Tumor-specific clonal immunoglobulin expressed by B-cell lymphomas (idiotype [Id]) can serve as a target for active immunotherapy. We have previously described the vaccination of 4 patients with follicular lymphoma using dendritic cells (DCs) pulsed with tumor-derived Id protein and now report on 35 patients treated using this approach. Among 10 initial patients with measurable lymphoma, 8 mounted T-cell proliferative anti-Id responses, and 4 had clinical responses—2 complete responses (CRs) (progression-free [PF] for 44 and 57 months after vaccination), 1 partial response (PR) (PF for 12 months), and 1 molecular response (PF for 75+ months). Subsequently, 25 additional patients were vaccinated after first chemotherapy, and 15 of 23 (65%) who completed the vaccination schedule mounted T-cell or humoral anti-Id responses. Induction of high-titer immunoglobulin G anti-Id antibodies required coupling of Id to the immunogenic carrier protein keyhole limpet hemocyanin (Id-KLH). These antibodies could bind to and induce tyrosine phosphorylation in autologous tumor cells. Among 18 patients with residual tumor at the time of vaccination, 4 (22%) had tumor regression, and 16 of 23 patients (70%) remain without tumor progression at a median of 43 months after chemotherapy. Six patients with disease progression after primary DC vaccination received booster injections of Id-KLH protein, and tumor regression was observed in 3 of them (2 CRs and 1 PR). We conclude that Id-pulsed DC vaccination can induce T-cell and humoral anti-Id immune responses and durable tumor regression. Subsequent boosting with Id-KLH can lead to tumor regression despite apparent resistance to the primary DC vaccine.
Introduction
Low-grade follicular B-cell non-Hodgkin lymphoma (NHL) is an incurable disease characterized by relatively slow growth and excellent initial responsiveness to chemotherapy but also by a continuous pattern of relapse and death.1 Despite recent advances in chemotherapy, radiation therapy, and supportive care, overall survival for low-grade NHL has not significantly improved in the past 3 decades. This fact, coupled with the substantial toxicities of standard treatments, has fueled the search for novel and more tumor-selective therapies.
Fortunately, there is a target expressed by B-cell lymphomas that can be exploited using an active immunotherapeutic approach. This target is the idiotype (Id), which is composed of the unique antigenic determinants in the variable regions of the clonal immunoglobulin (Ig) expressed by the tumor cells. Each normal B cell expresses an immunoglobulin with unique variable region sequences in the heavy and light chains that together form the antigen-binding site. When a B cell undergoes malignant transformation, these sequences are maintained by the malignant clone and can thus serve as a tumor-specific antigen. Id proteins contain structures that can be recognized by antibodies2-5 and by CD4+ T cells5-8 and CD8+ T cells9-12 and can be isolated from autologous tumor cells and formulated into a custom-made vaccine.
In preclinical studies, vaccination of mice with Id protein coupled to the highly immunogenic carrier protein keyhole limpet hemocyanin (Id-KLH) has been shown to protect animals from subsequent tumor challenge5,13-16 and to cure established lymphoma when combined with chemotherapy.16,17 In our previous clinical studies, vaccination with Id-KLH plus a chemical adjuvant was found to induce anti-Id immune responses in approximately half the patients with follicular NHL. These immune responses were correlated with improved disease-free survival and overall survival.6 7Subsequently, ways have been sought to improve the frequency and the potency of anti-Id immune responses in patients with NHL.
The critical role played by dentritic cells (DCs) in the initiation of immune responses makes them an attractive addition to cancer vaccine strategies.18,19 When pulsed (cocultured) with tumor-derived peptides or proteins or when transduced with tumor antigen-encoding viruses or nucleic acids and administered as a cellular vaccine, DCs have been shown to promote protective and even therapeutic antitumor immunity in murine tumor models. These studies have provided a compelling rationale for the clinical use of DCs in active vaccination strategies against human cancer.20Based on studies in a murine lymphoma model showing that vaccination with Id-pulsed DCs could induce protective antitumor immunity,21 we initiated a pilot clinical trial in 1993 of Id-pulsed DC vaccination for patients with follicular NHL. In the preliminary report of our first 4 patients,22 we described the induction of cellular anti-Id responses and evidence of antitumor effects in 3 of 4 patients. Since our initial report, clinical activity of DC-based vaccines has also been demonstrated in melanoma,23,24 prostate carcinoma,25,26 renal cell carcinoma,27 and carcinoembryonic antigen–expressing cancers.28
We now report on our expanded experience and long-term follow-up of 35 patients with follicular NHL treated with Id-pulsed DC vaccination, including a cohort of 25 patients treated in first remission after their initial cytoreductive chemotherapy. Our observations include durable, objective tumor regression in patients with measurable disease, induction of T-cell anti-Id immune responses, and induction of anti-Id antibodies capable of specifically recognizing and mediating signal transduction in autologous tumor cells. In addition, we found that patients who were resistant to or who had relapses after primary Id-pulsed DC vaccination could achieve complete tumor regression after the administration of booster vaccinations of Id-KLH protein. These findings confirm the clinical activity of Id-pulsed DCs and Id-KLH vaccination in follicular NHL and provide the rationale for applying similar strategies to other B-cell malignancies.
Patients, materials, and methods
Patients
Patients were adults with stage III or IV follicular B-cell NHL (follicular small-cleaved cell or follicular mixed small-cleaved and large-cell by Working Formulation, follicle center cell grades I and II by REAL classification). Each patient had a peripheral lymph node that measured at least 2 × 2 cm and that was accessible to excisional biopsy for diagnostic confirmation and vaccine production. Monoclonal surface immunoglobulin expression was verified using flow cytometry. The first 10 patients enrolled in the study (pilot phase) had measurable relapsed or residual tumor after one or more previous therapies (Table 1), and the remaining 25 patients were vaccinated after an attempt at remission induction with chemotherapy (Table 2). Each patient's physician determined the timing and choice of chemotherapy, and therapy was continued until a maximal response was achieved. Clinical responses were classified according to the standardized response criteria of Cheson et al.29 Vaccinations were initiated at least 2 months after the completion of chemotherapy according to the schedule below. Prevaccine patient evaluation included physical examination, complete blood count, lymphocyte subset analysis, serum chemistry and rheumatoid factor analyses, computed tomography (CT) of the chest, abdomen, and pelvis, and bilateral iliac crest bone marrow biopsy. These studies were repeated 1 month after the completion of vaccination, and subsequent follow-up examinations performed every 3 months included physical examination, complete blood count, and serum chemistry analysis. Repeat CT was performed every 6 months for the first 2 years and annually thereafter, or as clinically indicated. Prevaccine tumor status of patients was classified as either no evidence of disease (NED), minimal residual disease (MRD), or residual disease (RD), corresponding to the attainment and maintenance of a complete response (CR), complete response unconfirmed (CRu), or partial responses (PR), respectively, as specified in the Cheson criteria. The study was approved by the Institutional Review Board of Stanford University Medical Center, and all patients supplied written informed consent.
