Abstract
T-cell receptors (TCRs) of a series of minor histocompatibility antigen (mHag) HA-1–specific cytotoxic T-cell (CTL) clones isolated from 3 unrelated patients have been shown to use the same BV6S4A2 segment with conserved amino acids in the CDR3Vβ region. This suggests that different HA-1–specific TCRs interact similarly to the HA-1 antigen presented by the HLA-A2 molecule. The mHag HA-1 forms an immunogenic complex with HLA-A2 and induces strong alloimmune responses after stem cell transplantation (SCT). It was questioned, therefore, whether clonal and polyclonal HA-1–specific CTL responses can be antagonized by a single TCR antagonistic peptide. Functional analysis and molecular modeling of single and double amino acid substitutions of TCR contact residues, adjacent residues, and HLA-A2 binding residues resulted in 4 peptides with high affinity for HLA-A2 and with the capacity to inhibit the lysis of endogenously HA-1–expressing EBV-BLCL by 3 different HA-1–specific CTL clones. These peptides also efficiently antagonized HA-1–specific polyclonal CTL lines derived from 3 patients and significantly reduced the number of interferon-γ–producing HA-1–specific CTL of a patient with graft-versus-host disease after HA-1–mismatched SCT. These data show that general TCR antagonists can be developed that inhibit HLA-A2–restricted HA-1–specific CTL responses on the clonal and the polyclonal level and that TCR antagonists may modulate the immunodominant mHag HA-1 responses.
Introduction
T cells are activated by the interaction between the T-cell receptor (TCR) and its specific ligand—the combined structure of a major histocompatibility complex (MHC) allele and a peptide—on stimulatory cells. Specificity of TCR recognition is determined by the unique rearrangement of variable (V), diversity (D), and joining (J) gene segments for each of the 2 TCR chains. Two hypervariable regions are encoded by the V genes—complementarity-determining regions (CDRs) 1 and 2. Additional variation is achieved by the introduction of random nucleotides in the V/D/J junctional region, the CDR3 region. Four TCRs have been crystallized in conjunction with their MHC-peptide ligand.1-5 Two of these crystals consist of HLA-A2 presenting the HTLV-1 Tax peptide and 2 different TCRs, A6 and B7.1,5 In addition the murine H-2Kb molecule was crystallized with the dEV8 and VSV8 peptides and with their specific TCRs, 2C and N15, respectively.3 4 All 4 crystal structures revealed a blueprint for interaction between TCR chains and regions and the MHC and peptide. TCR is positioned diagonally on top of the MHC groove, so that Vα overlies the MHCα2 helix and Vβ overlies the MHCα1 helix. The TCR Vα chain thereby interacts with the N-terminal part of the peptide, whereas the Vβ chain is situated at the C-terminal part of the peptide. CDR1 and CDR3 regions of the Vα and Vβ chains interact with the peptide, whereas CDR2 regions mainly interact with MHC.
Subtle changes in the antigenic peptide can result in altered recognition or no recognition by the T cell. Partial agonists cause decreased activation of the T cell or induce a limited set of effector functions by the T cells. TCR antagonists are nonstimulatory ligands that compete with the agonist for the TCR.6-8 Hence, they can prevent proliferation, cytokine production, and cytotoxicity by MHC class II and class I restricted T-cell clones.9,10Antagonists have been generated in vitro by single or double amino acid substitution of the antigenic peptide.9-11 In addition, naturally occurring variants of human immunodeficiency virus, hepatitis B virus, and malaria encoding mutated epitopes that could down-regulate the CTL response by TCR antagonism have been described.12-14 TCR antagonists have been proposed as a potential therapy for autoimmune diseases. Although TCR antagonists have been developed and have been shown to decrease the severity of autoimmune diseases, such as experimental allergic encephalomyelitis (EAE) or adjuvant arthritis (AA),11,15-17 inhibition of polyclonal responses may be difficult to establish. Each different TCR apparently requires a different TCR antagonist.10,18Numerous T-cell responses have been described that use a restricted set of TCR Vα and or Vβ segments.19-24 Because those homologous TCRs may have similar interactions with the MHC antigenic peptide complex, it may be more feasible to develop a single TCR antagonist for these restricted T-cell responses.
Human minor histocompatibility antigen (mHag) HA-1 is a nonameric peptide VLHDDLLEA encoded by one allele of the KIAA0223gene.25 HA-1− cells express another allele of the KIAA0223 gene with 2 nucleotides difference resulting in a single amino acid difference. This single amino acid difference consists of an H to R substitution in the antigenic sequence. The HA-1R allelic peptide has a significantly lower binding affinity to HLA-A2 and cannot be detected in HLA-A2–binding peptides isolated from HA-1R expressing EBV-BLCL.
Five different CTL clones recognizing mHag HA-1 in the context of HLA-A2 express the same Vβ chain (BV6S4A2) and contain similar amino acids in the Vβ CDR3 region.24 HA-1–specific T-cell clones were isolated from several unrelated patients after stem cell transplantation (SCT).26,27 Incompatibility for mHag HA-1 was shown to be associated with the development of acute grade II-IV graft-versus-host disease (GvHD) after HLA-identical SCT.28,29 In addition, HA-1–specific CTLs could be demonstrated in the peripheral blood of stem cell recipients as early as 14 days after SCT and were significantly increased during acute GvHD.30 These data suggest an immunodominant behavior of HA-1.
Our goal was to develop antagonist peptides for the immunodominant HA-1–specific T cells. First, TCR contact residues in the HA-1 antigen were determined for 2 different HA-1–specific CTL clones. These amino acids and 2 adjacent amino acids were substituted with all 20 amino acids and tested for agonistic and antagonistic activity in cytotoxicity assays. To achieve physiologically relevant antagonistic activity, the inhibition of lysis of an endogenously HA-1–expressing EBV- BLCL by the HA-1–specific CTL was determined. Candidate antagonistic peptides were tested for their effect on HA-1–specific CTL clones, polyclonal CTL lines, and patient peripheral blood mononuclear cells (PBMCs) derived at the time of active GvHD after HA-1–mismatched SCT. The results show that it may be feasible to develop general antagonistic peptides that can inhibit T-cell recognition of the natural HLA-A2–HA-1 ligand.
