Key Points
Oligo- or monoclonal expansion of HTLV-1–infected T cells in asymptomatic carriers predicts the onset of ATL.
Progression to acute type from indolent ATL was observed only in cases with monoclonal expansion.
Abstract
Adult T-cell leukemia (ATL) is an aggressive T-cell malignancy caused by human T-cell leukemia virus type 1 (HTLV-1) that develops along a carcinogenic process involving 5 or more genetic events in infected cells. The lifetime incidence of ATL among HTLV-1–infected individuals is approximately 5%. Although epidemiologic studies have revealed risk factors for ATL, the molecular mechanisms that determine the fates of carriers remain unclear. A better understanding of clonal composition and related longitudinal dynamics would clarify the process of ATL leukemogenesis and provide insights into the mechanisms underlying the proliferation of a malignant clone. Genomic DNA samples and clinical information were obtained from individuals enrolled in the Joint Study for Predisposing Factors for ATL Development, a Japanese prospective cohort study. Forty-seven longitudinal samples from 20 individuals (9 asymptomatic carriers and 11 patients with ATL at enrollment) were subjected to a clonality analysis. A method based on next-generation sequencing was used to characterize clones on the basis of integration sites. Relationships were analyzed among clonal patterns, clone sizes, and clinical status, including ATL onset and progression. Among carriers, those exhibiting an oligoclonal or monoclonal pattern with largely expanded clones subsequently progressed to ATL. All indolent patients who progressed to acute-type ATL exhibited monoclonal expansion. In both situations, the major expanded clone after progression was derived from the largest pre-existing clone. This study has provided the first detailed information regarding the dynamics of HTLV-1–infected T-cell clones and collectively suggests that the clonality of HTLV-1–infected cells could be a useful predictive marker of ATL onset and progression.
Introduction
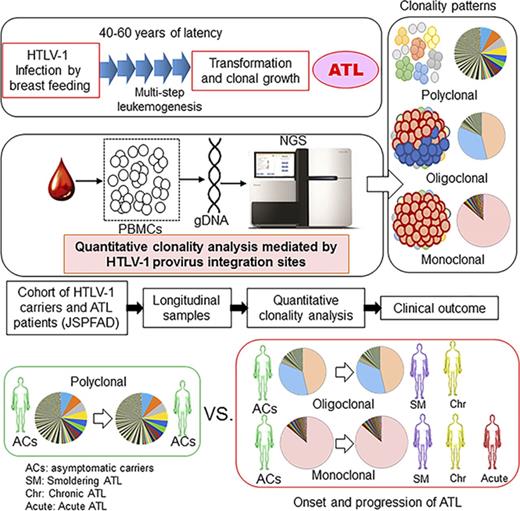
Adult T-cell leukemia/lymphoma (ATL) develops via the malignant transformation and clonal growth of human T-cell leukemia virus type 1 (HTLV-1)–infected T cells, a process that occurs after a clinically latent period of approximately 60 years after mother-to-child transmission via breastfeeding.1-5 Viral gene products play critical roles in HTLV-1–infected T-cell immortalization and malignant transformation.6-10 However, only 3% to 5% of HTLV-1–infected individuals (carriers) will develop ATL in their lifetimes.2 Although epidemiologic studies have revealed risk factors for ATL development, it remains difficult to predict which patients will develop ATL by using specific biomarkers.2,11
ATL cells are monoclonally expanded HTLV-1–infected cells, as demonstrated by Southern blot analyzes of HTLV-1 provirus integration.1,12,13 In a majority of patients, a single copy of the provirus is integrated into the chromosomal DNA of ATL cells; however, approximately 18% of patients harbor 2 or 3 integrated copies.14 The provirus integration site is considered an ideal molecular marker that discriminates individual HTLV-1–infected clonal cells. However, Southern blotting is not sufficiently sensitive to detect minor clones. To overcome this limitation, polymerase chain reaction (PCR)–based methods for clonality analysis, such as linker-mediated PCR and inverse long PCR, have been introduced.15-20 Previous studies based on those methods clearly demonstrated the presence of a polyclonally expanded background population of HTLV-1–infected cells in addition to monoclonally expanded HTLV-1–infected cells in ATL patients. Furthermore, these techniques could detect proliferation of either monoclonal or oligoclonal HTLV-1–infected cells, even in asymptomatic HTLV-1 carriers. More recently, next-generation sequencing (NGS) technology has been used to characterize the clonality of HTLV-1–infected cells in blood samples from carriers and ATL patients,21-25 and analyses using these new methods have clearly demonstrated the polyclonal compositions of infected cells in vivo.
The proviral load (PVL) is defined by the number of provirus DNA copies in peripheral blood mononuclear cells (PBMCs) and is usually expressed as a percentage representing the measured copy number per 100 PBMCs.26-28 An analysis of collected PBMC DNA samples from our cohort study, the Joint Study on Predisposing Factors of ATL Development (JSPFAD), identified a higher PVL as a major risk factor for ATL development. During the study follow-up, all 14 participants who progressed to overt ATL were among the 25% of carriers with PVLs exceeding 4%; in contrast, no ATL onset was observed among carriers with PVLs lower than 4%.26
Very low levels of viral replication and infection in vivo29,30 have led to speculation that high PVLs are maintained by persistent clonal proliferation and infected cell survival in vivo. This idea was supported by the detection of a specific HTLV-1 clone in the same carrier over the course of several years.20 These data also suggest that in patients, large numbers of HTLV-1–infected clones with varying sizes are present in the backgrounds of monoclonally expanded ATL cells. Therefore, HTLV-1–infected T cells exhibit polyclonal expansion according to provirus integration sites in adult carriers. Among a polyclonal population, monoclonal or oligoclonal expansion resulting from a growth advantage conferred by multiple gene mutations and epigenetic changes would eventually lead to clinical ATL onset.11
ATL can be classified into 4 subtypes: smoldering (SM), chronic, acute, and lymphoma.31 Among these, the acute and lymphoma types are clinically classified as aggressive-type ATL, a category that also includes some patients with chronic-type disease who have poor prognostic factors. In contrast, the SM and chronic types, which are not associated with poor prognosis and tend to correlate with longer survival, are classified as indolent ATL.32 Although there are exceptional cases, carriers are thought to progress initially to indolent-type ATL and later, in some cases, to aggressive-type ATL.