Characteristics and responses of initial patients with measurable disease (n = 10)
| Patient . | Age/ sex . | Histology . | Prior therapies . | Sites of disease . | Total DCs given* . | Anti-Id responses . | Clinical response, time to progression . | Current status, subsequent therapies, months since DC vaccine . | |
|---|---|---|---|---|---|---|---|---|---|
| T cell . | Ab . | ||||||||
| D1† | 59/F | FM | Chl, ChlVB | Paracardiac, mediastinal | 69 × 106 | + | − | CR, 44 months | RD after CHOP, rituximab, XRT, fludarabine, repeat DC vaccine, 94 mos |
| D2† | 44/F | FM | Chl, CVP | Peri-aortic, para-iliac | 22 × 106 | + | − | PR, 12 months | NED after Id-KLH/SAF,1-153 fludarabine, 83 mos |
| D3† | 34/F | F/DSC | CVP, fludarabine | BM | 14 × 106 | + | +μ‡ | Cleared BM PCR signal | NED 79 mos after DC vaccine without subsequent therapies |
| D4† | 45/M | FSC | CVP, fludarabine | Cervical, axillary, peri-aortic, iliac, inguinal | 16 × 106 | + | − | PD at 10 mos | NED after MINE, rituximab, 69 mos |
| D5 | 50/F | FM | CVP, fludarabine | Peri-aortic, BM | 23 × 106 | + | − | PD at 7 mos | NED after Id-KLH/SAF,1-153 rituximab, 65 mos |
| D6 | 47/M | FSC | Chl, CHOP | Peri-aortic, spleen, mesenteric | 22 × 106 | + | − | PD at 12 mos | RD after Id-KLH/SAF,1-153 rituximab, XRT, 63 mos |
| D7 | 43/F | FSC | CHOP | Axillary, mesenteric | 27 × 106 | + | − | PD at 9 mos | MRD after rituximab, 58 mos |
| D8 | 40/M | FSC | CVP, XRT, CEP, fludarabine | Axillary, peri-aortic | 10 × 106 | + | +μ‡ | PD at 5 mos | NED after Id-KLH/SAF,1-153 57 mos |
| D9 | 43/F | FSC | Chl, Id-KLH/SAF | Axillary, iliac, peri-aortic | 13 × 106 | − | − | CR, 57 mos | PD at single site 57 mos after DC vaccine |
| D10 | 51/F | FM | CVP, fludarabine, CHOP | Axillary, spleen | 12 × 106 | − | − | PD at 7 mos | NED after Id-KLH/SAF,1-153 tositumomab, 47 mos |
| Patient . | Age/ sex . | Histology . | Prior therapies . | Sites of disease . | Total DCs given* . | Anti-Id responses . | Clinical response, time to progression . | Current status, subsequent therapies, months since DC vaccine . | |
|---|---|---|---|---|---|---|---|---|---|
| T cell . | Ab . | ||||||||
| D1† | 59/F | FM | Chl, ChlVB | Paracardiac, mediastinal | 69 × 106 | + | − | CR, 44 months | RD after CHOP, rituximab, XRT, fludarabine, repeat DC vaccine, 94 mos |
| D2† | 44/F | FM | Chl, CVP | Peri-aortic, para-iliac | 22 × 106 | + | − | PR, 12 months | NED after Id-KLH/SAF,1-153 fludarabine, 83 mos |
| D3† | 34/F | F/DSC | CVP, fludarabine | BM | 14 × 106 | + | +μ‡ | Cleared BM PCR signal | NED 79 mos after DC vaccine without subsequent therapies |
| D4† | 45/M | FSC | CVP, fludarabine | Cervical, axillary, peri-aortic, iliac, inguinal | 16 × 106 | + | − | PD at 10 mos | NED after MINE, rituximab, 69 mos |
| D5 | 50/F | FM | CVP, fludarabine | Peri-aortic, BM | 23 × 106 | + | − | PD at 7 mos | NED after Id-KLH/SAF,1-153 rituximab, 65 mos |
| D6 | 47/M | FSC | Chl, CHOP | Peri-aortic, spleen, mesenteric | 22 × 106 | + | − | PD at 12 mos | RD after Id-KLH/SAF,1-153 rituximab, XRT, 63 mos |
| D7 | 43/F | FSC | CHOP | Axillary, mesenteric | 27 × 106 | + | − | PD at 9 mos | MRD after rituximab, 58 mos |
| D8 | 40/M | FSC | CVP, XRT, CEP, fludarabine | Axillary, peri-aortic | 10 × 106 | + | +μ‡ | PD at 5 mos | NED after Id-KLH/SAF,1-153 57 mos |
| D9 | 43/F | FSC | Chl, Id-KLH/SAF | Axillary, iliac, peri-aortic | 13 × 106 | − | − | CR, 57 mos | PD at single site 57 mos after DC vaccine |
| D10 | 51/F | FM | CVP, fludarabine, CHOP | Axillary, spleen | 12 × 106 | − | − | PD at 7 mos | NED after Id-KLH/SAF,1-153 tositumomab, 47 mos |
FSC indicates follicular small cleaved cell; FM, follicular mixed small cleaved and large cell; F/DSC, follicular and diffuse small cleaved cell; Chl, chlorambucil; ChlVB, chlorambucil, vincristine, and bleomycin; CVP, cyclophosphamide, vincristine, and prednisone; CHOP, cyclophosphamide, adriamycin, vincristine, and prednisone; CEP, cyclophosphamide, etoposide, and prednisone; Id-KLH/SAF, Id protein coupled to KLH plus Syntex adjuvant formulation; XRT, radiation therapy; MINE, mesna, ifosfamide, mitoxantrone, etoposide; PD, progressive disease; NED, no clinical evidence of disease; MRD, minimum residual disease; RD, residual disease; Ab, antibody; BM, bone marrow.
Total number of DCs infused over the course of 4 vaccinations.
Previously reported in original pilot study.22
IgM, but not IgG anti-Id antibodies detectable in serum.
Revaccinated with Id-KLH plus SAF adjuvant. See Table 3.
Characteristics and immune responses of patients vaccinated in first remission after chemotherapy (n = 25)
| Patient . | Age/sex . | Histology, stage . | Chemotherapy, no. cycles . | Prevaccine tumor status . | DCs pulsed with . | No. DCs* . | Anti-Id response . | Follow-up after chemotherapy . | |
|---|---|---|---|---|---|---|---|---|---|
| T cell . | Ab . | ||||||||
| D11 | 65/M | FM, IVA | CHOP × 4 | RD | Id | 33 × 106 | − | − | SD, 51 mos |
| CVP/F × 4 | |||||||||
| D12 | 44/F | FSC, IVA | CVP × 8 | MRD | Id | 9 × 106 | + | − | PD at 15 mos, NED at 62 mos‡ |
| D13 | 53/F | FSC, IIIA | CVP × 8 | RD | Id | 19 × 106 | − | − | NED, 52 mos |
| D14 | 60/M | FSC, IIIA | CVP × 6 | NED | Id | 9 × 106 | − | − | NED, 53 mos |
| D15 | 30/M | FSC, IVB | CVP × 8 | MRD | Id | 19 × 106 | + | − | SD, 47 mos |
| D16 | 34/M | FSC, IIIA | CVP × 6 | MRD | Id | 11 × 106 | + | − | NED, 52 mos |
| D17 | 44/F | FSC, IVA | CVP/F × 8 | MRD | Id | 17 × 106 | − | +μ† | NED, 56 mos |
| D18 | 52/F | FSC, IVA | Chl × 6 | MRD | Id | 17 × 106 | + | − | NED, 43 mos |
| D19 | 60/F | FSC, IVA | CVP × 8 | MRD | Id | 14 × 106 | − | − | SD, 46 mos |
| D20 | 48/M | FSC, IVA | CVP × 6 | PD | Id | 18 × 106 | − | − | PD at 9 mos, alive at 50 mos |
| D21 | 38/M | FM, IVA | CVP × 6 | MRD | Id | 8 × 106 | + | − | PD at 25 mos, dead at 32 mos2-153 |
| D22 | 40/M | FSC, IVA | CVP × 8 | MRD | Id | 19 × 106 | − | − | PD at 35 mos, alive at 44 mos |
| D23 | 55/F | FSC, IVA | CVP × 8 | RD | Id-KLH | 15 × 106 | − | +μ† | SD, 39 mos |
| D24 | 47/F | FM, IVA | CVP × 6 | NED | Id-KLH | 16 × 106 | + | − | NED, 38 mos |
| D25 | 30/M | FSC, IVA | CVP × 6 | RD | Id-KLH | 10 × 106 | − | − | PD at 18 mos, alive at 37 mos |
| D26 | 56/M | FM, IVA | CVP × 7 | NED | Id-KLH | 16 × 106 | + | + | NED, 45 mos |
| D27 | 50/F | FM, IVA | CVP × 6 | PD | Id-KLH | 21 × 106 | − | − | PD at 9 mos,2-153 alive at 35 mos |
| D28 | 61/M | FM, IVA | CVP × 6 | NED | Id-KLH | 10 × 106 | + | − | PD at 21 mos, alive at 36 mos |
| D29 | 50/M | FSC, IVB | CVP × 8 | RD | Id-KLH | 12 × 106 | − | + | MRD, 28 mos |
| D30 | 50/F | FM, IVA | CVP × 6 | MRD | Id-KLH | 20 × 106 | − | − | SD, 24 mos |
| CHOP × 2 | |||||||||
| D31 | 54/F | FSC, IVA | CVP × 8 | NED | Id-KLH | 10 × 106 | − | + | NED, 24 mos |
| D32 | 43/F | FSC, IVA | CVP × 8 | MRD | Id-KLH | 15 × 106 | − | +μ† | PD at 16 mos, alive at 25 mos |
| D33 | 55/M | FM, IIIA | CVP × 7 | RD | Id-KLH | 21 × 106 | − | − | SD, 23 mos |