Materials and methods
Cell lines
CD8+ HLA-A2–restricted T-cell clones 3HA15 and 5W38, clone 15, and the polyclonal CTL line A2Bx were derived from PBMCs of 2 patients who had undergone HLA-identical SCT, and they were cultured as described.28 CTL lines P.1.1 and K.1.1 were generated in vitro from HLA-A2+ HA-1− stem cell donors using HA-1 peptide-pulsed dendritic cells according to protocols described previously.31 CTL clones and lines were cultured for 3 days in RPMI 1640 medium containing 3 mM L-glutamine, penicillin–streptomycin, 15% human serum, 20 U/mL recombinant interleukin 2 and 10% T-cell growth factor before cytotoxicity assays. EBV-BLCL H02, H03, H05, and H06 and T2 were cultured in RPMI 1640 medium containing 3 mM L-glutamine, penicillin–streptomycin, and 10% fetal calf serum.
Peptides
Peptides were synthesized on an Abimed 422 multiple peptide synthesizer (Abimed Analyes-Technik, Langenfeld, Germany) using fluorenylmethoxycarbonyl-protected amino acids and TentagelS-AC resins (Rapp, Tubingen, Germany) as described. Purity of the peptides was determined by reverse-phase high-pressure liquid chromatography and was always higher than 90%. Peptides were weighed, and 10-mg/mL stock solutions were prepared in dimethyl sulfoxide. Peptides were further diluted in Hanks balanced salt solution containing 50 mM HEPES.
Peptide binding to HLA-A2
Binding of candidate peptides to HLA-A2 molecules was measured using recombinant HLA-A2 molecules in a cell-free competition-based assay as described previously.32 Relative binding affinities of the peptides are expressed as the IC50 values (concentration of the peptide that inhibits 50% of the binding of the reference peptide). HA-2 peptide (amino acid sequence, YIGEVLVSV) is used throughout as a negative control and binds effectively to HLA-A2 with IC50 value of 6.7 nM.32
Cytotoxicity assay
Chromium Cr 51-labeled target cells (2500 cells) in 25 μL RPMI 1640 medium with 3 mM L-glutamine and 15% human serum were incubated with 25 μL peptide solution for 30 minutes at 37°C. CTLs were then added, resulting in a final volume of 150 μL. After 4 hours at 37°C, 51Cr release in the supernatant was measured by standard methodology. HA-1− T2 or EBV-BLCL was used to determine the agonistic activity of the substituted peptides. Endogenously HA-1 expressing EBV-BLCL was used to determine the antagonistic activity of the substituted peptides. Peptide concentrations ranged from 5 μg/mL to 0.5 ng/mL during preincubation, giving final concentrations of 1.67 μg/mL to 0.167 ng/mL. Percentage inhibition was calculated as follows: 1 − (% Lysis peptide-loaded HA-1+ EBV-BLCL)/(% Lysis HA-1+ EBV-BLCL) × 100.
Interferon-γ ELISpot assay
PBMCs (1 × 105) were cocultured for 88 hours with equal numbers of HA-1 peptide-pulsed (0.1 μg/mL; 3 hours) donor EBV-BLCL in 96-well round-bottomed microtiter plates in the presence or absence of serial dilutions of antagonist peptides. After 72 hours the cells were transferred to the wells of anti–interferon (IFN)–γ–coated ELISpot plates (Mabtech, Cincinnati, OH). After culturing for another 20 hours, the IFN-γ spots were visualized using the commercial ELISpot (Mabtech) kit according to the instructions of the manufacturer. The number of IFN-γ spots in each well was determined with the use of a fully automated inverted light microscope.
Molecular modeling
All molecular modeling operations were performed on a Silicon Graphics O2 workstation (Mountain View, CA, USA). Quanta 4.0 molecular modeling suite (Molecular Simulations, San Diego, CA) was used as the software package. Several HLA-A2.1–peptide complexes were retrieved from the PDB, the Brookhaven Protein Databank,33including reference codes 1hhg, 1hhh, 1hhi, 1hhj, and 1hhk. The crystal structure of the complex between HLA-A2, a TAX peptide, and its TCR (A6) was kindly provided by Dr P. Ghosh.1 This latter structure holds the same peptide (LLFGYPVYV) present in the 1hhk file, though in a slightly different conformation. The modeling procedure consisted of the following steps. First, the peptide in the 1hhj file (ILKEPVHGV) was used as a template for the generation of a VLHDDLLEV conformer, one of the relevant peptides in the current study, because they share the highest homology. The directionality of the side chains in VLHDDLLEV was based on the conformation of the peptide in the HLA-TCR complex. Second, the peptide backbone of VLHDDLLEV was superimposed on the backbone of the peptide in the HLA-TCR complex as closely as possible. Third, TCR clone 5W38 was compared with crystalline TCR by means of protein homology modeling. Large, mutually conserved sequence elements in both TCRs served as anchor points to define the borders of the hypervariable regions. The TCRα chain was most in contact with the peptides, with important sequence differences between the 2 TCRs as indicated in Table1.