A collective study of the longitudinal dynamics of HTLV-1–infected T-cell clonality in vivo in both carriers and ATL patients would be indispensable to the delineation of the relationship between clonality and clinical status. In this study, we accessed accumulated biomaterials and clinical information from the JSPFAD and conducted a clonality analysis of longitudinal samples from 20 carriers and ATL patients. Our analysis was based on the Tag-NGS system,22 our new technique for the quantitative characterization of integration sites that measures the number of cells belonging to a specific clone.
Our study provides the first characterization of dynamic changes in clonality among HTLV-1–infected individuals, including carriers and ATL patients, with demonstrations of progression to ATL or an advanced clinical subtype. This study has provided novel information and raises questions regarding the relationship between clonality and disease progression that remain to be addressed.
Materials and methods
Clinical samples
Forty-seven clinical samples from 20 HTLV-1–infected individuals (carriers and ATL patients) were obtained from JSPFAD. The research plan was approved by the research ethics committee of the University of Tokyo (approval No. 10-50 and No. 14-155) and conducted in accordance with the Declaration of Helsinki. Patients’ ATL subtypes were diagnosed according to the Shimoyama criteria.31 Genomic DNA was isolated from PBMCs by using the QIAamp DNA Blood Kit (QIAGEN, Venlo, The Netherlands). PVLs were measured by real-time PCR on an ABI PRISM 7000 Sequence Detection System (Applied Biosystems, Inc., Foster City, CA), as described previously.26
Clonality analysis based on provirus integration sites
A specifically designed library preparation protocol was used to isolate HTLV-1 integration sites; details about the design and protocols were reported previously.22 Briefly, starting template DNA was sonicated to yield fragments of 300 to 700 base pairs (bp), which were assessed by using an Agilent 2100 Bioanalyzer and DNA 7500 kit (Agilent Technologies, Santa Clara, CA). A library of fragmented DNA samples was constructed via the following steps: end repair, A-tailing, adaptor ligation, size selection, and nested PCR. The generated products were sequenced on a HiSequation 2000/2500 platform (Illumina, Inc., San Diego, CA). Raw sequencing data were used to obtain information about HTLV-1 integration sites and clone sizes according to a previously described workflow.22 After removing a 23-bp sequence corresponding to the long terminal repeat primer, a search was performed by using the Basic Local Alignment Search Tool (BLAST)33 against the long terminal repeat reference sequence for the next 27 bp. The remaining 40 bp from BLAST hits were subjected to a BLAST search against an HTLV-1 reference sequence.34 Subsequently, reads confirmed to be from the HTLV-1 genome were removed, and sequences and identifications (IDs) from the remaining reads were considered human. We then collected Read-3 reads with IDs corresponding to those from Read-1 and mapped paired, matched-length Read-1 and Read-3 sequences against hg19 by using the Bowtie method.35 Two million mapped reads per sample were used in the subsequent analysis. The 5′ positions of mapped reads were identified as integration sites. Isolated integration site outputs used the format of chromosome: position: (strand) (eg, chr1: 121251270: (-)). Finally, clone sizes were measured on the basis of the frequency of unique Read-2 tags per integration site.
Categorizing absolute clone sizes
Clone sizes were determined by quantifying absolute numbers of infected cells using our high-throughput Tag-NGS system.22 Subsequently, clones were categorized by using previously published criteria.36 Briefly, the observed clones were categorized into 4 distinct size groups based on the distributions of clone sizes among clonality data from asymptomatic carriers (ACs) and ATL samples: very small (VS; 1-128 infected cells), small (S; 129-512 infected cells), big (B; 513-2048 infected cells), and very big (VB; >2048 infected cells).
Results
We selected 9 carriers and 11 ATL patients from the JSPFAD cohort. The follow-up periods of these 20 cases ranged from 15 months to 7 years 6 months. Clinical and laboratory data for the enrolled carriers and patients are provided in Table 1. Subtype diagnosis and classification were performed as described previously.31 Progression was defined as a change in the clinical status of an enrolled individual (eg, from carrier to any type of ATL or among subtypes of ATL). We quantified the population of each clone by using our high-throughput method and samples that had been longitudinally collected at more than 2 time points. In addition, we retrospectively characterized clonality dynamics on the basis of clone size and clinical status. The sizes and categories of the top 5 largest clones across the analyzed samples are provided in supplemental Table 1.