| CHOP × 4 | |||||||||
| D34 | 53/M | FSC, IVA | CVP × 6 | RD | Id-KLH | 35 × 106 | − | + | SD, 25 mos |
| CHOP × 2 | |||||||||
| D35 | 47/M | FSC, IVA | CVP × 8 | MRD | Id-KLH | 28 × 106 | + | − | PD at 28 mos, alive at 33 mos |
| Patient . | Age/sex . | Histology, stage . | Chemotherapy, no. cycles . | Prevaccine tumor status . | DCs pulsed with . | No. DCs* . | Anti-Id response . | Follow-up after chemotherapy . | |
|---|---|---|---|---|---|---|---|---|---|
| T cell . | Ab . | ||||||||
| D11 | 65/M | FM, IVA | CHOP × 4 | RD | Id | 33 × 106 | − | − | SD, 51 mos |
| CVP/F × 4 | |||||||||
| D12 | 44/F | FSC, IVA | CVP × 8 | MRD | Id | 9 × 106 | + | − | PD at 15 mos, NED at 62 mos‡ |
| D13 | 53/F | FSC, IIIA | CVP × 8 | RD | Id | 19 × 106 | − | − | NED, 52 mos |
| D14 | 60/M | FSC, IIIA | CVP × 6 | NED | Id | 9 × 106 | − | − | NED, 53 mos |
| D15 | 30/M | FSC, IVB | CVP × 8 | MRD | Id | 19 × 106 | + | − | SD, 47 mos |
| D16 | 34/M | FSC, IIIA | CVP × 6 | MRD | Id | 11 × 106 | + | − | NED, 52 mos |
| D17 | 44/F | FSC, IVA | CVP/F × 8 | MRD | Id | 17 × 106 | − | +μ† | NED, 56 mos |
| D18 | 52/F | FSC, IVA | Chl × 6 | MRD | Id | 17 × 106 | + | − | NED, 43 mos |
| D19 | 60/F | FSC, IVA | CVP × 8 | MRD | Id | 14 × 106 | − | − | SD, 46 mos |
| D20 | 48/M | FSC, IVA | CVP × 6 | PD | Id | 18 × 106 | − | − | PD at 9 mos, alive at 50 mos |
| D21 | 38/M | FM, IVA | CVP × 6 | MRD | Id | 8 × 106 | + | − | PD at 25 mos, dead at 32 mos2-153 |
| D22 | 40/M | FSC, IVA | CVP × 8 | MRD | Id | 19 × 106 | − | − | PD at 35 mos, alive at 44 mos |
| D23 | 55/F | FSC, IVA | CVP × 8 | RD | Id-KLH | 15 × 106 | − | +μ† | SD, 39 mos |
| D24 | 47/F | FM, IVA | CVP × 6 | NED | Id-KLH | 16 × 106 | + | − | NED, 38 mos |
| D25 | 30/M | FSC, IVA | CVP × 6 | RD | Id-KLH | 10 × 106 | − | − | PD at 18 mos, alive at 37 mos |
| D26 | 56/M | FM, IVA | CVP × 7 | NED | Id-KLH | 16 × 106 | + | + | NED, 45 mos |
| D27 | 50/F | FM, IVA | CVP × 6 | PD | Id-KLH | 21 × 106 | − | − | PD at 9 mos,2-153 alive at 35 mos |
| D28 | 61/M | FM, IVA | CVP × 6 | NED | Id-KLH | 10 × 106 | + | − | PD at 21 mos, alive at 36 mos |
| D29 | 50/M | FSC, IVB | CVP × 8 | RD | Id-KLH | 12 × 106 | − | + | MRD, 28 mos |
| D30 | 50/F | FM, IVA | CVP × 6 | MRD | Id-KLH | 20 × 106 | − | − | SD, 24 mos |
| CHOP × 2 | |||||||||
| D31 | 54/F | FSC, IVA | CVP × 8 | NED | Id-KLH | 10 × 106 | − | + | NED, 24 mos |
| D32 | 43/F | FSC, IVA | CVP × 8 | MRD | Id-KLH | 15 × 106 | − | +μ† | PD at 16 mos, alive at 25 mos |
| D33 | 55/M | FM, IIIA | CVP × 7 | RD | Id-KLH | 21 × 106 | − | − | SD, 23 mos |
| CHOP × 4 | |||||||||
| D34 | 53/M | FSC, IVA | CVP × 6 | RD | Id-KLH | 35 × 106 | − | + | SD, 25 mos |
| CHOP × 2 | |||||||||
| D35 | 47/M | FSC, IVA | CVP × 8 | MRD | Id-KLH | 28 × 106 | + | − | PD at 28 mos, alive at 33 mos |
FSC indicates follicular small cleaved cell; FM, follicular mixed small cleaved and large cell; F/DSC, follicular and diffuse small cleaved cell; CHOP, cyclophosphamide, adriamycin, vincristine, and prednisone; CVP, cyclophosphamide, vincristine, and prednisone; CVP/F, CVP alternating with fludarabine; Chl, chlorambucil; RD, residual disease; MRD, minimum residual disease; SD, stable disease; PD, progressive disease; NED, no clinical evidence of disease; Ab, antibody.
Total number of DCs infused over the course of 4 vaccinations.
IgM, but not IgG anti-Id antibodies detectable in serum.
Revaccinated with Id-KLH plus SAF adjuvant. See Table 3.
Histologic transformation to diffuse large cell lymphoma.
Vaccine production
In most cases, lymphoma Id proteins were isolated using the rescue hybridoma method.6,22 This technique yields Id proteins of either the IgG or the IgM isotype, depending on that produced by tumor cells. An alternative strategy, molecular rescue, was used to prepare Id proteins for patients D17 and patients D21 to D26. In this method, the genes encoding the heavy and light chain variable regions from tumor Ig were amplified by polymerase chain reaction (PCR) using family-specific immunoglobulin variable region primers,30 cloned into plasmid vectors containing human IgG constant regions, and cotransfected into a mammalian cell line31 that then secreted the tumor-specific Ig product. All tumor Id proteins were purified by affinity chromatography as previously described7 and, where indicated, were chemically conjugated with pyrogen-free KLH (Calbiochem, San Diego, CA) as previously described.6
We used peripheral blood DCs for our Id-pulsed DC vaccine approach given the proven capacity of these cells to prime naive CD4+ and CD8+ T cells in vitro.32,33 DCs were isolated from leukapheresis products by a series of density gradient centrifugation steps, as previously described.22 Peripheral blood mononuclear cells (PBMCs) were obtained by leukapheresis using a COBE cell separator apparatus followed by Ficoll-Hypaque sedimentation (Pharmacia, Uppsala, Sweden). Monocytes were then removed by a discontinuous (50%) Percoll gradient (Pharmacia). The resultant high-density fraction was cultured overnight in RPMI 1640 plus 10% human AB serum in Teflon vessels (Savillex, Minneapolis, MN) along with antigen at 2 μg/mL. During this period, DC precursors spontaneously matured without exogenous cytokines and acquired the low-density characteristic of DCs. The next day, high-density lymphocytes were removed using a 15% metrizamide gradient (Sigma, St Louis, MO), and the low-density, DC-enriched fraction was then recultured overnight in antigen at 50 μg/mL and was harvested, washed, and resuspended in sterile saline for re-infusion. The choice of antigen concentrations for sequential overnight incubations with DCs was based on our earlier in vitro studies using nominal antigens32,33 and on the limited availability of Id proteins. DCs were enumerated by flow cytometry as lineage (CD3, CD14, CD19) negative and HLA-DR bright, and purity was confirmed by morphology. For patients D1 to D22, DCs were divided and pulsed separately with Id and KLH as in our original report,22but patients D23 to D35 received DCs pulsed with Id-KLH conjugate as indicated in Table 2.
Vaccine treatments
Antigen-pulsed DCs were administered intravenously after premedication with acetaminophen and diphenhydramine. Patients received 3 monthly infusions, followed by a fourth given 2 to 6 months later. Each infusion was followed 2 weeks later by subcutaneous injections of 0.5 mg soluble Id and KLH proteins (without DCs). In patients D1 to D22, Id and KLH proteins were injected separately at different sites as in our original report,22 whereas in patients D23 to D35, Id-KLH conjugate was administered at a single site. Where indicated (Table 3), certain patients received further subcutaneous booster vaccinations consisting of 0.5 mg Id protein coupled to 0.5 mg KLH and emulsified in the chemical adjuvant SAF1 as previously described.6 7 Five of these vaccinations were administered at weeks 0, 4, 8, 12, and 20.