Theoretical alignment of TCRα amino acid sequences of HLA-A2–restricted T-cell clones used for molecular modeling
| Clone . | Fragment V . | Fragment N . | Fragment J . |
|---|---|---|---|
| A6 | PSDSATYLCA | VT | TDSWGKLQFG |
| 5W38 | GLYLCA | GARGWEN | SNSGYALNFG |
| Clone . | Fragment V . | Fragment N . | Fragment J . |
|---|---|---|---|
| A6 | PSDSATYLCA | VT | TDSWGKLQFG |
| 5W38 | GLYLCA | GARGWEN | SNSGYALNFG |
Homology modeling of fragment N is most speculative. Fragments V and J both share significant homology among the 2 receptors, and an initial model for fragments V and J of clone 5W38 was easily obtained by the on-screen mutation of A6. For fragment N, the following procedure was used. A sequence analogy search in the PDB identified reference code 1aih with the element GARWSEA. Highest homology of all sequences available with the N fragment (GARGWEN) was in clone 5W38. Three-dimensional topology of GARWSEA (slightly alpha-helical in nature) was used as a template for the conformation of the GARGWEN element. The resultant GARGWEN conformer was patched onto the crystal structure of A6 after on-screen excision of the di-peptidic element VT. This procedure required energy minimization of the new fragment. For this purpose Quanta's CHARMm force field was used in a default operating mode.
Fourth, the eventual VLHDDLLEV (peptide)-5W38 (TCR) complex was further energetically optimized by keeping the peptide fixed and the variable loops of 5W38 flexible. Similarly, the variable regions of the TCRβ chain of clone 5W38 was modeled based on the crystal structure of the equivalent A6 structure. The NDN segment in A6 (RPGLAGGR) was replaced by the 5W38 equivalent NDN fragment RSTGGVAL. All figures in the current study were based on this final structure.
Results
Determination of general TCR contact residues in HA-1
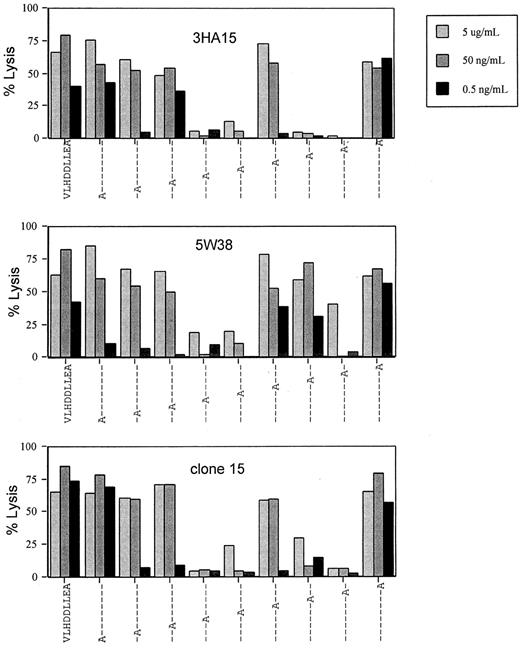
The interaction of 3 HA-1–specific HLA-A2–restricted CTL clones—3HA15, 5W38, and clone 15—with the HA-1 peptide was studied by analyzing the recognition of alanine-substituted peptides. All 3 clones express different TCRs but share the same BV6S4A2 segment. T-cell clones 3HA15 and clone 15 even share the same Vα- and β-chains but are still different in J sequences and CDR3 regions (Table2). Binding capacity of alanine-substituted peptides to the HLA-A2 molecule was first determined using a cell-free assay based on the inhibition of binding of a fluorescence peptide. Except for the peptides with alanine at positions 2 and 6, all alanine-substituted peptides were good HLA-A2 binders (data not shown). In addition, the alanine-substituted peptides were titrated on the TAP-deficient and efficient exogenous peptide-presenting T2 cell line and were evaluated for recognition by the 3 CTL clones in a cytotoxicity assay (Figure1). Substitution of both aspartic acids at positions 4 and 5 and of the glutamic acid at position 8 prevented recognition by all 3 clones. Substitution of leucine at position 7 inhibited the recognition of 2 of 3 clones, 3HA15 and clone 15. These data suggested that positions 4, 5, and 8 are important for TCR recognition of all 3 clones.
Alignment of amino acid sequences of TCRVα and TCRVβ CDR3 regions of HA-1–specific HLA-A2–restricted CTL clones
| T-cell clone . | Vα . | N . | Jα . | Vβ . | NDN . | Jβ . |
|---|---|---|---|---|---|---|
| 3HA15 | AV3S1 | 22 | BV6S4A2 | 2S7 | ||
| ASYFCAT | AVI | SSGSARQLTFGSGTQL | AMYLCASS | LVRT | YEQYFGPGTRLTVT | |
| 5W38 | AV10S1A3T | 41 | BV6S4A2 | 1S1 | ||
| GLYLCAG | ARWGE | NSNSGYALNFGKGTSL | AMYLCAS | RSTGGVAL | TEAFFGQGTRLTVV | |
| Clone 15 | AV3S1 | 39 | BV6S4A2 | 1S4 | ||
| ASYF | FL | NNAGNMLTFGGGTRL | AMYLCASS | STRL | EKLFFGSGTQLSVL |
| T-cell clone . | Vα . | N . | Jα . | Vβ . | NDN . | Jβ . |
|---|---|---|---|---|---|---|
| 3HA15 | AV3S1 | 22 | BV6S4A2 | 2S7 | ||
| ASYFCAT | AVI | SSGSARQLTFGSGTQL | AMYLCASS | LVRT | YEQYFGPGTRLTVT | |
| 5W38 | AV10S1A3T | 41 | BV6S4A2 | 1S1 | ||
| GLYLCAG | ARWGE | NSNSGYALNFGKGTSL | AMYLCAS | RSTGGVAL | TEAFFGQGTRLTVV | |
| Clone 15 | AV3S1 | 39 | BV6S4A2 | 1S4 | ||
| ASYF | FL | NNAGNMLTFGGGTRL | AMYLCASS | STRL | EKLFFGSGTQLSVL |
Agonistic activity of single alanine substitutions of HA-1 on HA-1–specific CTL clones 3HA15 and 5W38 and clone 15.
HA-1− T2 cells were incubated at 37°C with the indicated peptide concentrations. After 30 minutes CTLs were added, and percentage lysis was determined 4 hours later.
Agonistic activity of single alanine substitutions of HA-1 on HA-1–specific CTL clones 3HA15 and 5W38 and clone 15.
