Key Points
BR is an effective induction regimen in both transplant-eligible and -ineligible MCL patients, with favorable response rates and outcomes.
BR may not be an optimal induction regimen in MCL with high-risk MIPI, Ki-67 ≥50%, and blastoid/pleomorphic histology.
Abstract
Rituximab-containing chemotherapy regimens constitute standard first-line therapy for mantle cell lymphoma (MCL). Since June 2013, 190 patients ≥18 years of age with MCL in British Columbia have been treated with bendamustine and rituximab (BR). The overall response rate to BR was 88% (54% complete response). Of these, 61 of 89 patients (69%) aged ≤65 years received autologous stem cell transplantation and 141 of 190 patients (74%) from the entire cohort received maintenance rituximab. Twenty-three patients (12%) had progressive disease, associated with high risk per the Mantle Cell Lymphoma International Prognostic Index (MIPI), Ki-67 ≥50%, and blastoid/pleomorphic histology. Outcomes were compared with a historical cohort of 248 patients treated with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP; January 2003 to May 2013). Treatment with BR was associated with significant improvements in progression-free survival (PFS), but not overall survival (OS), compared with R-CHOP in the whole cohort (3-year PFS, 66% BR vs 51% R-CHOP, P = .003; 3-year OS, 73% BR vs 66% R-CHOP, P = .054) and in those >65 years of age (3-year PFS, 56% BR vs 35% R-CHOP, P = .001; 3-year OS, 64% BR vs 55% R-CHOP, P = .063). Outcomes in transplanted patients were not statistically significantly different compared with R-CHOP (3-year PFS, 85% BR vs 76% R-CHOP, P = .135; 3-year OS, 90% BR vs 88% R-CHOP, P = .305), although in multivariate analyses, treatment with BR was associated with improved PFS (hazard ratio, 0.40 [95% confidence interval, 0.17-0.94]; P = .036) but not OS. BR is an effective first-line option for most patients with MCL, however, outcomes are suboptimal for those with high-risk features and further studies integrating novel agents are warranted.
Introduction
Mantle cell lymphoma (MCL) is an incurable B-cell non-Hodgkin lymphoma associated with poor outcomes. First-line treatment regimens incorporate cytotoxic chemotherapeutic agents and rituximab, but there is no single standard of care.1 For over 2 decades, consolidative high-dose chemotherapy and autologous stem cell transplantation (ASCT) have been used in responding young, fit patients given an association with improved progression-free survival (PFS) and overall survival (OS).2,3 Maintenance rituximab (MR) after first-line immunochemotherapy, with or without consolidative ASCT, is also associated with improved outcomes, although it is unclear whether the choice of particular first-line immunochemotherapy regimens may influence the effectiveness of MR.4-6
In British Columbia (BC), between January 2003 and May 2013, the combination of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) was the preferred induction regimen. In June 2013, based on results from the STiL-1 trial demonstrating improved response rates and prolonged PFS with bendamustine and rituximab (BR) compared with R-CHOP in the first-line setting, including in the subgroup with MCL, BR became the recommended first-line therapy for all patients with MCL regardless of age.7 The aim of this study was to assess the efficacy of BR as an induction regimen in all patients with MCL, regardless of candidacy for subsequent ASCT or MR.
Methods
Patient, disease, and treatment characterization
Patients ≥18 years of age with biopsy-proven MCL treated with first-line BR prior to December 2018 were identified in the BC Cancer clinical and pathology databases, which have been previously described,8 as well as the Leukemia/Bone Marrow Transplant (L/BMT) Program of BC database. Prospective data collection and entry was performed for these databases, and selected additional study-specific information was collected and added retrospectively for this analysis. Protocols and policies for MCL management are set centrally via BC Cancer and followed with care delivered in a standard manner to all such patients in BC. Patient and disease characteristics, as well as follow-up data, were verified using the Cancer Agency Information System. Pathology was centrally reviewed at the time of diagnosis, and only cases positive for cyclin D1 by immunohistochemistry and/or translocation (11; 14) by fluorescence in situ hybridization were included. Bone marrow biopsies and contrast-enhanced computed tomography (CT) scans of the chest, abdomen, and pelvis were routinely performed to determine Ann Arbor stage. Gastrointestinal endoscopies and positron-emission tomography (PET) scans were not routinely performed for staging purposes.
Treatment data, including protocol delivered, date of first cycle, number of cycles, high-dose chemotherapy, and maintenance rituximab were verified using the BC Cancer Provincial Pharmacy database. Patients were included if they had received at least 1 cycle of BR and had not received any other prior systemic treatments for MCL. Prior observation for asymptomatic MCL and/or local therapy for localized disease, including radiotherapy or surgery (ie, splenectomy), were permitted. Patients diagnosed with MCL after June 2013 but not treated with BR, including on clinical trials, were excluded; reasons for not choosing BR were collected and described.
BR was administered IV at standard doses every 4 weeks for up to 6 cycles.7,9 Response to BR was retrospectively assessed through review of end-of-treatment CT scan and bone marrow biopsy reports, although the latter were not consistently performed in all patients. For consistency, information from the few available PET scans was not incorporated into this assessment. Patients were categorized as responders if they achieved a complete or partial response (CR or PR, respectively) by CT criteria,10 or as nonresponders if they developed progressive disease (PD) during first-line treatment with BR or within 3 months of the last cycle.