Characteristics and responses of patients revaccinated with Id-KLH protein + SAF1
| Patient . | Interval from DC to Id-KLH vaccination . | Sites of disease . | Anti-Id response . | Clinical response, time to progression . | |
|---|---|---|---|---|---|
| T cell . | Ab . | ||||
| D2 | 18 mos | Cervical, iliac, peri-aortic | + | − | PR, 14 mos |
| D5 | 16 mos | Skin, cervical, axillary, inguinal | − | − | PD |
| D6 | 16 mos | Parotid, axillary, mesenteric, peri-aortic | − | − | PD |
| D8 | 8 mos | Axillary, peri-aortic | − | +μ3-150 | CR, 48+ mos |
| D10 | 22 mos | Skin, peri-aortic, axillary, inguinal | − | − | PD |
| D123-151 | 33 mos | Cervical, axillary, iliac, inguinal, BM | + | − | CRu, 16 mos |
| 49 mos3-152 | Cervical, BM | + | − | CR, 1+ mos | |
| Patient . | Interval from DC to Id-KLH vaccination . | Sites of disease . | Anti-Id response . | Clinical response, time to progression . | |
|---|---|---|---|---|---|
| T cell . | Ab . | ||||
| D2 | 18 mos | Cervical, iliac, peri-aortic | + | − | PR, 14 mos |
| D5 | 16 mos | Skin, cervical, axillary, inguinal | − | − | PD |
| D6 | 16 mos | Parotid, axillary, mesenteric, peri-aortic | − | − | PD |
| D8 | 8 mos | Axillary, peri-aortic | − | +μ3-150 | CR, 48+ mos |
| D10 | 22 mos | Skin, peri-aortic, axillary, inguinal | − | − | PD |
| D123-151 | 33 mos | Cervical, axillary, iliac, inguinal, BM | + | − | CRu, 16 mos |
| 49 mos3-152 | Cervical, BM | + | − | CR, 1+ mos | |
CR indicates complete response; CRu, complete response unconfirmed (normalization of radiographs, equivocal findings in bone marrow); PR, partial response; PD, progressive disease; BM, bone marrow.
IgM, but not IgG anti-Id antibodies detectable in serum.
Adjuvant = ISAF (“incomplete” SAF1, without threonyl MDP component).
Repeat course of Id-KLH protein + ISAF vaccinations (vaccine course 3).
Cellular immune response assessments
T-cell proliferation assays were performed as reported previously.6,7 22 Fresh PBMCs were cultured in quadruplicate in media alone or with tumor Id, irrelevant Id proteins, or KLH at 0.1, 1.0, 10, and 100 μg/mL. Incorporation of [3H]-thymidine (Amersham Pharmacia Biotech, Piscataway, NJ; 185 GBq/mmol) was measured after an overnight pulse (1 μCi [0.037 MBq]/well) on day 5. A response was interpreted as positive when the incorporation of more than twice the background (media alone) with at least one antigen concentration was observed on 2 or more occasions.
Tumor-specific cytotoxicity assays for patient D12 were adapted from methods described by Schultze et al.34 Autologous follicular lymphoma cells were activated by 3- to 5-day coculture with irradiated (55 Gy) CD40L-expressing mouse L cells (a gift of Yong-Jun Liu, DNAX, Palo Alto, CA). Activated tumor cells were then irradiated (64 Gy) and were used to restimulate cryopreserved prevaccine and postvaccine (vaccine course 3) PBMCs for 2 weekly cycles. Interleukin-2 (Chiron, Emeryville, CA) was added at 30 IU/mL on the third day of culture, with fresh medium added every 3 days thereafter. On day 14, cytotoxicity against unmodified, cryopreserved autologous follicular lymphoma cells, irrelevant tumor cells (non-HLA matched follicular lymphoma), and normal B cells was assessed in a standard 4-hour chromium Cr 51 release assay.12 Normal autologous peripheral blood B cells were isolated by negative selection using the RosetteSep reagent (StemCell Technologies, Vancouver, BC, Canada), and flow cytometry showed that they were more than 95% CD19+and without evidence of light chain restriction (data not shown). Target cells were labeled with 51Cr (Amersham, 150 μCi (555 MBq/mL) per 106 target cells) for 1.5 to 2 hours and were washed 3 times, and 10 000 targets were incubated with effector cells at the effector–target (E/T) ratios of 100, 30, 10, and 3. Unlabeled K562 erythroleukemia cells were added at a ratio of 50:1 to labeled targets to quench natural killer cell–mediated lysis as previously described.12 Spontaneous release of51Cr was less than 20% for all target cell populations. The percentage specific lysis is expressed as the mean of triplicate values, determined using the equation: % Specific lysis = (experimental cpm − spontaneous release cpm)/(maximum release cpm − spontaneous release cpm) × 100.
Humoral immune response assessments
Enzyme-linked immunosorbent assay was used to analyze sera of all patients for anti-Id antibodies. Autologous IgM tumor Id proteins or irrelevant IgM Id proteins were captured onto microtiter plates coated with goat anti–human IgM (Biosource International, Camarillo, CA). When the tumor Id was an IgG, the plates were coated directly either with F(ab′)2 fragments of the IgG Id produced by digestion with immobilized pepsin (Pierce, Rockford, IL) or with whole IgG Id. Prevaccine and postvaccine sera were serially diluted and allowed to bind to the target Id proteins. Reagents used to detect bound antibodies depended on the isotype of the target Id molecule. When the Id protein was an IgM or a F(ab′)2 fragment of IgG, bound anti-Id antibodies of the IgG subclass were detected using polyclonal goat anti–human IgG coupled to horseradish peroxidase (HRP; Southern Biotech, Birmingham, AL). When the target Id protein was whole IgG, bound anti-Id antibodies were detected with polyclonal anti–human IgM HRP or with antibodies to the immunoglobulin light chain (κ or λ) opposite that of the tumor Id. A response was considered positive when a 4-fold increase in anti-Id titer was found compared to the prevaccine serum and the isotype-matched irrelevant Id proteins used as specificity controls. Antibody responses to KLH were measured as previously described.7 For tumor cell staining experiments, cryopreserved IgM-positive tumor cells were incubated with 1% prevaccine or postvaccine serum overnight at 4°C, washed, and incubated with goat anti–human IgG F(ab′)2-phycoerythrin (PE) followed by counter-staining with anti–human IgM F(ab′)2–fluorescein isothiocyanate (FITC; Biosource). Stained cells were analyzed using a FACScan flow cytometer (Becton Dickinson, San Jose, CA).
Analysis of signal transduction in tumor cells
Cryopreserved tumor cells (2 × 106/mL) were incubated with 1% relevant or irrelevant prevaccine or postvaccine serum for 3 hours on ice to allow the binding of anti-Id antibodies to the cell surface, warmed to 37°C for 2 minutes, and immediately chilled by the addition of ice-cold phosphate-buffered saline plus 1 mM Na3VO4 and washed once. Goat F(ab′)2 anti-μm antibodies (Biosource) served as a positive control. Western blotting was performed as previously described.35 Briefly, aliquots of cells treated as above were lysed, and solubilized proteins were separated by 8% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions and were transferred to nitrocellulose (Schleicher and Schuell, Keene, NJ). The blot was then stained with antiphosphotyrosine antibody 4G10 (Upstate Biotechnology, Lake Placid, NY) followed by goat anti–mouse IgG-HRP (Southern Biotech) and was developed using the enhanced chemiluminescence reagent (Amersham Pharmacia Biotech). For the detection of tyrosine phosphorylation by flow cytometry, cells treated as above were fixed and permeabilized (FACS Lysing Solution; Becton Dickinson), then were stained with antibody 4G10 coupled to FITC (Upstate Biotechnology). Aliquots of cells counter-stained with phycoerythrin-labeled anti-CD20 or anti-CD3 antibodies were fixed in 1% paraformaldehyde, and phosphotyrosine content was quantitated by FACScan.
Results
Pilot experience in patients with measurable follicular lymphoma
Idiotype-pulsed DC vaccination was first applied in the pilot phase of our study to 10 patients with measurable relapsed or residual tumor after chemotherapy. Characteristics of these patients are shown in Table 1. In this group, native, unmodified Id protein was used for pulsing the DCs. Patients D1 to D4 were described in our initial report.22 Eight of the 10 patients underwent 2 or more chemotherapy regimens before vaccination, and all but one had multiple sites of evaluable tumor. Total numbers of DCs administered over the 4 infusions were dictated by cell yield and ranged from 12 to 69 × 106 (median, 19 × 106; range, 2-32 × 106/infusion). Side effects (mild to moderate chills, rigors, or fevers) were associated with less than 20% of infusions and were self-limiting.
After vaccination, T-cell proliferative responses to Id protein developed in 8 of the 10 patients. Serum anti-Id antibodies of the IgG class were not found in any patient using our traditional anti-IgG detection strategy,6 7 but 2 patients (D3 and D8) were found to have serum anti-Id antibodies of the IgM class after vaccination. All 10 patients mounted humoral and cellular proliferative responses to KLH.