HA-1− T2 cells were incubated at 37°C with the indicated peptide concentrations. After 30 minutes CTLs were added, and percentage lysis was determined 4 hours later.
Molecular modeling of the VLHDDLLEA–5W38 peptide–TCR complex
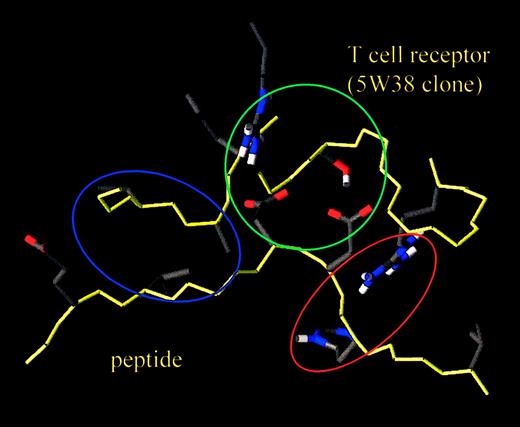
Proposed interactions between the HA-1 peptide and the T-cell clones were further studied by a computer model of the HLA-A2 molecule with the HA-1 peptide in conjunction with 5W38. The model was based on the crystal structure of HLA-A2 with the tax peptide and its specific TCR, A6. In Figure 2 a close-up of the peptide binding site on the TCR clone 5W38 is represented. Aspartic acid side chains on positions 4 and 5 are in close contact with the N and J fragments of the clone. The first serine in Jα and an arginine in the NDN region of CDR3Vβ seem to be important for this interaction. Interestingly, the histidine side chain on position 3 may also interact with the TCR because of the large CDR3Vα chain. In the model, the arginine residue in the CDR3 Vα fragment GARGWEN is particularly close to position 3 of the peptide. Positions 7 and 8 of the peptide with a leucine and a glutamic acid side chain, respectively, appear to be close to the TCRVβ chain NDN region. In fact, a hydrogen bond is feasible between the glutamic acid–NH group in the peptide backbone and the C = O backbone moiety of the first glycine residue in the β chain NDN fragment. As a consequence, the leucine side chain at position 7 in the peptide is oriented toward a hydrophobic region, consisting of the tripeptide Val-Ala-Leu in the NDN fragment. The orientation of the Glu8 side chain in the peptide is arbitrary because there is no immediate TCR counterpart in its vicinity. Overall, the binding between peptide and receptor is compatible with the diagonal mode, as reported for the 2 crystal structures available.1 5 Finally, positions 2, 6, and 9 of the peptide are directed toward the HLA molecule and do not interfere with TCR binding.
Close-up of the peptide-binding site of TCR clone 5W38.
The green zone encompasses the peptide's aspartate residues 4 and 5 interacting with the N and J fragments of the clone. Arginine in the NDN region of the Vβ chain and the first serine residue of the Jα seem of particular relevance. The blue zone highlights a more lipophilic zone consisting of leucine 7 of the peptide and the valine-alanine-leucine tripeptide in the NDN fragment of the Vβ chain. The red area shows the potential interaction between histidine 3 of the peptide and an arginine in the CDR3 Vα fragment GARGWEN. Peptide and protein backbone are in yellow, and the amino acid side chains are in half-bond color, respectively.
Close-up of the peptide-binding site of TCR clone 5W38.
The green zone encompasses the peptide's aspartate residues 4 and 5 interacting with the N and J fragments of the clone. Arginine in the NDN region of the Vβ chain and the first serine residue of the Jα seem of particular relevance. The blue zone highlights a more lipophilic zone consisting of leucine 7 of the peptide and the valine-alanine-leucine tripeptide in the NDN fragment of the Vβ chain. The red area shows the potential interaction between histidine 3 of the peptide and an arginine in the CDR3 Vα fragment GARGWEN. Peptide and protein backbone are in yellow, and the amino acid side chains are in half-bond color, respectively.
Selection of HA-1 TCR antagonists
Peptides were synthesized with every possible amino acid substitution at the proposed TCR contact residues 4, 5, and 8 and at 2 adjacent residues, positions 3 and 7. These peptides were first tested for their agonistic and antagonistic activity against CTL clones 3HA15 and 5W38 in cytotoxicity assays. To test for agonistic activity, the peptides were incubated with an HA-1− EBV-BLCL for 30 minutes before the addition of CTL. To test for antagonistic activity, the peptides were incubated with an HA-1+ EBV-BLCL. This would enable the selection of antagonistic peptides capable of inhibiting the recognition of naturally expressed HA-1 antigen. Substitution of position 5 with every amino acid completely abolished the recognition by the 2 clones because no agonistic or antagonist activity could be detected (Table 3). Position 4 substitution resulted in the loss of agonistic activity for both clones. Approximately half of the position 4 substitutions had antagonizing activity on 3HA15 but not on 5W38. These results, together with the modeling data, strongly suggest that DD at positions 4 and 5 are essential TCR contact sites for 3HA15 and 5W38 and that they may be considered primary TCR contact residues. Substitution of positions 3, 7, and 8 led to peptides with different activities, including antagonistic activity (Table 3). From the 100 different tested peptides we could not detect strong (more than 50% inhibition) antagonizing peptides for 5W38, though we could find strong antagonists for 3HA15.