Fit patients who met specific L/BMT Program of BC–predefined disease response and organ function criteria and who responded to BR were considered eligible for high-dose carmustine, etoposide, cytarabine, and melphalan (BEAM) followed by ASCT. During the early period of this study, age restrictions were in place to consider in general only those age ≤65 years for ASCT; however, over time selected fit patients >65 years could proceed with ASCT depending on their fitness. In BC, 375 mg/m2 MR IV weekly for 4 weeks at 3 and 9 months (total 8 doses) was given only to responding patients post-ASCT starting in 2004.11 Since mid-2012, 375 mg/m2 MR IV every 3 months for 2 years (total 8 doses) has been given to all patients responding to first-line chemoimmunotherapy, with or without consolidative ASCT, based on prior studies establishing benefit of MR after other first-line regimens.4
Historical comparison cohort
Characteristics and outcomes in BR-treated patients were compared with an earlier cohort of MCL patients from BC consecutively and uniformly treated with R-CHOP between January 2003 and May 2013. R-CHOP was typically given every 3 weeks for 6 to 8 cycles. The indications for consolidative ASCT and MR were similar to those applied during the BR era. The same methodology described in the previous section was used to identify and describe the comparison cohort.
Statistical analysis
Group comparisons were performed with the χ2 test or Student t test. PFS was defined as the time from the date of initiation of first-line systemic therapy to date of MCL relapse/progression or death from any cause. OS was defined as the time from the date of initiation of first-line systemic therapy to death from any cause. PFS and OS were estimated using the Kaplan-Meier method, and group comparisons were made with the log-rank test.
Hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated in univariate and multivariate Cox proportional hazards models. Baseline patient or disease characteristics with P < .1 in univariate analysis were entered into the multivariate models, which used a backward likelihood ratio selection method, and results with P < .05 were considered statistically significant in the final models. In multivariate models exploring the impact of the first-line immunochemotherapy regimen adjusted for other variables, BR vs R-CHOP was included regardless of significance in univariate analysis. To account for guarantee time bias in analyses exploring the impact of receipt of MR on outcomes, univariate landmark analyses were performed by excluding patients with PD, death, or loss to follow-up within 18 months of BR/R-CHOP initiation. This cutoff was chosen to ensure that all eligible patients had completed first-line immunochemotherapy, ASCT, response assessment, and at least 1 cycle of MR. Additionally, MR was analyzed as a time-dependent covariate in the multivariate models. Coefficients of all variables were calculated under the assumption of a constant HR except the time-dependent adjustment term. All statistical analyses were performed with SPSS version 14.0. The study was approved by the University of BC/BC Cancer Research Ethics Board.
Results
Baseline and response characteristics
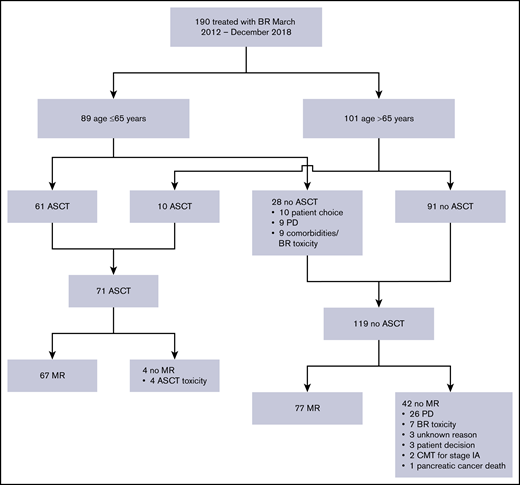
In total, 190 patients with MCL treated with first-line BR prior to December 2018 were identified. This included 3 patients who received BR prior to the introduction of the BR policy in June 2013. An additional 60 patients diagnosed with MCL between June 2013 and December 2018 were identified who were not treated with BR for the following reasons: 28 observation; 14 palliative therapies; 11 R-CHOP plus or minus dexamethasone, cytarabine, cisplatin (DHAP) due to physician preference; 7 treatment refused/unknown. Following BR, 61 of 89 patients (69%) aged ≤65 years received ASCT, and 144 of 190 patients (76%) from the entire cohort received MR. The most frequent reasons for not proceeding to ASCT and/or MR included PD and treatment toxicity (Figure 1).
Identification of the BR-treated cohort and description of subsequent therapies. CMT, combined modality therapy.
Identification of the BR-treated cohort and description of subsequent therapies. CMT, combined modality therapy.