Sampleinformation
| ID . | Status* . | Time point (month/day/year) . | Proviral load (%) . | WBC, ×109/L . | Lymphocyte count (%) . | Abnormal lymphocyte (flower cell) count (%) . | Soluble interleukin-2 receptor, U/mL . |
|---|---|---|---|---|---|---|---|
| P1-1 | Asymptomatic carrier | 1/8/2008 | 1.1 | 6.00 | 37 | 3 | 307 |
| P1-2 | Asymptomatic carrier | 4/17/2013 | 1.24 | 5.80 | 12 | 5 | 4 790 |
| P2-1 | Asymptomatic carrier | 6/25/2010 | 5.24 | 3.40 | 48 | 0.5 | 232 |
| P2-2 | Asymptomatic carrier | 3/26/2013 | 1.23 | 3.69 | 40 | 2.3 | 435 |
| P3-1 | Asymptomatic carrier | 12/14/2007 | 1.15 | 5.18 | 32.5 | 2 | 435 |
| P3-2 | Asymptomatic carrier | 1/28/2010 | 3.68 | 5.56 | 37 | 0 | 373 |
| P4-1 | Asymptomatic carrier | 10/21/2009 | 3.52 | 9.40 | 29 | 1 | 504 |
| P4-2 | Asymptomatic carrier | 6/24/2013 | 8.73 | 7.40 | 22 | 0 | 291 |
| P5-1 | Asymptomatic carrier | 12/1/2011 | 7.16 | 6.48 | 34.5 | 4.5 | 453 |
| P5-2 | Asymptomatic carrier | 12/10/2013 | 12.76 | 5.37 | 36 | 2.3 | 292 |
| P6-1 | Asymptomatic carrier | 9/14/2007 | 11.82 | 6.32 | 35.5 | 2.5 | 325 |
| P6-2 | Smoldering | 10/11/2011 | 11.51 | 5.88 | 36 | 7 | 355 |
| P6-3 | Smoldering | 4/1/2014 | 15.41 | 5.66 | 33.5 | 7 | 300 |
| P6-4 | Smoldering | 3/30/2015 | 23.45 | 6.06 | 37.5 | 6.5 | 268 |
| P7-1 | Asymptomatic carrier | 9/7/2005 | 18.22 | 8.48 | 41 | 0 | 595 |
| P7-2 | Asymptomatic carrier | 9/6/2007 | 11.66 | 9.48 | 37.5 | 3.5 | 544 |
| P7-3 | Chronic | 5/2/2011 | 56.79 | 10.14 | 14.5 | 52 | 6 800 |
| P7-4 | Acute | 11/21/2011 | 98.31 | 27.23 | 8.4 | 65.4 | 10 700 |
| P8-1 | Asymptomatic carrier | 4/13/2011 | 13.41 | 4.30 | 39 | 3 | 626 |
| P8-2 | Smoldering | 6/20/2012 | 17.54 | 5.00 | 33 | 13 | 467 |
| P8-3 | Smoldering | 5/28/2014 | 45.27 | NA | NA | NA | NA |
| P8-4 | Chronic | 8/19/2015 | 41.02 | NA | NA | NA | NA |
| P9-1 | Asymptomatic carrier | 2/27/2012 | 19.82 | NA | NA | NA | NA |
| P9-2 | Smoldering | 5/1/2014 | 21.78 | NA | NA | NA | NA |
| P9-3 | Smoldering | 5/1/2015 | 43.85 | NA | NA | NA | NA |
| P10-1 | Smoldering | 9/6/2011 | 20.46 | 5.10 | 37 | 6 | 523 |
| P10-2 | Smoldering | 5/12/2015 | 40.67 | 6.20 | 34 | 6 | 643 |
| P11-1 | Smoldering | 10/4/2012 | 36.63 | 6.04 | 28.5 | 24.5 | 1 700 |
| P11-2 | Chronic | 7/18/2013 | 37.32 | 6.92 | 13.5 | 51 | 7 000 |
| P12-1 | Smoldering | 7/27/2009 | 23.56 | 5.00 | 23 | 18 | 992 |
| P12-2 | Chronic | 6/1/2013 | 65.78 | NA | NA | NA | NA |
| P13-1 | Smoldering | 9/29/2010 | 31.1 | 10.20 | 21 | 15 | 1 320 |
| P13-2 | Chronic | 7/26/2012 | 32.15 | 9.40 | 51 | 14 | 1 780 |
| P14-1 | Smoldering | 11/10/2009 | 55.12 | 14.00 | 7 | 52 | 1 940 |
| P14-2 | Chronic | 5/23/2013 | 54.39 | 11.80 | 16 | 35 | 2 260 |
| P15-1 | Smoldering | 5/11/2007 | 16.9 | 9.80 | 44 | 0 | 833 |
| P15-2 | Chronic | 11/10/2010 | 28.53 | 12.70 | 52 | 14 | 823 |
| P16-1 | Smoldering | 10/17/2002 | 44.80 | 7.40 | 15 | 32 | 1 250 |
| P16-2 | Acute | 5/20/2004 | 105.98 | 12.80 | 4 | 76 | 12 800 |
| P17-1 | Chronic | 12/15/2010 | 59.3 | NA | NA | NA | 1 700 |
| P17-2 | Chronic | 11/26/2012 | 56.6 | NA | NA | NA | 7 000 |
| P18-1 | Chronic | 12/15/2008 | 41.47 | 9.40 | 38 | 22 | 3 180 |
| P18-2 | Chronic | 1/6/2011 | 34.23 | 9.10 | 46 | 10 | 2 610 |
| P19-1 | Chronic | 3/13/2008 | 63.07 | 18.10 | 65 | 9 | 3 590 |
| P19-2 | Chronic | 6/16/2010 | 73.27 | 13.10 | 54 | 12 | 5 390 |
| P20-1 | Chronic | 9/16/2009 | 83.81 | 50.32 | 39.5 | 40.5 | 14 200 |
| P20-2 | Acute | 9/21/2011 | 51.42 | 52.71 | 0.4 | 97.8 | 300 |
| ID . | Status* . | Time point (month/day/year) . | Proviral load (%) . | WBC, ×109/L . | Lymphocyte count (%) . | Abnormal lymphocyte (flower cell) count (%) . | Soluble interleukin-2 receptor, U/mL . |
|---|---|---|---|---|---|---|---|
| P1-1 | Asymptomatic carrier | 1/8/2008 | 1.1 | 6.00 | 37 | 3 | 307 |
| P1-2 | Asymptomatic carrier | 4/17/2013 | 1.24 | 5.80 | 12 | 5 | 4 790 |
| P2-1 | Asymptomatic carrier | 6/25/2010 | 5.24 | 3.40 | 48 | 0.5 | 232 |
| P2-2 | Asymptomatic carrier | 3/26/2013 | 1.23 | 3.69 | 40 | 2.3 | 435 |
| P3-1 | Asymptomatic carrier | 12/14/2007 | 1.15 | 5.18 | 32.5 | 2 | 435 |
| P3-2 | Asymptomatic carrier | 1/28/2010 | 3.68 | 5.56 | 37 | 0 | 373 |
| P4-1 | Asymptomatic carrier | 10/21/2009 | 3.52 | 9.40 | 29 | 1 | 504 |