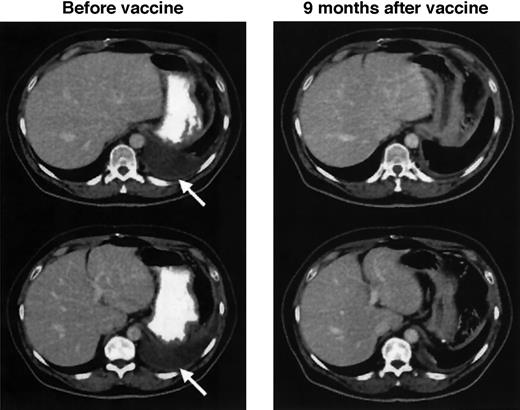
Evidence of antitumor activity was seen in 4 of the 10 patients, including 2 who experienced CR. Complete regression of paracardiac and mediastinal lymph nodes occurred in patient D1.22 This patient's remission lasted 44 months, after which recurrent disease was confined almost exclusively to bone, while all known prior sites of disease were spared. Patient D9 experienced complete regression of axillary (Figure 1), peri-aortic, and iliac lymph nodes. This patient's CR lasted 57 months, when recurrent disease was detected in a single previously uninvolved site (left thyroid). Patient D2 had a PR of peri-aortic and para-iliac nodes lasting 12 months. In patient D3, the only evidence of disease at the time of vaccination was a tumor-specific PCR signal in the bone marrow that became negative after vaccination,22 and she has remained without clinical evidence of disease progression for more than 6 years. Of the 4 patients with clinical responses, 3 of 4 had T-cell anti-Id immune responses, and patient D9 had no detectable in vitro anti-Id response despite having achieved durable CR. There was no correlation of immune or clinical responses to the number of DCs infused. Follow-up status of the 10 patients is listed in Table 1. All 10 patients remain alive at a median follow-up of 64 months after vaccination (range, 47-94 months) and 103 months after diagnosis (range, 81-276 months). Patient D3 has not received any subsequent treatments, and the remaining patients have undergone additional treatments as indicated. Patient D8 remains in CR after booster vaccinations with Id-KLH protein (see below and Table 3).
Complete regression of lymphoma after idiotype-pulsed dendritic cell vaccination.
CT images of the chest of patient D9 show multiple enlarged left axillary lymph nodes that normalized 11 months after vaccination. Arrows indicate sites of disease before vaccination. Similarly sized lymph nodes in the periaortic and iliac regions also completely regressed.
Complete regression of lymphoma after idiotype-pulsed dendritic cell vaccination.
CT images of the chest of patient D9 show multiple enlarged left axillary lymph nodes that normalized 11 months after vaccination. Arrows indicate sites of disease before vaccination. Similarly sized lymph nodes in the periaortic and iliac regions also completely regressed.
Vaccination of patients in first remission
Given the results of the pilot phase, we applied this treatment to a group of 25 patients with advanced-stage follicular lymphoma after an attempt at remission induction with chemotherapy. The characteristics of these patients are shown in Table 2. Chemotherapy was administered to achieve the best clinical response (response plateau with last 2 cycles). Patients were then observed until vaccine production was completed (median, 5 months; range, 3-18 months). After chemotherapy and at the time of vaccination, patients achieved and sustained the following responses: CR in 5 patients, CRu in 11 patients, and PR in 7 patients (designated prevaccine tumor status of NED, MRD, and RD, respectively). Two patients (patients D20 and D27) had progressive disease (PD) at the time of vaccination (both at 9 months after chemotherapy) and completed only 3 vaccinations before they needed chemotherapy and had to be removed from the study. Numbers of DCs administered (median, 16 × 106 over 4 infusions; range, 8-35 × 106 total DCs given, 0.7-12 × 106/infusion) and toxicities observed (less than 20% incidence of transient flulike symptoms) were comparable to those in the pilot phase of the study.
Immune responses of patients in first remission
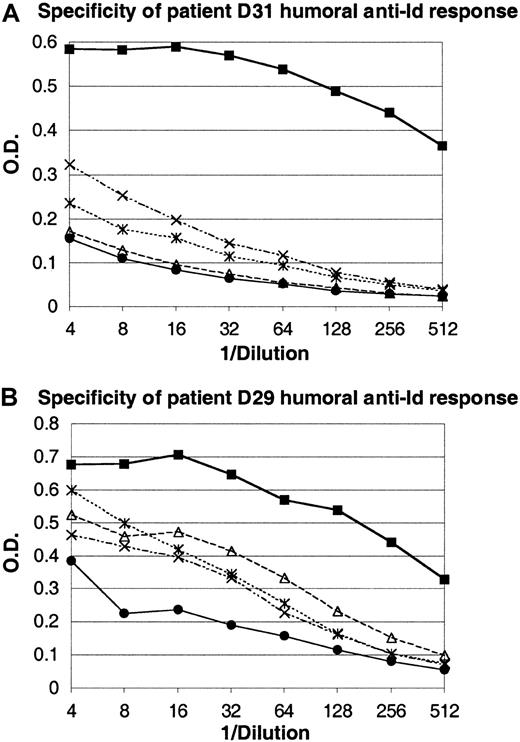
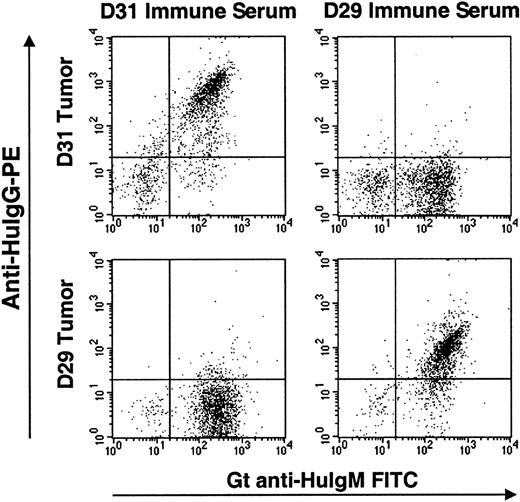
Twenty-three patients completed the series of 4 vaccinations and were considered evaluable for immune responses. All 23 patients mounted humoral and cellular proliferative responses to KLH. Overall, 15 of the 23 (65%) patients mounted T-cell or humoral anti-Id responses. In patients D11 to D22, native, unmodified Id protein was used for DC pulsing as in the pilot study. Five of the 11 evaluable patients in this subgroup mounted T-cell anti-Id responses, and anti-Id antibodies of the IgM class, mirroring the low frequency of anti-Id antibodies seen in the pilot phase, developed in one patient. To improve the induction of Id-specific humoral immunity, 13 additional patients (patients D23-D35) were immunized with DCs pulsed with the Id-KLH conjugate. This modification was based on recent studies of Id-pulsed DC vaccination in a murine lymphoma model, demonstrating that the use of Id-KLH conjugate for DC pulsing led to improved induction of anti-Id antibodies and tumor protection over native Id protein.36Among 12 evaluable patients vaccinated in this manner, 4 mounted cellular anti-Id responses, and 6 developed humoral anti-Id responses. In 4 of these patients, high titers of anti-Id antibodies of the IgG class were detectable. In 2 additional patients, anti-Id antibodies were of the IgM class. The Id specificity of the humoral anti-Id response evoked in 2 representative patients (D29 and D31) after vaccination with Id-KLH–pulsed DCs is shown in Figure2. These serum antibodies showed a high level of binding to the relevant (autologous) tumor Id protein but a low degree of binding to irrelevant Id proteins from other patients' tumors. To further characterize these humoral anti-Id responses, we next asked whether these sera could recognize tumor Id protein as presented in its native conformation by tumor cells. Tumor cells were incubated with either relevant or control prevaccine or postvaccine serum and were stained for bound anti-Id antibodies. Figure3 shows a flow cytometric analysis demonstrating the ability of IgG antibodies in postvaccine serum to specifically recognize autologous tumor cells but not control, irrelevant tumor cells from another patient. Postvaccine sera could specifically recognize tumor cells in 4 of 5 patients for whom tumor cells were available for testing.
Idiotype specificity of the humoral anti-Id response after vaccination with Id-KLH–pulsed dendritic cells.