A historical comparison cohort of 248 patients treated with R-CHOP between January 2003 and May 2013 was identified. Baseline characteristics were generally similar between patients treated with BR compared with R-CHOP (Table 1). A similar proportion of patients underwent ASCT in both groups (37% BR vs 35% R-CHOP; P = .561). ASCT was performed after a median of 263 days (range, 179-363 days) from the first dose of BR compared with 185 days (range, 109-437 days) from the first dose of R-CHOP (P < .001). More patients in the BR-treated cohort received MR, partly due to the fact that the routine use of MR for all patients was not introduced until 2013. The MR strategy of 4 weekly rituximab doses for 2 cycles was used exclusively after ASCT; all 109 patients who received this protocol had a prior ASCT (31 BR, 78 R-CHOP). A total of 67 of 71 BR patients received MR post-ASCT (32 weekly rituximab ×4 doses for 2 cycles, 35 rituximab every 3 months for 8 cycles), whereas 79 of 86 R-CHOP patients received MR post-ASCT (78 weekly rituximab ×4 doses for 2 cycles, 1 rituximab every 3 months for 8 cycles; P < .001).
Baseline characteristics
| Characteristic . | All BR patients . | Patients ≤65 y of age at chemotherapy . | Patients >65 y of age at chemotherapy . | ||||
|---|---|---|---|---|---|---|---|
| BR, n = 190 . | BR, n = 89 . | R-CHOP, n = 142 . | P . | BR, n = 101 . | R-CHOP, n = 106 . | P . | |
| Median age (range), y | 66 (37-90) | 58 (37-65) | 57 (31-65) | .282 | 73 (63-90) | 72 (63-87) | .631 |
| Male | 121 (64) | 55 (62) | 110 (78) | .010 | 66 (65) | 74 (70) | .493 |
| Performance status >1 | 36/158 (23) | 16/77 (21) | 23/136 (17) | .483 | 20/81 (25) | 31/93 (33) | .212 |
| Elevated LDH | 40/140 (29) | 24/71 (34) | 45/127 (35) | .817 | 16/69 (23) | 31/83 (37) | .060 |
| Ann Arbor stage III/IV | 166/180 (92) | 79/84 (94) | 129/142 (91) | .390 | 87/96 (91) | 95/103 (93) | .685 |
| Marrow/blood involvement | 132 (69) | 65 (73) | 113 (80) | .250 | 67 (66) | 73 (69) | .697 |
| B symptoms | 41/180 (23) | 22/84 (26) | 46/142 (32) | .326 | 19/96 (20) | 26/103 (25) | .358 |
| Largest mass ≥5 cm | 52/109 (48) | 24/52 (46) | 60/123 (49) | .751 | 28/57 (49) | 46/85 (54) | .559 |
| MIPI | |||||||
| High risk | 21/125 (17) | 9/64 (14) | 14/122 (12) | .519 | 12/61 (20) | 31/74 (42) | .007 |
| Intermediate risk | 38/125 (30) | 14/64 (22) | 20/122 (16) | 24/61 (39) | 28/74 (38) | ||
| Low risk | 66/125 (53) | 41/64 (64) | 88/122 (72) | 25/61 (41) | 15/74 (20) | ||
| Blastoid/pleomorphic | 13 (7) | 7 (8) | 14 (10) | .608 | 6 (6) | 15 (14) | .051 |
| Median Ki67 (range) | 30 (5-90) | 25 (5-90) | 25 (1-99) | .317 | 30 (5-90) | 33 (2-100) | .192 |
| Ki67 ≥30% | 92/171 (54) | 36/80 (45) | 42/88 (48) | .723 | 56/91 (62) | 34/64 (53) | .296 |
| Ki67 ≥50% | 45/171 (26) | 17/80 (21) | 26/88 (30) | .218 | 28/91 (31) | 22/64 (34) | .636 |
| Prior observation | 21 | 7 (8) | 6 (4) | .243 | 14 (14) | 6 (6) | .046 |
| Prior local therapy | 6 | 1 (1) | 3 (3) | 1.0 | 5 (5) | 1 (1) | .112 |
| Median no. of chemotherapy cycles (range) | 6 (1-6) | 6 (1-6) | 6 (1-8) | .127 | 6 (1-6) | 6 (1-8) | .890 |
| ≥6 cycles of chemotherapy | 154 (81) | 78 (88) | 123 (87) | .822 | 76 (75) | 82 (77) | .721 |
| ASCT | 71 (37) | 61 (69) | 81 (57) | .081 | 10 (10) | 5 (5) | .150 |
| Rituximab maintenance | 141 (74) | 72 (81) | 80 (56) | <.001 | 72 (71) | 25 (24) | <.001 |
| Rituximab maintenance protocol | |||||||
| Weekly × 4 doses ×2 cycles | 31/141 (22) | 27/72 (37) | 73/80 (91) | <.001 | 5/72 (7) | 5/25 (20) | .064 |
| Every 3 mo ×8 cycles | 110/141 (78) | 45/72 (63) | 7/80 (9) | 67/72 (93) | 20/25 (80) | ||