| P4-2 | Asymptomatic carrier | 6/24/2013 | 8.73 | 7.40 | 22 | 0 | 291 |
| P5-1 | Asymptomatic carrier | 12/1/2011 | 7.16 | 6.48 | 34.5 | 4.5 | 453 |
| P5-2 | Asymptomatic carrier | 12/10/2013 | 12.76 | 5.37 | 36 | 2.3 | 292 |
| P6-1 | Asymptomatic carrier | 9/14/2007 | 11.82 | 6.32 | 35.5 | 2.5 | 325 |
| P6-2 | Smoldering | 10/11/2011 | 11.51 | 5.88 | 36 | 7 | 355 |
| P6-3 | Smoldering | 4/1/2014 | 15.41 | 5.66 | 33.5 | 7 | 300 |
| P6-4 | Smoldering | 3/30/2015 | 23.45 | 6.06 | 37.5 | 6.5 | 268 |
| P7-1 | Asymptomatic carrier | 9/7/2005 | 18.22 | 8.48 | 41 | 0 | 595 |
| P7-2 | Asymptomatic carrier | 9/6/2007 | 11.66 | 9.48 | 37.5 | 3.5 | 544 |
| P7-3 | Chronic | 5/2/2011 | 56.79 | 10.14 | 14.5 | 52 | 6 800 |
| P7-4 | Acute | 11/21/2011 | 98.31 | 27.23 | 8.4 | 65.4 | 10 700 |
| P8-1 | Asymptomatic carrier | 4/13/2011 | 13.41 | 4.30 | 39 | 3 | 626 |
| P8-2 | Smoldering | 6/20/2012 | 17.54 | 5.00 | 33 | 13 | 467 |
| P8-3 | Smoldering | 5/28/2014 | 45.27 | NA | NA | NA | NA |
| P8-4 | Chronic | 8/19/2015 | 41.02 | NA | NA | NA | NA |
| P9-1 | Asymptomatic carrier | 2/27/2012 | 19.82 | NA | NA | NA | NA |
| P9-2 | Smoldering | 5/1/2014 | 21.78 | NA | NA | NA | NA |
| P9-3 | Smoldering | 5/1/2015 | 43.85 | NA | NA | NA | NA |
| P10-1 | Smoldering | 9/6/2011 | 20.46 | 5.10 | 37 | 6 | 523 |
| P10-2 | Smoldering | 5/12/2015 | 40.67 | 6.20 | 34 | 6 | 643 |
| P11-1 | Smoldering | 10/4/2012 | 36.63 | 6.04 | 28.5 | 24.5 | 1 700 |
| P11-2 | Chronic | 7/18/2013 | 37.32 | 6.92 | 13.5 | 51 | 7 000 |
| P12-1 | Smoldering | 7/27/2009 | 23.56 | 5.00 | 23 | 18 | 992 |
| P12-2 | Chronic | 6/1/2013 | 65.78 | NA | NA | NA | NA |
| P13-1 | Smoldering | 9/29/2010 | 31.1 | 10.20 | 21 | 15 | 1 320 |
| P13-2 | Chronic | 7/26/2012 | 32.15 | 9.40 | 51 | 14 | 1 780 |
| P14-1 | Smoldering | 11/10/2009 | 55.12 | 14.00 | 7 | 52 | 1 940 |
| P14-2 | Chronic | 5/23/2013 | 54.39 | 11.80 | 16 | 35 | 2 260 |
| P15-1 | Smoldering | 5/11/2007 | 16.9 | 9.80 | 44 | 0 | 833 |
| P15-2 | Chronic | 11/10/2010 | 28.53 | 12.70 | 52 | 14 | 823 |
| P16-1 | Smoldering | 10/17/2002 | 44.80 | 7.40 | 15 | 32 | 1 250 |
| P16-2 | Acute | 5/20/2004 | 105.98 | 12.80 | 4 | 76 | 12 800 |
| P17-1 | Chronic | 12/15/2010 | 59.3 | NA | NA | NA | 1 700 |
| P17-2 | Chronic | 11/26/2012 | 56.6 | NA | NA | NA | 7 000 |
| P18-1 | Chronic | 12/15/2008 | 41.47 | 9.40 | 38 | 22 | 3 180 |
| P18-2 | Chronic | 1/6/2011 | 34.23 | 9.10 | 46 | 10 | 2 610 |
| P19-1 | Chronic | 3/13/2008 | 63.07 | 18.10 | 65 | 9 | 3 590 |
| P19-2 | Chronic | 6/16/2010 | 73.27 | 13.10 | 54 | 12 | 5 390 |
| P20-1 | Chronic | 9/16/2009 | 83.81 | 50.32 | 39.5 | 40.5 | 14 200 |
| P20-2 | Acute | 9/21/2011 | 51.42 | 52.71 | 0.4 | 97.8 | 300 |
ID, identifier; NA, not available; WBC, white blood cell count.
Clinical diagnosis of each sample, based on Shimoyama criteria.
ACs
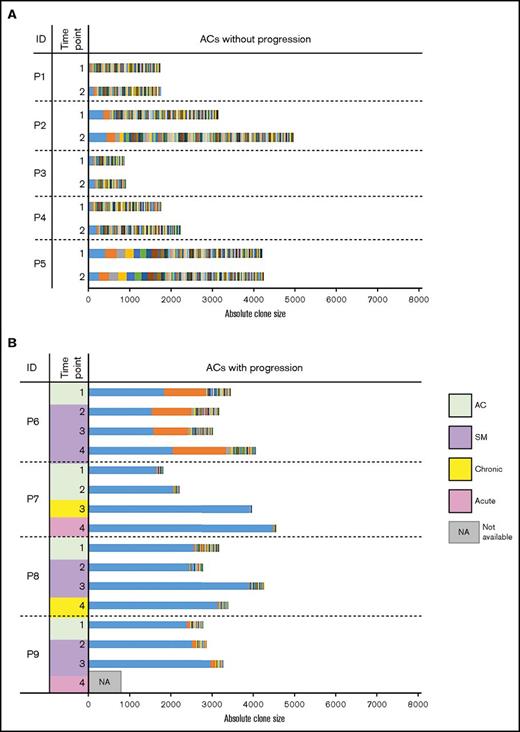
Longitudinally collected samples from nine ACs (P1-P9) were subjected to clonality analysis. Five (P1-P5) remained ACs during the observation period (Figure 1A), and 4 (P6-P9) progressed to ATL. All ACs without progression harbored both S and VS clones (ie, polyclonal pattern). The largest and second-largest clone sizes, clonality patterns, and clinical statuses are summarized in Table 2. Carriers without progression (P1-P5) exhibited small-size clones and polyclonal patterns. In contrast, carriers who progressed to ATL (P6-P9) exhibited oligoclonal or monoclonal patterns (Figure 1B; Table 2). Carrier P6, who was monitored for 7 years 6 months, developed SM-type ATL at 49 months after enrollment and maintained this status until the last observation. Carrier P7, who was monitored for 6 years 2 months, developed chronic-type ATL at time point 3 (68 months) and progressed to acute-type ATL at time point 4. Carrier P8 progressed to SM-type at time point 2 (14 months), maintained this status until time point 3 (37 months), and progressed to chronic-type ATL at time point 4 (52 months). Carrier P9, who was monitored for 3 years 2 months, developed SM-type ATL at time point 2 (26 months) and maintained this status until time point 3 (38 months). Although this patient eventually progressed to acute-type ATL, a post-progression blood sample was not available (Figure 1; Table 1).