Prevaccine or postvaccine sera from 2 patients (D31 in panel A, D29 in panel B) were serially diluted and incubated on plates coated with the patients' autologous tumor Id (relevant Id) or with Id proteins from 3 other patient's tumors (irrelevant Id). Binding of IgG anti-Id antibodies was detected by anti–human IgG antibodies conjugated to HRP, and expressed as optical density (O.D.). Panel A: ● indicates prevaccine sera on relevant Id (D31); ▪, postvaccine sera on relevant Id (D31); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; , postvaccine sera on irrelevant Id no. 3. Panel B: ● indicates prevaccine sera on relevant Id (D29); ▪, postvaccine sera on relevant Id (D29); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; and
, postvaccine sera on irrelevant Id no. 3. Panel B: ● indicates prevaccine sera on relevant Id (D29); ▪, postvaccine sera on relevant Id (D29); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; and  , postvaccine sera on irrelevant Id no. 3.
, postvaccine sera on irrelevant Id no. 3.
Idiotype specificity of the humoral anti-Id response after vaccination with Id-KLH–pulsed dendritic cells.
Prevaccine or postvaccine sera from 2 patients (D31 in panel A, D29 in panel B) were serially diluted and incubated on plates coated with the patients' autologous tumor Id (relevant Id) or with Id proteins from 3 other patient's tumors (irrelevant Id). Binding of IgG anti-Id antibodies was detected by anti–human IgG antibodies conjugated to HRP, and expressed as optical density (O.D.). Panel A: ● indicates prevaccine sera on relevant Id (D31); ▪, postvaccine sera on relevant Id (D31); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; , postvaccine sera on irrelevant Id no. 3. Panel B: ● indicates prevaccine sera on relevant Id (D29); ▪, postvaccine sera on relevant Id (D29); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; and
, postvaccine sera on irrelevant Id no. 3. Panel B: ● indicates prevaccine sera on relevant Id (D29); ▪, postvaccine sera on relevant Id (D29); ▵, postvaccine sera on irrelevant Id no. 1; ×, postvaccine sera on irrelevant Id no. 2; and  , postvaccine sera on irrelevant Id no. 3.
, postvaccine sera on irrelevant Id no. 3.
Flow cytometric analysis of tumor-specific cell staining by sera from patients vaccinated with Id-KLH–pulsed dendritic cells.
Tumor cells from patients D31 and D29 (upper and lower sets of panels, respectively) were incubated with autologous postvaccine serum or that of the other patient as a control. Bound anti-Id IgG antibodies were detected by anti-IgG-PE (y-axis), and tumor B cells (IgM+) were counterstained by anti-IgM–FITC (x-axis). Each patient's serum specifically recognizes only the autologous tumor cells but not the control, irrelevant tumor cells.
Flow cytometric analysis of tumor-specific cell staining by sera from patients vaccinated with Id-KLH–pulsed dendritic cells.
Tumor cells from patients D31 and D29 (upper and lower sets of panels, respectively) were incubated with autologous postvaccine serum or that of the other patient as a control. Bound anti-Id IgG antibodies were detected by anti-IgG-PE (y-axis), and tumor B cells (IgM+) were counterstained by anti-IgM–FITC (x-axis). Each patient's serum specifically recognizes only the autologous tumor cells but not the control, irrelevant tumor cells.
Evoked antibodies induce signal transduction in tumor B cells
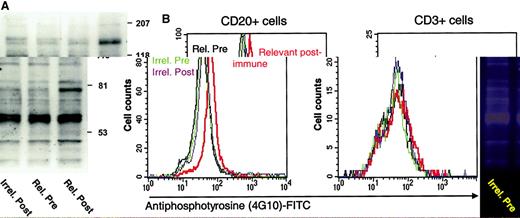
We next investigated whether these serum anti-Id antibodies could mediate signal transduction after binding to tumor cells. When B-cell surface immunoglobulin is engaged and cross-linked by antigen, tyrosine kinases are recruited to phosphorylate tyrosine residues in a number of cellular proteins.37 Anti-Id antibodies, because they also specifically cross-link surface immunoglobulin, can transmit a similar signal. Previous studies have established that tumor regression induced by passively administered anti-Id antibodies38 can be correlated with ability of the antibodies to induce tyrosine phosphorylation in tumor cells.35 However, such effects have not been described using anti-Id antibodies from actively vaccinated patients. Therefore, we treated tumor cells with immune sera and assayed for the induction of tyrosine phosphorylation. As shown in the Western blot in Figure4A, only cells treated with the relevant postvaccine serum displayed the distinct pattern of signal intensities, with more prominent bands at 140 and 75 kd. To verify and further quantitate these effects on a per-cell basis, we used flow cytometric analysis for cellular phosphotyrosine content. Using this method, permeabilized tumor cells stained for total intracellular phosphotyrosine could be counterstained for surface markers to determine the population accounting for the increased signal. As shown in Figure 4B, only treatment of cells with the relevant postimmune serum results in a rightward shift in signal, indicating increased phosphotyrosine content within the CD20+ tumor cell population (left panel), whereas no such shift is seen in the CD3+ tumor-infiltrating T cells (right panel). Mean fluorescence intensities of the phosphotyrosine (4G10) signal induced in the CD20+ cells by prevaccine serum, postvaccine serum, or anti-μm antibodies (positive control) were 48, 103, and 110, respectively. These results indicate that serum anti-Id antibodies induced by active vaccination can initiate signal transduction specifically in tumor B cells. Such effects were observed in 2 of the 3 patients for whom tumor cells were available for testing.
Idiotype-immune serum induces tyrosine phosphorylation specifically in tumor B cells.
(A) Detection of tyrosine phosphorylation in tumor cells by Western blotting. Tumor cells from patient D31 were incubated with the indicated sera and then lysed. Solubilized proteins were separated by SDS-PAGE and transferred to nitrocellulose. The blot was stained with antiphosphotyrosine antibody 4G10. (B) Detection of tyrosine phosphorylation in tumor cells by flow cytometry. Tumor cells from patient D31 were treated with the indicated sera, permeabilized, and stained with antibody 4G10 coupled to FITC. Phosphotyrosine content was then quantitated in aliquots of cells counterstained with either phycoerythrin-labeled anti-CD20 or anti-CD3.
Idiotype-immune serum induces tyrosine phosphorylation specifically in tumor B cells.
(A) Detection of tyrosine phosphorylation in tumor cells by Western blotting. Tumor cells from patient D31 were incubated with the indicated sera and then lysed. Solubilized proteins were separated by SDS-PAGE and transferred to nitrocellulose. The blot was stained with antiphosphotyrosine antibody 4G10. (B) Detection of tyrosine phosphorylation in tumor cells by flow cytometry. Tumor cells from patient D31 were treated with the indicated sera, permeabilized, and stained with antibody 4G10 coupled to FITC. Phosphotyrosine content was then quantitated in aliquots of cells counterstained with either phycoerythrin-labeled anti-CD20 or anti-CD3.
Clinical responses and follow-up of patients in first remission
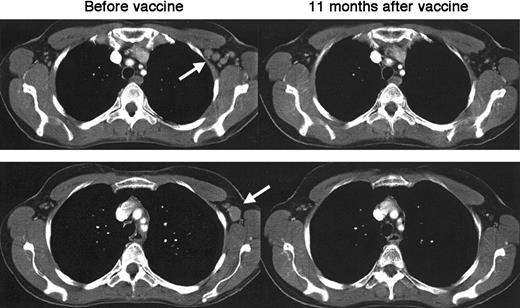
In evaluable patients with less than CR at the time of vaccination (n = 18), sustained regression of residual tumor (in bone marrow, lymph nodes, or pleura) was seen in 4 patients (22%; patients D13, D15, D16, and D29). Patient D29, whose postvaccine sera specifically bound to tumor cells, had regression of lymphomatous pleural disease after vaccination (Figure 5). Pleural effusion shown in the CT images of the chest was refractory to cyclophosphamide, vincristine, and prednisone (CVP) chemotherapy and remained cytologically positive before vaccination. However, 9 months after vaccination with Id-KLH–pulsed DCs, it had almost completely resolved. This patient also had a marked reduction in bone marrow disease after vaccination and has remained without disease progression for more than 21 months. Long-term follow-up of patients vaccinated during first remission shows that 16 of 23 (70%) remain progression free at a median of 43 months after the completion of chemotherapy (range, 23-56 months). There has been a single death (patient D21) associated with a transformation to aggressive diffuse large-cell lymphoma. Six of 7 (86%) patients mounting humoral anti-Id responses have remained progression free compared with 10 of 16 (63%) without antibody responses, though this difference is not statistically significant (P = .40, log-rank statistic). The proportion of patients remaining progression free did not differ between patients mounting T-cell or humoral responses (10 of 15, 67%) versus those without immune responses (6 of 8, 75%).
Regression of lymphomatous pleural effusion after vaccination with Id-KLH–pulsed dendritic cells.