| Characteristic . | All BR patients . | Patients ≤65 y of age at chemotherapy . | Patients >65 y of age at chemotherapy . | ||||
|---|---|---|---|---|---|---|---|
| BR, n = 190 . | BR, n = 89 . | R-CHOP, n = 142 . | P . | BR, n = 101 . | R-CHOP, n = 106 . | P . | |
| Median age (range), y | 66 (37-90) | 58 (37-65) | 57 (31-65) | .282 | 73 (63-90) | 72 (63-87) | .631 |
| Male | 121 (64) | 55 (62) | 110 (78) | .010 | 66 (65) | 74 (70) | .493 |
| Performance status >1 | 36/158 (23) | 16/77 (21) | 23/136 (17) | .483 | 20/81 (25) | 31/93 (33) | .212 |
| Elevated LDH | 40/140 (29) | 24/71 (34) | 45/127 (35) | .817 | 16/69 (23) | 31/83 (37) | .060 |
| Ann Arbor stage III/IV | 166/180 (92) | 79/84 (94) | 129/142 (91) | .390 | 87/96 (91) | 95/103 (93) | .685 |
| Marrow/blood involvement | 132 (69) | 65 (73) | 113 (80) | .250 | 67 (66) | 73 (69) | .697 |
| B symptoms | 41/180 (23) | 22/84 (26) | 46/142 (32) | .326 | 19/96 (20) | 26/103 (25) | .358 |
| Largest mass ≥5 cm | 52/109 (48) | 24/52 (46) | 60/123 (49) | .751 | 28/57 (49) | 46/85 (54) | .559 |
| MIPI | |||||||
| High risk | 21/125 (17) | 9/64 (14) | 14/122 (12) | .519 | 12/61 (20) | 31/74 (42) | .007 |
| Intermediate risk | 38/125 (30) | 14/64 (22) | 20/122 (16) | 24/61 (39) | 28/74 (38) | ||
| Low risk | 66/125 (53) | 41/64 (64) | 88/122 (72) | 25/61 (41) | 15/74 (20) | ||
| Blastoid/pleomorphic | 13 (7) | 7 (8) | 14 (10) | .608 | 6 (6) | 15 (14) | .051 |
| Median Ki67 (range) | 30 (5-90) | 25 (5-90) | 25 (1-99) | .317 | 30 (5-90) | 33 (2-100) | .192 |
| Ki67 ≥30% | 92/171 (54) | 36/80 (45) | 42/88 (48) | .723 | 56/91 (62) | 34/64 (53) | .296 |
| Ki67 ≥50% | 45/171 (26) | 17/80 (21) | 26/88 (30) | .218 | 28/91 (31) | 22/64 (34) | .636 |
| Prior observation | 21 | 7 (8) | 6 (4) | .243 | 14 (14) | 6 (6) | .046 |
| Prior local therapy | 6 | 1 (1) | 3 (3) | 1.0 | 5 (5) | 1 (1) | .112 |
| Median no. of chemotherapy cycles (range) | 6 (1-6) | 6 (1-6) | 6 (1-8) | .127 | 6 (1-6) | 6 (1-8) | .890 |
| ≥6 cycles of chemotherapy | 154 (81) | 78 (88) | 123 (87) | .822 | 76 (75) | 82 (77) | .721 |
| ASCT | 71 (37) | 61 (69) | 81 (57) | .081 | 10 (10) | 5 (5) | .150 |
| Rituximab maintenance | 141 (74) | 72 (81) | 80 (56) | <.001 | 72 (71) | 25 (24) | <.001 |
| Rituximab maintenance protocol | |||||||
| Weekly × 4 doses ×2 cycles | 31/141 (22) | 27/72 (37) | 73/80 (91) | <.001 | 5/72 (7) | 5/25 (20) | .064 |
| Every 3 mo ×8 cycles | 110/141 (78) | 45/72 (63) | 7/80 (9) | 67/72 (93) | 20/25 (80) | ||
Values in the table body are shown as n (%) or n/N (%), unless otherwise stated in row headings.
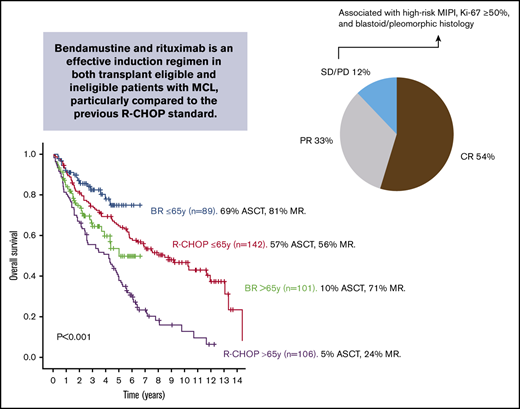
In total, 188 of 190 BR-treated patients were evaluable for response. Two patients, both over the age of 75 years, died of infectious complications during immunochemotherapy and were considered not evaluable. One hundred three of 188 patients (54%) achieved a CR and 62 of 188 (33%) a PR, for an 88% overall response rate. However, 23 of 188 patients progressed during or within 3 months of receiving BR, which included 4 patients with stable disease on end-of-treatment response assessment but who progressed shortly after. Response rates were similar between patients ≤65 years of age (CR, 54 of 89 [61%]; PR, 25 of 89 [28%]; PD, 10 of 89 [11%]) and those >65 years of age (CR, 49 of 99 [50%]; PR, 37 of 99 [37%); PD, 13 of 99 [13%]). Clinical characteristics associated with nonresponse (PD) following BR include poor performance status, elevated lactate dehydrogenase (LDH), high-risk Mantle Cell Lymphoma International Prognostic Index (MIPI), highly proliferative MCL (Ki-67 ≥50%), and blastoid/pleomorphic histology. (Table 2)
Characteristics of BR nonresponders vs responders
| Characteristic . | Responders, n = 165 . | Nonresponders, n = 23 . | P . |