Agonist and antagonist activity of single amino acid substitutions
| Position . | Substituted amino acids . | 3HA15 . | 5W38 . | ||
|---|---|---|---|---|---|
| Agonist . | Antagonist . | Agonist . | Antagonist . | ||
| 3 (H) | A, Q, L | ++ | − | − | +/− |
| R, K | − | +/− | − | − | |
| N, C, V | ++ | − | +/− | − | |
| D, E, F | ++ | − | − | − | |
| G, S | − | ++ | − | +/− | |
| M, T, I | ++ | − | ++ | − | |
| P, W | − | ++ | − | − | |
| Y | ++ | +/− | − | − | |
| 4 (D) | N, Q, I | − | ++ | − | − |
| H, M, F, W, Y, V | − | +/− | − | − | |
| Rest | − | − | − | − | |
| 5 (D) | All | − | − | − | − |
| 7 (L) | A, F | − | ++ | +/− | − |
| R | − | − | +/− | +/− | |
| N, H | − | +/− | − | − | |
| D | − | − | − | +/− | |
| C | − | ++ | ++ | − | |
| Q | − | +/− | − | +/− | |
| E | +/− | − | +/− | − | |
| G, Y | − | +/− | +/− | − | |
| I, M | +/− | − | ++ | − | |
| K | − | − | ++ | − | |
| P | − | ++ | − | +/− | |
| S, T, V | − | ++ | − | − | |
| W | − | +/− | − | − | |
| 8 (E) | A | +/− | − | − | +/− |
| R, L | − | − | − | − | |
| D | +/− | ++ | ++ | − | |
| C | ++ | − | ++ | − | |
| N, Q, H | − | +/− | − | +/− | |
| G, S | − | ++ | ++ | − | |
| K | ++ | +/− | ++ | − | |
| M | +/− | − | − | − | |
| I, F, P, T, W, Y | − | +/− | − | − | |
| V | − | − | − | +/− | |
| Position . | Substituted amino acids . | 3HA15 . | 5W38 . | ||
|---|---|---|---|---|---|
| Agonist . | Antagonist . | Agonist . | Antagonist . | ||
| 3 (H) | A, Q, L | ++ | − | − | +/− |
| R, K | − | +/− | − | − | |
| N, C, V | ++ | − | +/− | − | |
| D, E, F | ++ | − | − | − | |
| G, S | − | ++ | − | +/− | |
| M, T, I | ++ | − | ++ | − | |
| P, W | − | ++ | − | − | |
| Y | ++ | +/− | − | − | |
| 4 (D) | N, Q, I | − | ++ | − | − |
| H, M, F, W, Y, V | − | +/− | − | − | |
| Rest | − | − | − | − | |
| 5 (D) | All | − | − | − | − |
| 7 (L) | A, F | − | ++ | +/− | − |
| R | − | − | +/− | +/− | |
| N, H | − | +/− | − | − | |
| D | − | − | − | +/− | |
| C | − | ++ | ++ | − | |
| Q | − | +/− | − | +/− | |
| E | +/− | − | +/− | − | |
| G, Y | − | +/− | +/− | − | |
| I, M | +/− | − | ++ | − | |
| K | − | − | ++ | − | |
| P | − | ++ | − | +/− | |
| S, T, V | − | ++ | − | − | |
| W | − | +/− | − | − | |
| 8 (E) | A | +/− | − | − | +/− |
| R, L | − | − | − | − | |
| D | +/− | ++ | ++ | − | |
| C | ++ | − | ++ | − | |
| N, Q, H | − | +/− | − | +/− | |
| G, S | − | ++ | ++ | − | |
| K | ++ | +/− | ++ | − | |
| M | +/− | − | − | − | |
| I, F, P, T, W, Y | − | +/− | − | − | |
| V | − | − | − | +/− | |
Values represent lysis (agonist) and inhibition of lysis (antagonist) activity of single amino acid substitutions at positions 3, 4, 5, 7, and 8 of the HA-1 peptide with CTL clones 3HA15 and 5W38.
− Indicates less than 25% lysis/inhibition of lysis; ±, 25% to 50% lysis/inhibition of lysis; and ++, more than 50% lysis/inhibition of lysis.
Analysis of candidate antagonists
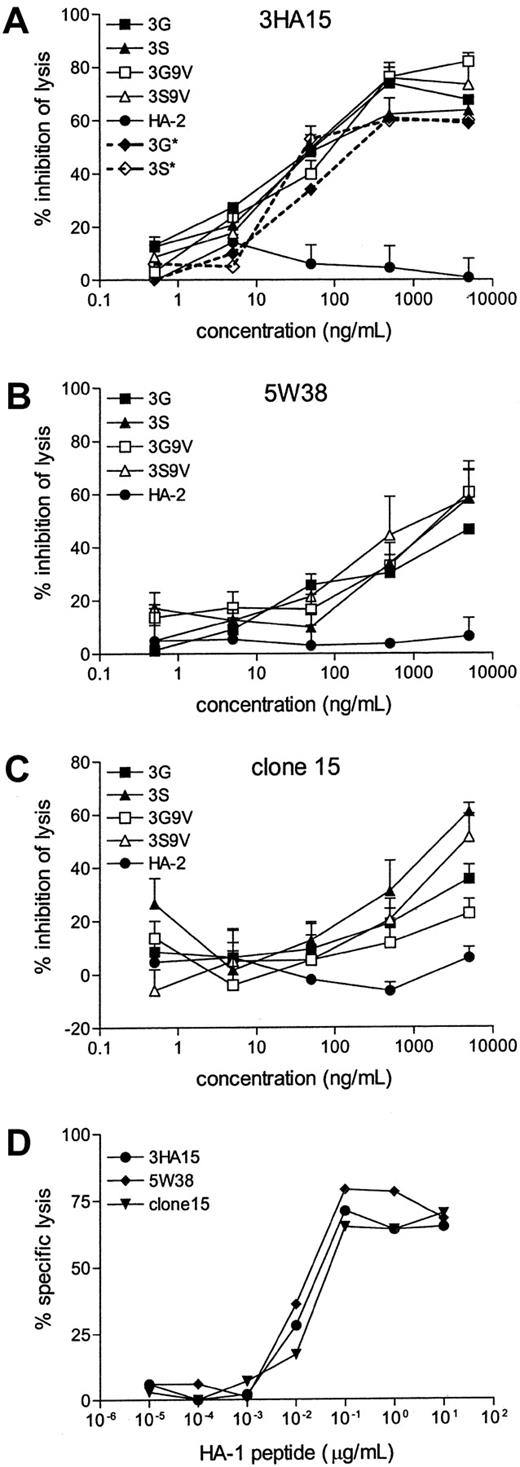
Among the 7 peptides that showed moderate to strong antagonistic but no agonistic activity for the 3HA15 and the 5W38 clones, 2 peptides that contained substitutions at position 3 (G or S) were selected for further analysis on the basis of reproducibly antagonizing 3HA15 and 5W38. Additional substitution of position 9 (V instead of A) of these 2 peptides resulted in peptides with higher affinity for HLA-A2 (IC50 of 18 nM for 3G9V and 3S9V compared to IC50 of 82 and 66 nM for 3G and 3S, respectively). All 4 peptides (3G, 3S, 3G9V, 3S9V) were efficient antagonists of clone 3HA15 and completely abolished the lysis of an HA-1–presenting EBV-BLCL (Figure 3A), whereas CTL clone 5W38 could be inhibited 50% by the 4 peptide antagonists (Figure 3B). The latter 4 antagonist peptides were subsequently used to antagonize a third HA-1–specific CTL clone, clone 15. Like clone 3HA15, clone 15 is derived from the polyclonal CTL line (A2Bx), and it expresses the same TCR Vα (AV3S1) and Vβ (BV6S4A2) chains but different J and CDR3 regions. In spite of the highly homologous TCRs, the 2 clones reacted differently to the 4 peptides. Whereas all 4 peptides strongly antagonized 3HA15, the inhibition of clone 15 was partial, with best inhibition levels of 50% to 60% by 3S and 3S9V (Figure 3C).