Clonality of asymptomatic HTLV-1 carriers over time. Each colored bar segment represents a unique clone, and the segment width represents the clone size. (A) Clonality among ACs who remained ACs over time. These samples exhibited polyclonal patterns (uniform distribution) over all analyzed time points. (B) Clonality among ACs who progressed to different ATL subtypes over time. These samples largely exhibited expanded oligoclonal or monoclonal patterns.
Clonality of asymptomatic HTLV-1 carriers over time. Each colored bar segment represents a unique clone, and the segment width represents the clone size. (A) Clonality among ACs who remained ACs over time. These samples exhibited polyclonal patterns (uniform distribution) over all analyzed time points. (B) Clonality among ACs who progressed to different ATL subtypes over time. These samples largely exhibited expanded oligoclonal or monoclonal patterns.
Clone sizes, clonality patterns, and clinical progression among carriers
| ACs . | Time point 1 . | Clonality pattern . | Time point 2 . | Clonality pattern . | Clinical progression . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Top . | Second . | Top + second . | Top . | Second . | Top + second . | ||||
| P1 | 46 | 31 | 77 | Polyclonal | 112 | 54 | 166 | Polyclonal | − |
| P2 | 357 | 147 | 504 | Polyclonal | 433 | 208 | 641 | Polyclonal | − |
| P3 | 105 | 34 | 139 | Polyclonal | 153 | 25 | 178 | Polyclonal | − |
| P4 | 77 | 29 | 106 | Polyclonal | 187 | 37 | 224 | Polyclonal | − |
| P5 | 388 | 287 | 675 | Polyclonal | 244 | 243 | 487 | Polyclonal | − |
| P6 | 1852 | 1007 | 2859 | Oligoclonal | 1553 | 962 | 2515 | Oligoclonal | − |
| P7 | 1655 | 26 | 1681 | Monoclonal | 2083 | 11 | 2094 | Monoclonal | +* |
| P8 | 2572 | 39 | 2611 | Monoclonal | 2460 | 10 | 2470 | Monoclonal | − |
| P9 | 2392 | 81 | 2473 | Monoclonal | 2527 | 94 | 2621 | Monoclonal | +* |
| ACs . | Time point 1 . | Clonality pattern . | Time point 2 . | Clonality pattern . | Clinical progression . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Top . | Second . | Top + second . | Top . | Second . | Top + second . | ||||
| P1 | 46 | 31 | 77 | Polyclonal | 112 | 54 | 166 | Polyclonal | − |
| P2 | 357 | 147 | 504 | Polyclonal | 433 | 208 | 641 | Polyclonal | − |
| P3 | 105 | 34 | 139 | Polyclonal | 153 | 25 | 178 | Polyclonal | − |
| P4 | 77 | 29 | 106 | Polyclonal | 187 | 37 | 224 | Polyclonal | − |
| P5 | 388 | 287 | 675 | Polyclonal | 244 | 243 | 487 | Polyclonal | − |
| P6 | 1852 | 1007 | 2859 | Oligoclonal | 1553 | 962 | 2515 | Oligoclonal | − |
| P7 | 1655 | 26 | 1681 | Monoclonal | 2083 | 11 | 2094 | Monoclonal | +* |
| P8 | 2572 | 39 | 2611 | Monoclonal | 2460 | 10 | 2470 | Monoclonal | − |
| P9 | 2392 | 81 | 2473 | Monoclonal | 2527 | 94 | 2621 | Monoclonal | +* |
−, Maintained clinical status; +, progressed to advanced subtype; second, second-largest clone; sum, sum of top and second clone sizes; top, largest clone.
Progressed to aggressive subtype.
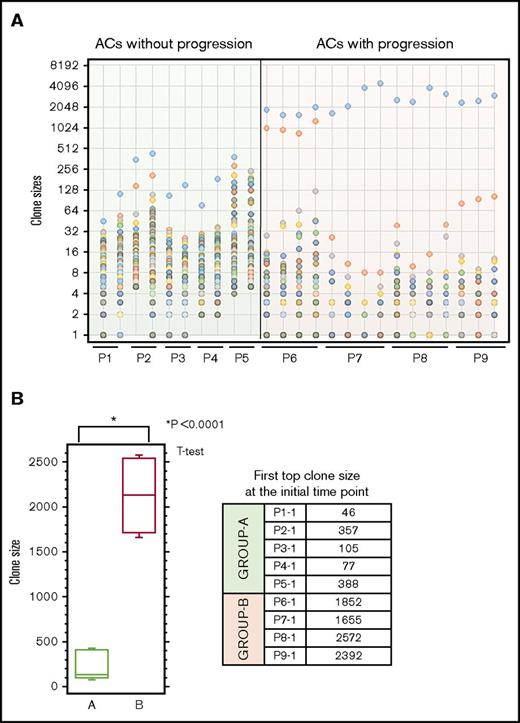
Next, we analyzed the clone sizes of carriers who eventually progressed to ATL. As shown in Table 2, all samples from carriers who exhibited clinical progression harbored 1 or 2 large clones categorized as B or VB, which represented monoclonal or oligoclonal growth, at both time points. The clone size distribution among these samples is shown in Figure 2A. The sizes of the top clones were significantly larger in ACs with progression compared with those in ACs without progression (P = .0001, Student t test) (Figure 2B). Clone sizes, clonality patterns, and clinical progression among carriers (P1-P9) are also summarized in Table 2. Briefly, these findings suggest that ACs harboring oligoclonal or monoclonal patterns are at risk of progression to ATL, whereas those with a polyclonal pattern remained as carriers and thus were considered to be the low risk group for development of ATL.