|---|---|---|---|
| CR, n = 103; PR, n = 62 . | PD, n = 23 . | ||
| Age >65 y at BR initiation | 79 (48) | 10 (44) | .692 |
| Male | 105 (64) | 15 (65) | .882 |
| Performance status >1 | 21/135 (16) | 13/21 (62) | <.001 |
| Elevated LDH | 31/124 (25) | 9/15 (60) | .005 |
| Ann Arbor stage III/IV | 143/156 (92) | 22/23 (96) | .506 |
| Marrow/blood involvement | 115 (70) | 16 (70) | .990 |
| B symptoms | 32/156 (21) | 8/23 (35) | .125 |
| Largest mass ≥5 cm | 46/96 (48) | 5/12 (42) | .683 |
| MIPI | |||
| High risk | 13/109 (12) | 8/15 (53) | <.001 |
| Intermediate risk | 33/109 (30) | 4/15 (27) | |
| Low risk | 63/109 (58) | 3/15 (20) | |
| Blastoid/pleomorphic | 7 (4) | 6 (26) | <.001 |
| Median Ki67 (range) | 30 (5-90) | 50 (5-90) | .003 |
| Ki67 ≥30% | 79/149 (53) | 12/21 (57) | .723 |
| Ki67 ≥50% | 32/149 (22) | 12/21 (57) | <.001 |
| Prior observation | 18 (11) | 3 (13) | .726 |
| Prior local therapy | 6 (4) | 0 (0) | 1.0 |
| Median no. of chemotherapy cycles (range) | 6 (2-6) | 3 (1-6) | <.001 |
| ≥6 cycles of chemotherapy | 146 (87) | 8 (35) | <.001 |
| Characteristic . | Responders, n = 165 . | Nonresponders, n = 23 . | P . |
|---|---|---|---|
| CR, n = 103; PR, n = 62 . | PD, n = 23 . | ||
| Age >65 y at BR initiation | 79 (48) | 10 (44) | .692 |
| Male | 105 (64) | 15 (65) | .882 |
| Performance status >1 | 21/135 (16) | 13/21 (62) | <.001 |
| Elevated LDH | 31/124 (25) | 9/15 (60) | .005 |
| Ann Arbor stage III/IV | 143/156 (92) | 22/23 (96) | .506 |
| Marrow/blood involvement | 115 (70) | 16 (70) | .990 |
| B symptoms | 32/156 (21) | 8/23 (35) | .125 |
| Largest mass ≥5 cm | 46/96 (48) | 5/12 (42) | .683 |
| MIPI | |||
| High risk | 13/109 (12) | 8/15 (53) | <.001 |
| Intermediate risk | 33/109 (30) | 4/15 (27) | |
| Low risk | 63/109 (58) | 3/15 (20) | |
| Blastoid/pleomorphic | 7 (4) | 6 (26) | <.001 |
| Median Ki67 (range) | 30 (5-90) | 50 (5-90) | .003 |
| Ki67 ≥30% | 79/149 (53) | 12/21 (57) | .723 |
| Ki67 ≥50% | 32/149 (22) | 12/21 (57) | <.001 |
| Prior observation | 18 (11) | 3 (13) | .726 |
| Prior local therapy | 6 (4) | 0 (0) | 1.0 |
| Median no. of chemotherapy cycles (range) | 6 (2-6) | 3 (1-6) | <.001 |
| ≥6 cycles of chemotherapy | 146 (87) | 8 (35) | <.001 |
Values in the table body are shown as n (%) or n/N (%), unless otherwise stated in row headings. Response assessment was not performed in 2 patients who were excluded.
Stem cell collection rates post-BR have been previously described and were not the focus of the current study.12 Initial failure of stem cell collection, defined as yield <1 × 106 CD34+ cells per kilogram on apheresis day 1, was observed in 45% of BR vs 10% R-CHOP–treated patients (P < .001), primarily when granulocyte colony-stimulating factor (G-CSF) alone was used for mobilization. Subsequent strategies, such as delaying stem cell collection 2 to 3 months after BR, G-CSF plus cyclophosphamide mobilization, and/or the use of plerixafor reduced the overall rate of stem cell collection failure (defined as <2 × 106 CD34+ cells per kilogram after all apheresis days) after BR to 11%.12
Survival outcomes
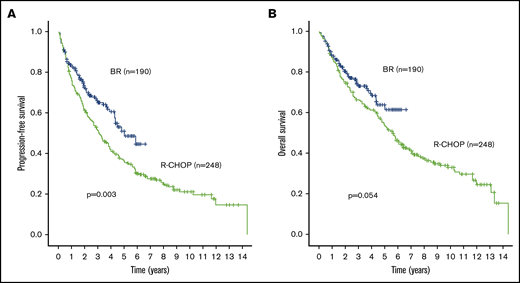
With a median follow-up time in living patients of 3.1 years (range, 0.4-6.6 years) after initiation of BR, the 3-year PFS of the whole cohort was 66% (95% CI, 65.2-66.8) and 3-year OS was 73% (95% CI, 72.3-73.7). The historical cohort had longer median follow-up time of 8.1 years (range, 0.8-13.7 years) after initiation of R-CHOP with 3-year PFS of 51.2% (95% CI, 50.6-51.8; P = .003 compared with BR) (Figure 2A) and 3-year OS of 66.2% (95% CI, 65.7-66.7; P = .054 compared with R-CHOP) (Figure 2B).