Antagonistic activity of selected peptides on HA-1–specific CTL clones.
(A-C) Peptides were incubated with 51Cr labeled, HA-1 expressing EBV-BLCL at 37°C with the indicated peptide concentrations 30 minutes before the addition of CTL clones 3HA15 (A) 5W38 (B) and clone 15 (C). Percentage inhibition of lysis by antagonist peptides was calculated as described in “Materials and methods.” Average percentage inhibition of at least 3 independent experiments is shown with the SEM. Mean percentages lysis of HA-1+ EBV-BLCL in the absence of antagonistic peptides were 66% for 3HA15, 59% for 5W38, and 45% for clone 15. For the CTL clone 3HA15, the antagonistic effects of 3G and 3S were also determined in response to HA-1− target cells pulsed with 0.1 μg/mL HA-1 peptide (3G*, 3S*). (D) Response of HA-1–specific CTL clones to serial dilutions of HA-1 peptide loaded on HA-1− EBV-BLCLs.
Antagonistic activity of selected peptides on HA-1–specific CTL clones.
(A-C) Peptides were incubated with 51Cr labeled, HA-1 expressing EBV-BLCL at 37°C with the indicated peptide concentrations 30 minutes before the addition of CTL clones 3HA15 (A) 5W38 (B) and clone 15 (C). Percentage inhibition of lysis by antagonist peptides was calculated as described in “Materials and methods.” Average percentage inhibition of at least 3 independent experiments is shown with the SEM. Mean percentages lysis of HA-1+ EBV-BLCL in the absence of antagonistic peptides were 66% for 3HA15, 59% for 5W38, and 45% for clone 15. For the CTL clone 3HA15, the antagonistic effects of 3G and 3S were also determined in response to HA-1− target cells pulsed with 0.1 μg/mL HA-1 peptide (3G*, 3S*). (D) Response of HA-1–specific CTL clones to serial dilutions of HA-1 peptide loaded on HA-1− EBV-BLCLs.
The differential effect of antagonistic peptides on the 3 CTL clones could be a reflection of their different affinity to the HA-1 peptide. To verify this possibility, the CTL clones were assayed against target cells in HA-1 peptide titration assays. All 3 CTL clones reacted similarly to the HLA-A2–HA-1 peptide ligand (Figure 3D).
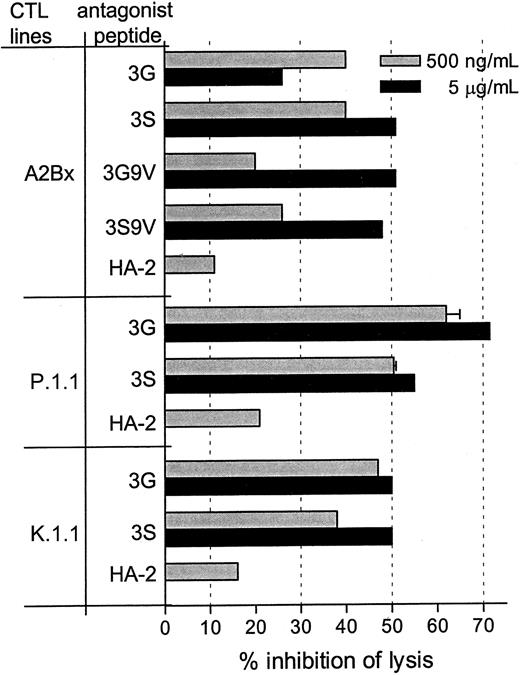
Antagonist peptides were subsequently used to antagonize polyclonal HA-1–specific CTL lines derived from 3 different patients. The CTL line A2Bx, the original CTL line of the CTL clone 3HA15 and clone 15, was inhibited 50% with the peptides 3S, 3G9V, and 3S9V and was inhibited 40% with peptide 3G (Figure4). CTL lines P.1.1 and K.1.1 were also effectively inhibited by the antagonistic peptides 3G and 3S, with maximal inhibition levels of 50% to 70% with 5 μg/mL peptides. These results show that at the clonal and at the polyclonal level, CTL responses can be inhibited by antagonist peptides.
Antagonistic activity of selected peptides on polyclonal HA-1–specific CTL lines.
Peptides were incubated with 51Cr labeled, HA-1– expressing EBV-BLCL at 37°C with the indicated peptide concentrations 30 minutes before the addition of the indicated CTL in standard 4-hour51Cr release assays. Percentage inhibition of lysis by antagonist peptides was calculated as described in “Materials and methods.” Data are representative of at least 2 independent experiments in which the mean percentages lysis of HA-1+target cells without antagonistic peptides were 35% for A2Bx, 54% for P.1.1, and 58% for K.1.1.