Clone size distributions among ACs. Clones are displayed in descending order based on size. Three main patterns were observed across the samples: polyclonal, oligoclonal, and monoclonal. (A) Left-most section of the graph: ACs whose clinical status did not progress. None of the samples with polyclonal patterns showed progression over time. Right-most section of the graph: ACs whose clinical status progressed. All ACs with atypical oligoclonal or monoclonal patterns showed progression. (B) Comparison of clone sizes between ACs with and without progression. Group A, ACs without progression; Group B, ACs with progression. Clone sizes of the first top clones among ACs who remained ACs vs those who developed ATL. The intergroup difference regarding the size of the first top clone was significant (Student t test, P = .0001).
Clone size distributions among ACs. Clones are displayed in descending order based on size. Three main patterns were observed across the samples: polyclonal, oligoclonal, and monoclonal. (A) Left-most section of the graph: ACs whose clinical status did not progress. None of the samples with polyclonal patterns showed progression over time. Right-most section of the graph: ACs whose clinical status progressed. All ACs with atypical oligoclonal or monoclonal patterns showed progression. (B) Comparison of clone sizes between ACs with and without progression. Group A, ACs without progression; Group B, ACs with progression. Clone sizes of the first top clones among ACs who remained ACs vs those who developed ATL. The intergroup difference regarding the size of the first top clone was significant (Student t test, P = .0001).
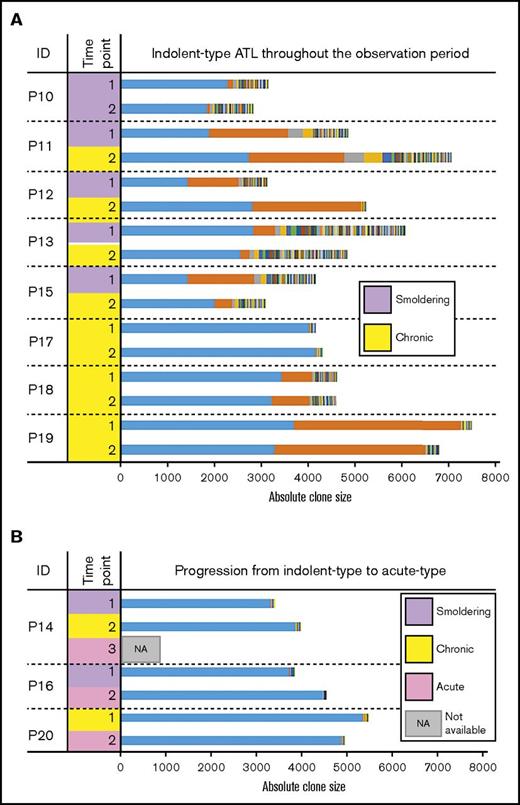
Indolent ATL cases
Seven patients (P10-P16) were diagnosed with SM-type ATL upon study enrollment (time point 1). Among them, one (P10) maintained SM-type disease throughout an observation period of 3 years 8 months. The remaining 5 cases (P11-P15) progressed to chronic-type ATL at time point 2. Although P14 progressed to acute-type ATL 4 months after time point 2, postprogression blood samples were not available for study. P16 directly progressed to acute-type ATL 19 months after time point 1 without progression to indolent-type ATL (Table 1; Figure 3).
Clonality among patients with indolent-type ATL. Each colored bar segment represents a unique clone, and the segment width indicates the clone size. (A) Longitudinal analysis of clonality among patients with indolent types of ATL (SM and chronic ATL) who exhibited oligoclonal or monoclonal patterns. (B) Clonality among individuals with SM and chronic ATL who progressed to acute ATL over time. These samples largely exhibited monoclonal patterns.
Clonality among patients with indolent-type ATL. Each colored bar segment represents a unique clone, and the segment width indicates the clone size. (A) Longitudinal analysis of clonality among patients with indolent types of ATL (SM and chronic ATL) who exhibited oligoclonal or monoclonal patterns. (B) Clonality among individuals with SM and chronic ATL who progressed to acute ATL over time. These samples largely exhibited monoclonal patterns.
The results of longitudinal clone size measurements among patients initially diagnosed with SM- or chronic-type ATL are shown in Figure 3. The clone sizes, clonality patterns, and clinical progression of these patients (P10-P20) are also summarized in Table 3 and Figure 4. Among them, 1 patient with SM-type (P10) and 3 with chronic-type ATL (P17-P19) maintained their diagnostic status throughout the observation period. The other 7 patients experienced progression: 5 patients with SM-type (P11-P15) progressed to chronic-type ATL, and 1 of them (P14) later progressed to acute-type ATL. One chronic-type ATL patient (P20) progressed to acute-type ATL at time point 2 (Figure 4B).