Outcome comparisons between the entire BR (blue line) and R-CHOP (green line) cohorts. (A) PFS. (B) OS.
Outcome comparisons between the entire BR (blue line) and R-CHOP (green line) cohorts. (A) PFS. (B) OS.
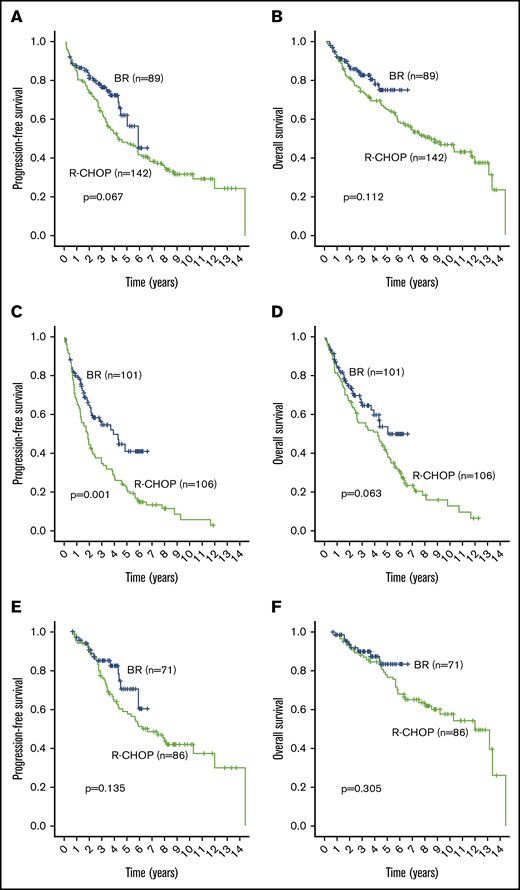
In patients ≤65 years of age, 3-year outcomes in the BR-treated cohort were not significantly different compared with the historical cohort. Specifically, the 3-year PFS was 76.5% BR (95% CI, 75.1-78) vs 63.6% R-CHOP (95% CI, 62.7-64.5) (P = .067) (Figure 3A). The 3-year OS was 82.4% BR (95% CI, 81.2-83.6) vs 74.3% R-CHOP (95% CI, 73.6-75) (P = .112) (Figure 3B).
Outcome comparisons between subgroups of the BR (blue line) and R-CHOP (green line) cohorts. (A) PFS in patients ≤65 years of age. (B) OS in patients ≤65 years of age. (C) PFS in patients >65 years of age. (D) OS in patients >65 years of age. (E) PFS in ASCT patients regardless of age. (F) OS in ASCT patients regardless of age.
Outcome comparisons between subgroups of the BR (blue line) and R-CHOP (green line) cohorts. (A) PFS in patients ≤65 years of age. (B) OS in patients ≤65 years of age. (C) PFS in patients >65 years of age. (D) OS in patients >65 years of age. (E) PFS in ASCT patients regardless of age. (F) OS in ASCT patients regardless of age.
In patients >65 years of age, 3-year outcomes in the BR-treated cohort were statistically significantly improved compared with the R-CHOP cohort. Specifically, the 3-year PFS was 56.4% BR (95% CI, 54.5-58.3) vs 34.7% R-CHOP (95% CI, 33.2-36.2) (P = .001) (Figure 3C). The 3-year OS was 64.3% BR (95% CI, 62.6-66) vs R-CHOP 55.4% (95% CI, 54.1-56.6) (P = .063) (Figure 3D).
In the subgroup of patients who underwent ASCT, 3-year outcomes were not significantly different between the 2 cohorts. Specifically, the 3-year PFS was 85.1% BR (95% CI, 83.8-86.4) vs 76.2% R-CHOP (95% CI, 75.1-77.3) (P = .135) (Figure 3E). The 3-year OS was 90% BR (95% CI, 88.9-91.1) vs 88.2% R-CHOP (95% CI, 87.4-89) (P = .305) (Figure 3F). Similarly, PFS and OS calculated from the time of ASCT were not statistically different between the BR and R-CHOP cohorts. Specifically, the 3-year post-ASCT PFS was 82.2% BR (95% CI, 80.3-84.1) vs 67.8% R-CHOP (95% CI, 66.4-69.1) (P = .22). The 3-year post-ASCT OS was 87.2% BR (95% CI, 85.5-88.9) vs 84.5% R-CHOP (95% CI, 83.5-85.4) (P = .37).