Antagonistic activity of selected peptides on polyclonal HA-1–specific CTL lines.
Peptides were incubated with 51Cr labeled, HA-1– expressing EBV-BLCL at 37°C with the indicated peptide concentrations 30 minutes before the addition of the indicated CTL in standard 4-hour51Cr release assays. Percentage inhibition of lysis by antagonist peptides was calculated as described in “Materials and methods.” Data are representative of at least 2 independent experiments in which the mean percentages lysis of HA-1+target cells without antagonistic peptides were 35% for A2Bx, 54% for P.1.1, and 58% for K.1.1.
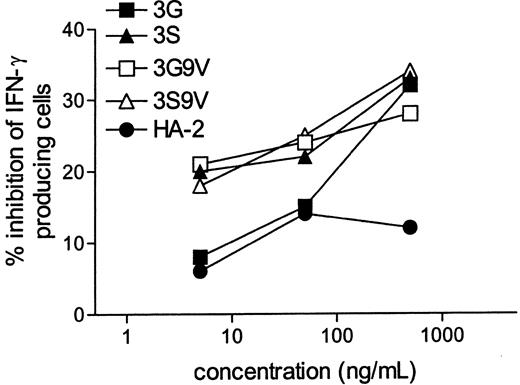
Finally, we performed ELISpot (Mabtech) assays to determine whether the antagonist peptides can inhibit the HA-1–specific CTL present in the PBMCs of a patient who acquired GvHD after HLA-matched–HA-1–mismatched SCT. Because the patient and donor have multiple mHag disparities besides HA-1, the patient's post-SCT PBMCs were stimulated with stem cell donor EBV-BLCLs pulsed with 0.1 μg/mL HA-1 peptide. Subsequent addition of antagonist peptides specifically reduced the number of IFN-γ–producing HA-1–specific T cells. Inhibition of 30% was achieved at 5 μg/mL of antagonist peptide concentrations (Figure 5). These results revealed that the antagonist peptides were also capable of significantly inhibiting the number of HA-1–specific T cells in the patient's PBMCs during GvHD.
Inhibitory activity of antagonistic HA-1 peptides on the number of IFN-γ–producing HA-1–specific T cells in the PBMCs of a patient with GvHD.
After SCT, PBMCs were stimulated in triplicate for 72 hours with HA-1− donor EBV-BLCLs pulsed with 0.1 μg/mL peptide. Antagonist peptides were added in the assay. The IFN-γ spots were visualized and counted as indicated in “Materials and methods.” Percentage inhibition of IFN-γ producing T cells was calculated as follows: (1 − (mean frequency of IFN-γ–producing cells in the presence of the antagonist peptide)/(mean frequency of IFN-γ–producing cells in the absence of the antagonist peptide) × 100%. The frequency of IFN-γ–producing HA-1–specific T cells in the absence of the antagonist peptide was 96 per 105 PBMCs.
Inhibitory activity of antagonistic HA-1 peptides on the number of IFN-γ–producing HA-1–specific T cells in the PBMCs of a patient with GvHD.
After SCT, PBMCs were stimulated in triplicate for 72 hours with HA-1− donor EBV-BLCLs pulsed with 0.1 μg/mL peptide. Antagonist peptides were added in the assay. The IFN-γ spots were visualized and counted as indicated in “Materials and methods.” Percentage inhibition of IFN-γ producing T cells was calculated as follows: (1 − (mean frequency of IFN-γ–producing cells in the presence of the antagonist peptide)/(mean frequency of IFN-γ–producing cells in the absence of the antagonist peptide) × 100%. The frequency of IFN-γ–producing HA-1–specific T cells in the absence of the antagonist peptide was 96 per 105 PBMCs.
Discussion
Four crystal structures of MHC–peptide–TCR complexes indicate that the diagonal mode of interaction between TCR and HLA molecules is a general feature of different TCRs.1-5 mHag HA-1–specific CTL clones have been characterized by the presence of the BV6S4A2 chain and conserved amino acids in the VβCDR3 region.24 This suggests that different HA-1–specific TCRs have similar interactions with the HLA-A2–HA-1 complex. Therefore, it may be feasible to identify general TCR antagonists capable of inhibiting a total HA-1–specific TCR response. Our results show that general antagonist peptides do exist that can efficiently inhibit clonal and polyclonal HA-1–specific CTL responses and can reduce IFN-γ–producing HA-1–specific T cells isolated from a patient during ongoing acute GvHD.
Interaction of HA-1 with 3HA15 and 5W38 TCRs
Three different HA-1–specific CTL clones, 3HA15, 5W38, and clone 15, were evaluated for their TCR contact residues in the HA-1 peptide. All clones used the BV6S4A2 segment, and 2 (3HA15 and clone15) used the same AV2S1 segment. Alanine substitution analysis revealed that positions 4, 5, and 8 were important for the TCR interaction with the 3 clones. Position 7 was essential for the 2 clones with the identical VαVβ chains. Recently, we identified a fourth HA-1–specific T-cell clone expressing BV6S4A2, but with a different TCR, it also was shown to have positions 4, 5, 7, and 8 as TCR contact sites (data not shown). Computer modeling of the HLA-A2–HA-1 complex with the TCR of clone 5W38 indicated that the side chains of the aspartic acids at positions 4 and 5 indeed pointed out from the groove and fit in the pocket formed by the 5W38 CDR3Vα and Vβ chains. Because other substitutions at these positions are as detrimental for T-cell recognition as the alanine substitution, it can be concluded that positions 4 and 5 are the most essential TCR contact sites in the HA-1 peptide. Positions 7 and 8 are more distant, but there seems to be contact with the 2 glycines and with Val-Ala-Leu in the CDR3Vβ region of 5W38. In the model, the histidine at position 3 could interact with the arginine of the CDR3Vα region ARWGE of 5W38. The absence of activity of most substitutions at this position for 5W38 supports this hypothesis.