Clone sizes, clonality patterns, and clinical progression among patients with adult T-cell leukemia
| Patients . | Time point 1 . | Clonality pattern . | Time point 2 . | Clonality pattern . | Clinical progression . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Top . | Second . | Sum . | Top . | Second . | Sum . | ||||
| P10 | 2287 | 120 | 2407 | Monoclonal | 1857 | 71 | 1928 | Monoclonal | − |
| P11 | 1904 | 1690 | 3594 | Oligoclonal | 2742 | 2055 | 4797 | Oligoclonal | − |
| P12 | 1446 | 1088 | 2534 | Oligoclonal | 2824 | 2318 | 5142 | Oligoclonal | − |
| P13 | 2851 | 456 | 3307 | Monoclonal | 2567 | 193 | 2760 | Monoclonal | − |
| P14 | 3343 | 6 | 3349 | Monoclonal | 3882 | 6 | 3888 | Monoclonal | +* |
| P15 | 1455 | 1407 | 2862 | Oligoclonal | 2029 | 361 | 2390 | Monoclonal | + |
| P16 | 3727 | 12 | 3739 | Monoclonal | 3735 | 2 | 3737 | Monoclonal | +* |
| P17 | 4033 | 22 | 4055 | Monoclonal | 4187 | 16 | 4203 | Monoclonal | − |
| P18 | 3457 | 649 | 4106 | Oligoclonal | 3246 | 797 | 4043 | Oligoclonal | − |
| P19 | 3700 | 3560 | 7260 | Oligoclonal | 3293 | 3226 | 6519 | Oligoclonal | − |
| P20 | 5377 | 14 | 5391 | Monoclonal | 4909 | 17 | 4926 | Monoclonal | +* |
| Patients . | Time point 1 . | Clonality pattern . | Time point 2 . | Clonality pattern . | Clinical progression . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Top . | Second . | Sum . | Top . | Second . | Sum . | ||||
| P10 | 2287 | 120 | 2407 | Monoclonal | 1857 | 71 | 1928 | Monoclonal | − |
| P11 | 1904 | 1690 | 3594 | Oligoclonal | 2742 | 2055 | 4797 | Oligoclonal | − |
| P12 | 1446 | 1088 | 2534 | Oligoclonal | 2824 | 2318 | 5142 | Oligoclonal | − |
| P13 | 2851 | 456 | 3307 | Monoclonal | 2567 | 193 | 2760 | Monoclonal | − |
| P14 | 3343 | 6 | 3349 | Monoclonal | 3882 | 6 | 3888 | Monoclonal | +* |
| P15 | 1455 | 1407 | 2862 | Oligoclonal | 2029 | 361 | 2390 | Monoclonal | + |
| P16 | 3727 | 12 | 3739 | Monoclonal | 3735 | 2 | 3737 | Monoclonal | +* |
| P17 | 4033 | 22 | 4055 | Monoclonal | 4187 | 16 | 4203 | Monoclonal | − |
| P18 | 3457 | 649 | 4106 | Oligoclonal | 3246 | 797 | 4043 | Oligoclonal | − |
| P19 | 3700 | 3560 | 7260 | Oligoclonal | 3293 | 3226 | 6519 | Oligoclonal | − |
| P20 | 5377 | 14 | 5391 | Monoclonal | 4909 | 17 | 4926 | Monoclonal | +* |
−, Maintained clinical status; +, progressed to advanced subtype; second, second-largest clone; sum, sum of top and second clone sizes; top, largest clone.
Progressed to aggressive subtype.
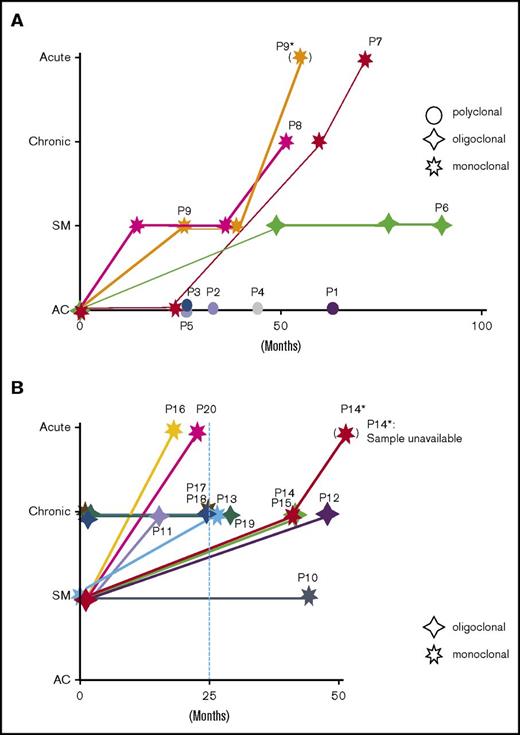
Schematic presentation of clinical courses and clonalities. (A-B) Schematic descriptions of the clinical courses of 9 asymptomatic HTLV-1 carriers and 11 patients with indolent-subtype ATL. The clinical status is shown on the vertical axis, and follow-up periods are indicated on the horizontal axis. The clonality of each patient is indicated in the figure by respective marks in the figure.
Schematic presentation of clinical courses and clonalities. (A-B) Schematic descriptions of the clinical courses of 9 asymptomatic HTLV-1 carriers and 11 patients with indolent-subtype ATL. The clinical status is shown on the vertical axis, and follow-up periods are indicated on the horizontal axis. The clonality of each patient is indicated in the figure by respective marks in the figure.
The largest clone (ie, top clone) sizes ranged from 1446 to 5377 at time point 1. Neither the largest clone size nor the sum of the 2 largest sizes correlated with progression (Figure 3; Table 3). Among the patients, 5 exhibited an oligoclonal pattern with the 2 largest clones having similar sizes (P11, P12, P15, P18, P19). However, at time point 2, P15 exhibited a monoclonal pattern and a significant decrease in the second-largest clone size (Figure 3; Table 3). Three patients (P11, P12, P15) exhibited clinical progression, and 2 patients (P18, P19) maintained chronic-type ATL (Figure 4B).
Taken together, these results show that highly expanded clones seemed to have been established in carriers several years before the onset of ATL. In addition, all patients who progressed to acute-type ATL exhibited monoclonal patterns (supplemental Figure 1).
Clones and other markers
An analysis of the measured PVLs relative to either the top clone or the sum of the top 2 clones revealed correlations with both parameters (supplemental Figure 3A-D). These findings support the PVL as an indicator of the total number of HTLV-1–infected peripheral blood cells, which mainly represent the sizes of major clones. Next, we studied the relationships between clone sizes (top clone, sum of top 2 clones) and soluble interleukin-2 receptor-α (sIL-2Ra) levels (supplemental Figure 3E-H) and observed correlations between the sizes of the major clones and the PVLs, except for 1 case with an exceptionally high sIL-2Ra level (supplemental Figure 3I-J). The results suggest that sIL-2Ra levels generally represent the total number of HTLV-1–infected T cells, but not phenotypic changes in the clonally expanded population, despite the presence of an exceptional case.