Despite chemotherapy, a left-sided pleural effusion persisted in patient D29 and remained cytologically positive before vaccination. Near-complete resolution is noted 9 months after completing vaccination with Id-KLH–pulsed dendritic cells.
Regression of lymphomatous pleural effusion after vaccination with Id-KLH–pulsed dendritic cells.
Despite chemotherapy, a left-sided pleural effusion persisted in patient D29 and remained cytologically positive before vaccination. Near-complete resolution is noted 9 months after completing vaccination with Id-KLH–pulsed dendritic cells.
Administration of booster vaccinations with Id-KLH protein plus chemical adjuvant
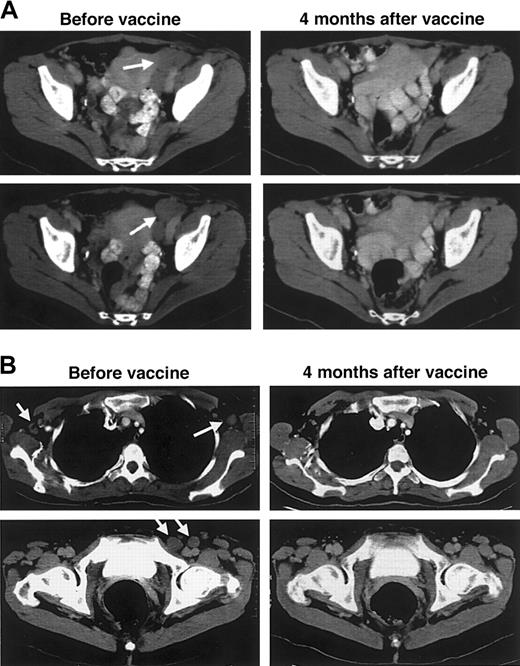
Patients with persistent or relapsed tumors after DC vaccination were offered booster vaccination with Id-KLH protein without DCs, as performed in our previous vaccine trials.7 Biopsy samples were again taken from tumors of 4 patients before revaccination (patients D5, D6, D10, D12). Each was found to have retained expression of surface immunoglobulin heavy and light chains of the same classes found on the original tumor. Therapy consisted of 5 subcutaneous injections of the same Id protein used for DC vaccination yet was coupled to KLH and was emulsified in a chemical adjuvant.7Six patients were treated—5 from the pilot study who had not fully responded and 1 who had a relapse after DC vaccination given during first remission (Table 3). The Id-KLH booster vaccinations were given at different lengths of time after the original DC vaccination series (range, 8-33 months), and all patients had multiple sites of progressive, evaluable tumor. Three of the patients had immune responses to the booster vaccine series; 2 were T-cell responses and one was a humoral response (Table 3). These responses were of the same type (T cell, humoral) and of similar magnitude to those each patient previously mounted to the DC vaccine, though the earlier responses had waned in the interval before boosting. Each of the 3 immune responders also had a corresponding clinical response; one PR lasting 14 months, one CR ongoing at 48+ months, and one CRu lasting 16 months. The latter patient (patient D12), treated 18 months after relapse following DC vaccination given in first remission, had extensive lymphadenopathy involving cervical, axillary, iliac, and inguinal lymph nodes, including a pelvic mass measuring more than 4 × 5 × 6 cm. Bone marrow was also involved. Within 2 weeks of the first injection of Id-KLH, all palpable lymphadenopathy began to rapidly regress, and physical examination results became normal in the next 4 weeks. Normalization of all nodes was documented by CT at 1 and 4 months after the completion of vaccination (Figure6A-B), and the only evidence of tumor was equivocal findings in the bone marrow. At 16 months after vaccination, small recurrent lymph nodes were detected in the right side of the neck. A repeat series of Id-KLH vaccinations (designated vaccine course 3) was then administered, and tumor regression occurred once again, with CR documented 7 months later accompanied by an ongoing Id-specific T-cell proliferative response (Table 3).
Regression of tumors after booster vaccination with Id-KLH + chemical adjuvant.
Patient D12 was given Id-pulsed DC vaccination during first remission after chemotherapy but relapsed 15 months later. Subsequent to the development of widespread disease 18 months later, she was given injections of Id-KLH protein plus a chemical adjuvant. (A) CT images of the pelvis show that bulky left-sided pelvic lymph nodes have completely regressed 4 months after booster vaccinations. (B) Regression of axillary (top panels) and inguinal (bottom panels) lymph nodes after booster vaccinations. Arrows indicate sites of disease before vaccination.
Regression of tumors after booster vaccination with Id-KLH + chemical adjuvant.
Patient D12 was given Id-pulsed DC vaccination during first remission after chemotherapy but relapsed 15 months later. Subsequent to the development of widespread disease 18 months later, she was given injections of Id-KLH protein plus a chemical adjuvant. (A) CT images of the pelvis show that bulky left-sided pelvic lymph nodes have completely regressed 4 months after booster vaccinations. (B) Regression of axillary (top panels) and inguinal (bottom panels) lymph nodes after booster vaccinations. Arrows indicate sites of disease before vaccination.
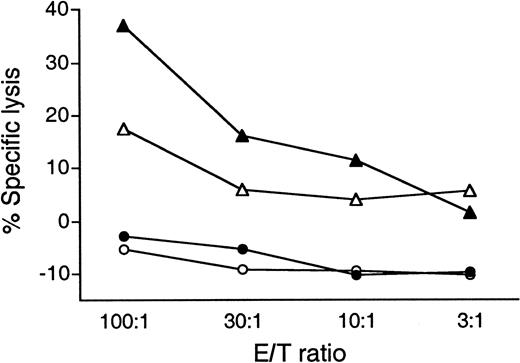
Given this patient's repeated tumor regression in response to vaccination, we looked for evidence of antitumor cytolytic activity in peripheral blood lymphocyte (PBL) effectors after restimulation with CD40L-activated tumor cells.39 As shown in Figure7, restimulated PBLs obtained after vaccine course 3 exhibited specific cytotoxicity toward autologous tumor cells. PBL obtained before vaccine course 3 had lower, but measurable, cytolytic activity. There was no significant lysis of purified autologous normal B cells (obtained after the attainment of CR) or of follicular lymphoma cells from a patient of unknown HLA haplotype (data not shown).
Tumor-specific cytolysis by peripheral blood lymphocytes derived from patient D12 before and after a repeat series of Id-KLH vaccinations (third course of vaccinations).
Prevaccine and postvaccine PBMC effectors were restimulated using 2 weekly cycles of CD40L-activated autologous tumor cells plus interleukin-2. After 14 days, the ability of effectors to lyse unmodified, cryopreserved tumor cells was assessed in a 4-hour51Cr release assay. Purified autologous normal peripheral blood B cells serve as negative control targets. The percentage specific lysis is expressed as the mean of triplicate values. Results are representative of 3 independent experiments. ▴ indicates postvaccine course no. 3 versus tumor targets; ▵ indicates prevaccine course no. 3 versus tumor targets; ● indicates postvaccine course no. 3 versus normal B cells; and ○ indicates prevaccine course no. 3 versus normal B cells.
Tumor-specific cytolysis by peripheral blood lymphocytes derived from patient D12 before and after a repeat series of Id-KLH vaccinations (third course of vaccinations).
Prevaccine and postvaccine PBMC effectors were restimulated using 2 weekly cycles of CD40L-activated autologous tumor cells plus interleukin-2. After 14 days, the ability of effectors to lyse unmodified, cryopreserved tumor cells was assessed in a 4-hour51Cr release assay. Purified autologous normal peripheral blood B cells serve as negative control targets. The percentage specific lysis is expressed as the mean of triplicate values. Results are representative of 3 independent experiments. ▴ indicates postvaccine course no. 3 versus tumor targets; ▵ indicates prevaccine course no. 3 versus tumor targets; ● indicates postvaccine course no. 3 versus normal B cells; and ○ indicates prevaccine course no. 3 versus normal B cells.