There were 53 deaths in the BR cohort: 4 toxic deaths (acute respiratory distress syndrome during maintenance rituximab, abdominal sepsis during BR, toxic megacolon during BR, abdominal sepsis post-ASCT), 13 unrelated deaths of which 10 were metastatic solid malignancies, and 36 PD. The majority of these deaths (n = 36) occurred in patients over the age of 65 years: 4 toxic deaths, 10 unrelated of which 8 were metastatic solid malignancies, and 22 PD.
Univariate and multivariate analyses
In the entire cohort of BR-treated patients, age >65 years, Eastern Cooperative Oncology Group (ECOG) performance status >1, elevated LDH, high-risk MIPI, blastoid/pleomorphic morphology, and Ki67 ≥50% were associated with an inferior PFS and OS in univariate analysis (supplemental Table 1). In multivariate analysis excluding MR, ECOG performance status >1 and Ki67 ≥50% were associated with inferior PFS and OS, whereas elevated LDH was associated with inferior PFS only (supplemental Table 1). In the 152 R-CHOP patients with available Ki67 data, Ki67 >30% was not associated with PFS (HR, 1.18 [95% CI, 0.81-1.71]; P = .390) or OS (HR, 1.42 [95% CI, 0.94-2.14]; P = .092). Similarly, Ki67 >50% was not associated with PFS (HR, 0.93 [95% CI, 0.62-1.40]; P = .723) or OS (HR, 1.12 [95% CI, 0.72-1.73]; P = .615).
Treatment with BR was associated with improved PFS (HR, 0.65 [95% CI, 0.49-0.86]; P = .003) but not OS (HR, 0.73 [95% CI, 0.53-1.01]; P = .055) compared with R-CHOP in univariate analysis (supplemental Table 4). In multivariate analysis, including only significant baseline patient/disease variables as well as immunochemotherapy regimen (but excluding MR due to substantial selection bias), treatment with BR was also associated with favorable PFS (HR, 0.58 [95% CI, 0.39-0.86]; P = .007) and OS (HR, 0.49 [95% CI, 0.29-0.84]; P = .010) (supplemental Table 4). The PFS effect was largely restricted to the subgroup of patients >65 years (supplemental Tables 5 and 6). Treatment with BR was not associated with favorable PFS or OS when MR was included in the multivariate models (supplemental Tables 4-6). In patients who received ASCT, treatment with BR was associated with improved PFS but not OS in multivariate analyses including and excluding MR (supplemental Table 7).
Receipt of MR was associated with improved PFS and/or OS in univariate (unadjusted) and multivariate analyses in BR-treated patients and the various subgroups shown in the supplemental Data. Univariate landmark analyses did not consistently show an association between MR and improved outcomes. In the 146 patients who received MR after ASCT, MR strategy did not impact PFS (P = .543), post-ASCT PFS (P = .328), OS (P = .657), or post-ASCT OS (P = .562) in crude analysis or after adjusting for BR vs R-CHOP (data not shown).
Discussion
In this retrospective population-based analysis, BR was an effective induction regimen for both transplant-eligible and -ineligible patients with MCL, with significant improvements in PFS, but not OS, compared with R-CHOP in the whole cohort and in the subset of patients >65 years of age. In patients ≤65 years of age, ASCT was feasible after BR although differences in PFS and OS were not statistically different compared with R-CHOP.
We observed progressive disease during or within 3 months of BR in 12% of patients as well as a sharp drop in the PFS curve within the first year after BR initiation, suggesting that BR may not be an optimal induction regimen across all patients with MCL. In particular, early BR failure was more common in those with biologically aggressive (ie, blastoid/pleomorphic morphology) or highly proliferative disease. In our BR cohort, a Ki67 cutoff ≥50% was significantly associated with worse outcomes compared with the ≥30% cutoff that has been identified with other immunochemotherapy regimens.13 However, similar to other series, Ki67 was not collected in all patients and other relevant molecular prognostic factors such as TP53 mutations, 17p deletions, and other markers of genomic complexity were not available.14 Such high-risk patients may benefit from alternative strategies, including those that incorporate more intensive cytarabine-containing regimens and/or the inclusion of novel agents, although it is unknown whether these treatment modifications may be sufficient to overcome adverse biology.15
Based on our data, BR appears to be associated with outcomes that are not statistically different from those observed with R-CHOP in patients ≤65 years of age (Figure 3A-B). The European MCL Younger study showed alternating R-CHOP/R-DHAP followed by high-dose cytarabine and total-body irradiation (TBI) containing conditioning regimen and ASCT was associated with longer time to treatment failure compared with R-CHOP induction followed by high-dose cyclophosphamide/TBI conditioning and ASCT, although there was no improvement in OS.16 However, the alternating regimen of R-CHOP/R-DHAP has never been compared with BR. The SWOG 1106 study compared R-HyperCVAD (rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone), a different intensive cytarabine-containing regimen, with BR in transplant-eligible patients. Although this study was ultimately underpowered as it was terminated early due to excess toxicity in the intensive arm, there were no differences in long-term outcomes between the regimens.17 Other cytarabine-based regimens (R-DHAP, R-BAC [rituximab, bendamustine, cytarabine], BR/CR) have been evaluated in phase 2 trials, but a true OS advantage from the inclusion of cytarabine in the first-line therapy of MCL has never been demonstrated.5,18,19 Whether BR and ASCT achieve outcomes as good as those with high-dose cytarabine-containing regimens and ASCT can only be determined with a well-powered, randomized trial.