Discussion
In this study, we conducted a longitudinal analysis of samples collected from the JSPFAD cohort, which began in 2002. Accordingly, some individuals in the cohort participated in follow-up for more than 10 years and provided blood samples at every annual clinic visit. The samples analyzed in this study included those of HTLV-1 carriers who subsequently developed ATL, as well as those of patients with ATL who progressed from indolent-type to aggressive-type disease during the observation period. Therefore, the most unique aspect of this study is the quantitative characterization of clonality in longitudinal clinical samples. This study would not have been possible without a large-scale cohort study and our new next-generation sequencing based method (Tag-NGS system).
We defined and classified clonal growth patterns of HTLV-1–infected T cells as polyclonal, oligoclonal, or monoclonal, depending on the sizes of the clones that collectively composed the in vivo HTLV-1–infected T-cell populations. Clonal patterns of individuals did not seem to change significantly during the follow-up period (Figures 1-3; supplemental Table 1; supplemental Figure 2). A schematic representation of the relationship between clonality and clinical progression is described in Figure 4 and supplemental Figure 4. Results from ACs and ATL patients with SM- or chronic subtype are presented in supplemental Figure 4A and 4B, respectively.
Here we provide a collective summary of the results with respect to clonality and clinical progression. Polyclonal pattern was observed only in ACs who did not show clinical progression. Oligoclonal pattern was observed in 6 cases, one AC, 3 SM-ATL, and 2 chronic-ATL, among which 1 AC progressed to SM-ATL, 3 SM-ATL cases progressed to chronic-ATL, and 2 chronic-ATL cases did not show progression. Thus, all cases exhibiting the oligoclonal pattern maintained indolent-type ATL (SM and chronic types). The monoclonal pattern was observed in 9 individuals: 3 ACs, 4 SM-ATL cases, and 2 chronic-ATL cases. Among them, 5 cases progressed to acute-type ATL: 2 ACs, 2 SM-ATL cases, and 1 chronic-ATL case. These results suggest that the monoclonal pattern indicates a higher risk of progression to aggressive ATL. Among the 7 SM-type cases, 4 exhibited monoclonal patterns and 3 exhibited oligoclonal patterns. These results are in line with previous Southern blot analyzes in which approximately half the analyzed SM-type cases yielded monoclonal bands.12 Taken together, patients with indolent-type ATL who harbor a monoclonal infected cell population are considered at high risk of progression to acute-type ATL. This finding contrasts with results suggesting that patients exhibiting oligoclonal patterns maintained chronic-type ATL, despite progression from SM-type disease.
Although our results provide important information that will elucidate the dynamics of clonal HTLV-1–infected cell expansion in vivo, this analysis should only be considered a pilot study. Accordingly, the results should be validated in a large sample. Regarding study limitations, the sampling method used in this study was not randomized to include all clinical categories other than acute-type ATL. Furthermore, the follow-up periods of enrolled individuals varied widely (from 15 months to 7 years 6 months).
In this article, the term “clone” refers to a population of HTLV-1–infected cells that originated from a single infected cell and thus harbored the same integration site. However, a tumor cell clone is defined as a cell population with specific common genomic mutations that are usually considered to be a driving force for tumor cell proliferation, as well as the molecular basis for the tumor cell phenotype. Therefore, the correlation between clones defined by infection status and those defined by specific genomic mutations remains to be determined. Studies of this issue will provide information about the molecular mechanism underlying the growth of malignant clones within an infected cell clone.
Another unaddressed issue involves carriers with low PVLs who nevertheless develop aggressive ATL. To date, we have identified 4 cases in our cohort that corresponded to this type of ATL onset. Studies of clonality before onset will provide valuable information relevant to the molecular events underlying ATL development.
In conclusion, this longitudinal analysis of clonal dynamics in HTLV-1–infected individuals revealed that an oligoclonal or monoclonal pattern of HTLV-1–infected cells is a risk factor for ATL development in carriers. In addition, patients with indolent ATL who harbor a monoclonal pattern are also at increased risk of progression to aggressive-type ATL. Therefore, the delineation of clonality in HTLV-1 carriers and ATL patients could serve as a powerful prognostic indicator.
The full-text version of this article contains a data supplement.
Acknowledgments
The authors thank the Joint Study for Predisposing Factors for ATL Development (JSPFAD) for providing clinical samples; M. Nakashima and T. Akashi for maintaining the JSPFAD; K. Abe, K. Imamura, and T. Horiuchi for sequencing technical support; and H. Nishijima for providing technical assistance. A.F. thanks the Otsuka Toshimi Scholarship Foundation for supporting his graduate studies. Computational analyzes were provided by the Super Computer System at the Human Genome Center, Institute of Medical Science, University of Tokyo.
This work was supported by a DC1 research fellowship from the Japanese Society for the Promotion of Science (JSPS, 24.6916), a Kakenhi grant from JSPS (17K15044), and by Japan Agency for Medical Research and Development grants numbers 16ck0106133h0103 and 16ck0106136h0103 (T.W.).
Samples are registered at the National Center for Biotechnology Information under BioProject ID numbers PRJNA36018136 and PRJNA335734.
Authorship
Contribution: T.W., Y.S., S.F., and A.F. conceived the project; S.F. designed and carried out the experiments and performed the wet laboratory experiments; A.F. and S.F. performed the in silico data analysis and interpretation; Y.S. performed sequencing of samples; Y.S. and K.N. contributed to the in silico data analysis and interpretation; K.U., M.I., and A.U. contributed to the medical data interpretation and sample collection; T.W., S.F., and A.F. wrote the manuscript; A.F., S.F., and T.W. designed the figures; T.W. supervised the study; and all authors assisted in drafting the manuscript, critically revising it, and reading and approving the final manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests
Correspondence: Toshiki Watanabe, Department of Advanced Medical Innovation, St. Marianna University Graduate School of Medicine, and Graduate School of Frontier Sciences, The University of Tokyo, Tokyo 108-8639, Japan; e-mail: tnabe@ims.u-tokyo.ac.jp.
References
Author notes
S.F. and A.F. contributed equally to this study.