Discussion
Our earliest studies of Id vaccination for NHL focused on describing the induction of anti-Id immune responses6 and their correlation with favorable clinical outcomes.7 With our more recently developed DC vaccine approach, we directly assessed antitumor effects by vaccination of larger numbers of patients with measurable tumors. In the pilot phase of our DC study, we observed antitumor activity in 4 of 10 patients consisting of 3 objective radiographic responses and 1 molecular complete response. In 3 patients, these responses were complete and durable (44, 57, and 73+ months). Among patients with residual disease in first remission after chemotherapy, 4 of 18 also had objective reductions in measured tumor burden. Two additional patients had tumor shrinkage after Id-KLH booster vaccinations. Thus, 10 of 28 (36%) patients with evaluable tumor had objective antitumor effects after our Id vaccine interventions. A novel finding in this study was the occurrence of tumor regression in patients with resistant or relapsed disease after retreatment with booster Id-KLH protein vaccinations. These observed tumor regressions are instructive of several points. First, revaccination using the same Id protein, reformulated as Id-KLH plus chemical adjuvant, can lead to tumor regression despite the lack of a clinical response to the initial Id-pulsed DC vaccine. Second, even relatively large tumor burdens can undergo regression after Id-KLH vaccination (Figure 6). Whether the prior immunization with Id-pulsed DCs contributed to the observed antitumor responses to Id-KLH is unknown. However, the rapid regression of tumor observed in patient D12, beginning within 2 weeks of Id-KLH initiation, suggests stimulation of a memory response, primed either through the previously administered DC vaccine or through endogenous mechanisms. Taken together, these observations provide unequivocal evidence of the clinical activity of Id vaccination.
Induction of anti-Id immune responses was another important endpoint of our study. The overall (humoral or T-cell) immune response rate of 65% compared favorably with the 49% response rate seen in our earlier study of Id-KLH vaccination.7 In keeping with the principal role of DCs in priming T-cell responses, vaccination with Id-pulsed DCs preferentially induced cellular responses over antibody responses (Tables 1-2). Although 62% of patients given native Id-pulsed DCs mounted cellular responses, only 14% produced anti-Id antibodies. However, the ability of DCs to function as adjuvants for the induction of a humoral anti-Id response varied according to the form of Id used for DC pulsing, as previously observed in a murine lymphoma model.36 Vaccination using DCs pulsed with native Id induced detectable anti-Id antibodies, always of the IgM class, in only 3 patients. Use of the Id-KLH conjugate for DC pulsing led to a greater frequency of humoral anti-Id responses (6 of 12 patients; 50%) including high-titer IgG antibodies, once again pointing to the capacity of DCs to prime B-cell responses.18 36
Induction of a humoral anti-Id response is thought to be an important goal of Id vaccination for several reasons. First, passively administered anti-Id murine monoclonal antibodies are able to induce tumor regression in two thirds of patients.38 In addition, in murine lymphoma models of Id vaccination, anti-Id antibodies alone can be sufficient to provide tumor protection.4,13,36,40Thus, a broad anti-Id immune response, including anti-Id T cells and antibodies, might have greater antitumor effects. Antibodies induced by vaccination with Id-pulsed DCs were highly relevant to tumor cells because they could bind selectively to autologous tumor cells in addition to the isolated Id protein. Furthermore, these antibodies had the capacity to stimulate signal transduction in tumor cells, as indicated by increased tyrosine phosphorylation. Induction of tyrosine phosphorylation by cross-linking surface immunoglobulin on B-lymphoma cells has been shown to be an initial step in the cascade of events leading to apoptosis,37,41,42 suggesting a direct mechanism whereby anti-Id antibodies may exert antitumor effects. Vuist et al35 previously demonstrated that in patients treated with passive anti-Id monoclonal antibodies, achievement of a clinical response was highly correlated with the ability of the antibodies to stimulate tyrosine phosphorylation in tumor cells. Similarly, we have found that among our treated patients, serum anti-Id antibodies capable of binding to tumor cells could induce tyrosine phosphorylation in 2 of 3 patients. Whether this signal transduction response leads to tumor cell apoptosis, correlates with clinical outcome, or can be detected in patients treated with other types of Id vaccines is the subject of ongoing studies.
Overall and progression-free survival of our selected group of 35 patients appears favorable in comparison to that historically observed in follicular NHL,43 though a controlled trial would be necessary to ascribe long-term outcomes to the vaccine interventions. Given the clinically significant tumor regressions observed in this study, however, it is possible that the natural history of the disease was altered in some patients. Bendandi et al39 have also described excellent long-term disease-free survival in 18 of 20 patients (median since chemotherapy, 42 months) with follicular lymphoma vaccinated with Id-KLH plus granulocyte macrophage–colony-stimulating factor. Because their study was limited to patients achieving CR after chemotherapy, few comparisons can be made with our current trial in which 20 of 25 patients had less than CR at the time of vaccination. Nevertheless, all but 1 of our 5 patients in CR has remained free of disease progression (Table 2).
Bendandi et al39 measured tumor-specific cytokine secretion by patient PBL cocultured with autologous tumor cells in 19 of 20 patients, and they measured antitumor cytotoxic T-cell activity in a subset of these patients. This 95% overall immune response rate is higher than that observed in the current study. The difference may be explained by the use of alternative assay techniques, their use of Id-KLH plus granulocyte macrophage–colony-stimulating factor adjuvant, or by selection of a more favorable first CR patient population. Indeed it does appear from previous work7 and our current study that immune responses after Id vaccination are more frequent in patients in CR at the time of vaccination (4 of 5 immune response-positive in the current study). Although we did not routinely measure antitumor cytotoxic T cells in our patients, in the one patient studied (patient D12) we did find considerable tumor-specific cytolytic activity in PBLs obtained after repeat booster vaccinations with Id-KLH and objective tumor regressions.
We believe that our findings have several implications for the design of future lymphoma vaccine trials. Our results in NHL once again confirm tumor antigen-pulsed DC vaccination as a promising platform for cancer immunotherapy.20 This approach is more technically demanding than traditional protein (ie, Id-KLH) vaccination methods, and it remains to be proven superior in formal comparisons. Before embarking on such studies, it will be important to further refine this technique, particularly with regard to the types and numbers of antigen-pulsed DCs administered.19,20 Our use of peripheral blood DCs isolated from leukapheresis product was based on the demonstrated T-cell stimulatory capacity of this DC fraction32,33,44 and its observed clinical activity early in our pilot study.22 Numbers of DCs obtainable using this method, approximately 5 to 10 million per infusion, do not allow for dose-escalation studies. However, newer methodologies for propagating DCs from monocytes45 or CD34+ hematopoietic progenitor cells46 or from the mobilization of peripheral blood DCs using Flt3 ligand28,47 now allow DC dose escalation to more than 108 cells. The use of greater numbers of antigen-pulsed cells, cultured under conditions that promote maturation and activation of DCs,48-50 may provide even greater potency at inducing anti-Id immune and clinical responses.
Our observation of significant clinical activity in DC-vaccinated patients after booster vaccination with Id-KLH suggests a potential new strategy for the immunotherapy of follicular NHL, and further studies using Id-KLH vaccines in tandem with Id-pulsed DCs are in progress.51 These observations may also herald a shift in our view of the optimal timing of Id vaccination. Earlier studies of Id vaccination have been principally confined to patients in remission after cytoreductive chemotherapy,6,7,39 based on the view that vaccination might be more effective when the level of tumor is minimal. Thus, little is known about the therapeutic effects of Id-KLH vaccination in patients with progressive lymphoma. Indeed, in our previous study, in vitro anti-Id immune responses were more frequently detectable in patients in CR at the time of vaccination than in those with residual disease,7 suggesting that the presence of tumor might impair the host's ability to mount an effective antitumor response. The finding that substantial tumor burdens may regress after Id-KLH vaccination (Table 3; Figure 6) provides rationale for further study of Id-KLH vaccination in patients with relapsed or even untreated follicular NHL. In the latter case, avoidance of the strongly immunosuppressive effects of chemotherapy before immunization might prove to be an important advantage.
Given its validation as a susceptible target, lymphoma idiotype will likely remain a highly relevant tumor antigen for NHL immunotherapy despite the need to produce patient-specific material. In multiple clinical trials, Id vaccine production has been shown to be feasible, and evidence of clinical activity has been obtained.6,22,39 Moreover, novel genetic approaches are now yielding recombinant forms of Id and will continue to make large-scale production of Id vaccines more practical.52-56These technologies offer new sources of Id for loading into DCs. In the future, as additional and possibly shared NHL-associated tumor antigens are described, these too will be candidates for evaluation using DC-based vaccine strategies.
We thank Hendrik Veelken for critical reading of the manuscript; Dan Denney and Diane Ingolia for help in producing recombinant tumor Id proteins; Yong-Jun Liu at DNAX for the gift of CD40 ligand-transfected L cells; and Patty Ciesla for assistance in preparing the manuscript.
Supported by National Institutes of Health grants HL57443 and CA33399. J.M.T. and T.A.D. are recipients of Clinical Associate Physician awards from the National Institutes of Health (RR-00070-CAP). R.L. is an American Cancer Society Clinical Research Professor.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Ronald Levy, Dept of Medicine, Division of Oncology, Stanford University Medical Center, CCSR Rm 1105, Stanford, CA 94305-5151; e-mail: levy@stanford.edu.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal