challenges with the use of ixazomib posttransplant. Hematologic and cutaneous toxicity, at least possibly related to ixazomib, was common and resulted in cycle delays and dose reductions in most patients. Despite the intention of completing 12 cycles of therapy, the median number of ixazomib cycles given was 3 with only 1 patient able to receive all 12 planned cycles. Rashes, occurring in the first several months posttransplant in approximately 75% of patients were unexpected, and that made it challenging to distinguish drug toxicity from skin-only aGVHD. The major reasons for early discontinuation of ixazomib were rashes, cytopenias, and relapse/progression (in order of frequency). However, severe nonhematologic toxicity was uncommon, and both cutaneous and hematologic toxicities were easily managed with appropriate dose modifications.
Over the last decade, there has been considerable interest in the use of PIs for GVHD prophylaxis. Although current GVHD prevention regimens exclusively target T cells, PIs possess unique immunomodulatory effects that target both T cells and dendritic cells, the latter of which plays a critical role in the generation and maintenance of alloreactivity.30 In the setting of HLA-mismatched murine transplant studies, early posttransplant use of bortezomib results in significant protection of mice from lethal aGVHD, while retaining the potent GVM effects of cytotoxic T cells.11 Furthermore, a clinical study that evaluated the addition of early posttransplant bortezomib to conventional GVHD prophylaxis in the context of reduced-intensity HLA-mismatched unrelated donor transplantation resulted in promising transplant outcomes.13 Notably, however, the large randomized Blood and Marrow Transplant Clinical Trials Network 1203 study failed to show a benefit for bortezomib in the setting of reduced-intensity HLA-matched donor transplantation when compared with standard conventional GVHD prophylaxis.31
Contrary to the beneficial effects of early bortezomib administration in murine transplant experiments, delayed or continuous use of bortezomib posttransplant in mice has led to aggravation of acute gastrointestinal GVHD, leading to enhanced lethality.32 This paradoxical effect of delayed posttransplant PI administration seems to be driven by increased proinflammatory cytokine release, an effect that can be negated by previous depletion of CD4+ but not CD8+ T cells from the murine allograft.32 Similarly, administering PTCy before administering PI has been shown to prevent the PI-associated increases in interleukin 1β (IL-1β) and tumor necrosis factor-α (TNF-α) release as well as the paradoxical aggravation of GVHD.15 The combination of PTCy and ixazomib was tested in a phase 1/2 clinical study in the context of reduced-intensity matched related or unrelated donor transplantation with promising results.33
Although overall NRM in this study was low (12% at 3 years posttransplant), NRM seemed higher in patients who received transplants with donors who expressed the more inhibitory KIR A/A haplotype (43% vs 0%). Given the critical importance of the immunoproteasome for CD8+ T-cell–mediated immune responses against intracellular infections,34,35 we speculate that more potent NK cells resulting from an activating KIR genotype may be important in controlling early infections posttransplant when using a PI-based GVHD prophylaxis regimen. To this point, an activating KIR B/x haplotype has been associated with lower infectious risk in the context of both hematopoietic and solid organ transplantation.36,37 Furthermore, activating KIR genes have also been associated with a lower incidence of CMV infection after allogenic transplantation.38-40
The finding of lower relapse/progression and improved PFS in patients with higher day +30 lymphocyte recovery and CD8+ T-cell recovery in particular, is also noteworthy and deserving of additional investigation. Of note, we found no similar association of pretransplant ALC with PFS or relapse/progression. Furthermore, there was no correlation of either pre- or posttransplant ALC with underlying disease. In previous studies, early ALC recovery has been shown to predict lower relapse and improved PFS after matched related and unrelated donor transplantation41-43 as well as in the context of ATG-based, T-replete HIDT using the Beijing protocol.44
There was a notable discrepancy between 3-year OS and PFS in this study, which were estimated at 74% and 44%, respectively. As of the data cutoff for this study, 7 of 10 patients with posttransplant relapse/progression remain alive and in CR with a median of 16 months (range, 8-46 months) of follow-up. Since that time, all 7 patients remain in CR with at least 16 months of follow-up (median, 24 months), suggesting a favorable postrelapse survival (PRS, ∼70%) in this study. Historically, our group has shown disappointing PRS after HIDT-PTCy (1-year PRS, 17%) when compared with similar patients after matched related and unrelated donor transplantation.1,45 However, small numbers of patients and the heterogeneity of the patient population make definitive comparisons of PRS with historical controls challenging.
Substituting the oral PI ixazomib for MMF after NMA PBSC-based HIDT-PTCy seems to be a safe and effective alternative GVHD prophylaxis strategy. Although our study failed to show an impact on relapse/progression risk, potential benefits include universal engraftment and favorable 3-year survival, associated with similar rates of GVHD, NRM, and immune reconstitution. Given the need for ixazomib dose reductions in multiple patients because of hematologic and cutaneous toxicity, further investigation into the optimal dosing strategy is warranted. Although the overall NRM in this study was low (12% at 3 years posttransplant), the increased NRM as a result of early infectious mortality in recipients with KIR A/A haplotype donors is cautionary and deserves further investigation. However, given the older age and high comorbidity status of this patient cohort, the low NRM in this study seems favorable, which leads to a promising 74% 3-year posttransplant OS rate.
Requests for data sharing should be e-mailed to Scott R. Solomon at ssolomon@bmtga.com.
Acknowledgments
The authors are grateful to all the clinicians and support staff at the Blood and Marrow Transplant Program at Northside Hospital Cancer Institute for their excellent care of our transplant patients.
This study was supported by Takeda Oncology (previously Millennium Pharmaceuticals), which also provided the study drug ixazomib.
Authorship
Contribution: S.R.S. designed the research; S.R.S., M.S., L.E.M., H.K.H., and A.B. treated patients; S.B. and K.C.J. collected the data; S.R.S. and X.Z. analyzed and interpreted the data; X.Z. performed the statistical analysis; S.R.S. wrote the manuscript; and all authors revised and approved the final version of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Scott R. Solomon, Northside Hospital Cancer Institute, 5670 Peachtree-Dunwoody Road NE, Suite 1000, 10th Floor, Atlanta, GA 30342; e-mail: ssolomon@bmtga.com.
References
Author notes
The full-text version of this article contains a data supplement.