We noted a high proportion (45%) of stem cell collection failure on day 1 of apheresis after BR. All stem cell collections in BC are performed in a single center through the Apheresis Program of BC, where typically G-CSF alone is used for stem cell mobilization for ASCT. However, after the introduction of additional strategies, 89% of BR patients ultimately had successful collections. Other MCL induction regimens are also associated with reduced stem cell collection rates. For example, in the European MCL Younger trial, successful collection (defined as ≥2 × 106 CD34+ cells per kilogram) was achieved in 85% of controls vs 66% of the cytarabine group.16 Stem cell mobilization and collection after BR induction for MCL is further detailed in a separate report by our group.12
Our study is limited by its retrospective, nonrandomized design as well as the relatively short follow-up time in the BR subgroup. The broad availability of MR for all patients with MCL after 2012 may have contributed to the favorable outcomes in the BR cohort, although whether MR truly improves PFS or OS after BR remains unknown.6 There was a significant nonrelapse-related fatal adverse event rate in the BR cohort (regardless of relation to BR), with most events occurring in patients >65 years of age. Despite differences in study populations, these findings are consistent with the GALLIUM trial, which showed a fatal adverse event rate >4% in patients with follicular lymphoma treated with BR and MR.20
Moving forward, BR will likely become a common backbone for the incorporation of other cytotoxic (ie, cytarabine) or novel (ie, Bruton tyrosine kinase inhibitors) agents in an attempt to further improve outcomes while maintaining an effective, safe, and ideally outpatient regimen for MCL. For example, the SHINE (NCT01776840) and ACE-LY-308 (NCT02972840) studies will inform the addition of ibrutinib and acalabrutinib, respectively, to BR in transplant-ineligible patients. ECOG-ACRIN 4181 (NCT04115631) is a randomized phase 2 study that will inform the addition of high-dose cytarabine and/or acalabrutinib to BR in transplant-eligible patients ≤70 years old with MCL. Although our data suggest that BR is an effective and well-tolerated induction regimen for patients with MCL, outcomes are suboptimal for those with high-risk features, and further studies integrating novel agents are warranted.
Presented in abstract form at the 61st annual meeting of the American Society of Hematology, Orlando, FL, 7-10 December 2019 (Abstract 2814).
For original data, please contact dvilla@bccancer.bc.ca.
Acknowledgment
Medical writers were not involved in the writing of this manuscript.
Authorship
Contribution: D.V., L.H.S., and A.S.G. designed the research; D.V. and A.S.G. performed the research and analyzed data; and all authors contributed to the acquisition, analysis, or interpretation of data for this article and drafts of the article, revised the manuscript critically for important intellectual content, approved the final version to be published, and agreed to be accountable for all aspects of the article.
Conflict-of-interest disclosure: D.V., L.H.S., K.J.S., C.L.F., D.W.S., and A.S.G. have received research funding from Roche to support the BC Cancer Centre for Lymphoid Cancer database. D.V. has received honoraria from and/or participated in advisory boards for Roche, AbbVie, Celgene, Seattle Genetics, Lundbeck, AstraZeneca, Nanostring, Janssen, Kite/Gilead, and Novartis; and has received research funding from AstraZeneca. L.H.S. has received honoraria from and/or participated in advisory boards for Karyopharm, Roche, Morphosys, Takeda, Verastem, Teva, Kite, Acerta, Seattle Genetics, AstraZeneca, Janssen, Merck, AbbVie, Amgen, Gilead, Celgene, Lundbeck, and TG Therapeutics. K.J.S. has received honoraria from and/or participated in advisory boards for BMS, Merck, Novartis, Verastem, AbbVie, Servier, and Seattle Genetics. C.L.T. has received honoraria from and/or participated in advisory boards for AbbVie, Janssen, Lundbeck, Roche, Teva, and AstraZeneca. K.S. has received honoraria from and/or participated in advisory boards for Celgene, Janssen, Takeda, Amgen, and Novartis. C.L.F. has received honoraria from and/or participated in advisory boards for Seattle Genetics, Janssen, Amgen, Celgene, and AbbVie. D.W.S. has received honoraria from and/or participated in advisory boards for Janssen; has received research funding from Roche; and has been named inventor on a patent licensed to NanoSting (institution). A.S.G. has received honoraria from and/or participated in advisory boards for Janssen, AbbVie, Lundbeck, Seattle Genetics, Celgene, and Novartis. W.D.d.B. declares no competing financial interests.
Correspondence: Diego Villa, BC Cancer–Vancouver Cancer Centre, 600 West 10th Ave, Vancouver, BC V5Z 4E6, Canada; e-mail: dvilla@bccancer.bc.ca.
References
Author notes
The full-text version of this article contains a data supplement.