We did not observe any evidence for interactions with the CDR1 of the 5W38 Vα and Vβ chains with HLA-A2–HA-1 in the model. This may be because of the relatively large CDR3 Vα and Vβ regions of the 5W38 TCR. Crystallized A6 TCR has a larger CDR3 Vβ region than crystallized 2C receptor,34 which probably causes the TCR to shift and prevents close contact between the Vβ domain and the HLA-A2 molecule. The 5W38 TCR has a similar CDR3 Vβ length, and the CDR3Vα is 5 residues longer than that of A6. This may explain the absence of interaction of CDR1 regions in 5W38 with the HLA-A2–HA-1 complex. In contrast, the CDR3Vα and Vβ regions of 3HA15 are, respectively, 2 and 3 amino acids shorter than 5W38. This may facilitate more HLA-A2 contact with 3HA15 TCR and may explain the loss of agonistic activity seen when position 7 is substituted. In addition, the shorter CDR3Vα in 3HA15 has probably fewer interactions with the histidine at position 3 in the HA-1 peptide than the CDR3Vα chain of 5W38. More substitutions are allowed at position 3 for recognition by 3HA15 than by 5W38.
Determination of general HA-1 antagonists
One hundred peptides were assayed for their antagonistic activity on HA-1–specific 3HA15 and 5W38 CTL clones. To determine the level of antagonizing activity, peptides were loaded on EBV-BLCL that endogenously expressed mHag HA-1. In our opinion, this is a more physiological test system than a prepulse assay.9 In a prepulse assay a target cell is loaded with suboptimal amounts of antigen. Nonbound antigen is washed away, and the antagonistic peptide is added. The antigen gradually dissociates from the MHC molecules, and its abundance decreases in time. In contrast, HA-1 expressing EBV-BLCL continuously expresses a similar amount of HA-1 antigen sufficient to efficiently activate T cells. Although prepulsed targets are loaded with suboptimal amounts of antigen, generally resulting in low lysis, the HA-1 expressing EBV-BLCL are lysed to 60%. Only peptides that can antagonize this lysis may be effective for in vivo protocols.
From the 100 peptides evaluated, only 2 peptides with a substitution on position 3 (3G and 3S) showed clear antagonizing activity toward the 3HA15 and the 5W38 clones in cytotoxicity assays. The potency of these antagonist peptides were additionally verified in the prepulse assay mentioned above. Figure 3A shows that when HA-1 peptide-pulsed target cells were used, efficient inhibition of target cell lysis by clone 3HA15 could be achieved by the addition of 5- to 50-fold 3G and 3S antagonist peptides.
Although several good antagonists were found for 3HA15, only a few moderate antagonists could be identified for 5W38. Possibly the longer CDR3 chains of 5W38 have fewer contact sites with HLA-A2–HA-1, allowing less variation. Similar to the inhibition patterns of clone 5W38, the third HA-1–specific clone, clone 15, was also less significantly inhibited than clone 3HA15, but the Vα and Vβ of both clones were identical. This suggests that the Vα and Vβ CDR3 regions contain relatively important areas that play roles in the interaction of TCRs with antagonist peptides. Alternatively, the affinity of the CTL clones to the HLA-A2–HA-1 ligand may be different. However, our HA-1 peptide titration studies did not reveal a difference between the CTL clones. This suggests that the peptide affinity differences between CTL clones may not explain the variation in the antagonistic effects among the CTL clones. Naturally, a peptide titration may not reflect the real TCR affinity differences between the CTL clones; direct TCR affinity measurements are required to answer this question.
Of interest is that the antagonists significantly inhibit the response of polyclonal HA-1–specific CTL lines from different patients by 50% or more. Moreover, the antagonistic peptides also reduced 30% of the IFN-γ–producing HA-1–specific CTL in the PBMCs of a patient with GvHD after HLA-matched, HA-1–mismatched SCT. These results underscore the apparent dominant HA-1 response in naturally multiple mHag-mismatched SCT. Because these assays were performed for technical reasons with peptide-pulsed EBV-BLCL, we expect higher levels of inhibition in response to the natural HA-1 ligand. Thus, the generation and identification of general antagonists is feasible for restricted TCR responses with restricted sets of Vα or Vβ domains and, perhaps more important, with conserved amino acids in the CDR3 regions.
Clinical relevance
The possible clinical relevance of these in vitro immunomodulation studies lies in the possibility of reducing the HA-1–induced GvHD in HA-1 disparate SCT donor–patient combinations. Three types of observations support the relevance of the latter proposition. First, HA-1 disparity is associated with the development of acute GvHD.28,29 Second, HA-1–specific CTLs have been isolated from different cohorts of patients with GvHD.26,27,30,35Third, the apparent exceptional restricted use of the TCR repertoire for HA-1 recognition makes the general TCR antagonists applicable in the HA-1–mismatched combinations.24 Especially early after SCT, immunomodulation of the HA-1 response may be important. This supposition is based on recent work by Schlomchik et al,36who described an MHC-identical, mHag-mismatched murine model of allo-SCT with bone marrow chimeric mice without class I on their antigen-presenting cells (APCs) but with class I on GvHD target tissues. This model was used to investigate the mechanism of GvHD activation. Several important conclusions have been drawn from these studies. They are that depleting host APCs before conditioning abrogates GvHD, replacing host with donor APCs abrogates T-cell activation, and cross-presenting host antigens by donor APCs does not cause GvHD; thus the target antigens for CD8 T cells in GvHD are restricted to proteins expressed by host APCs. In concordance with the results of the latter model system, we ascribe the association between HA-1 mismatch and the development of GvHD to the significant expression of HA-1 on the patients' professional APCs. We argue that through adequate presentation of HA-1 on the remaining patient's dendritic cells early after SCT, strong donor antihost allo-immune T cells are induced. This may give rise to the production of cytokines, subsequently leading to the activation of bystander allo-immune T cells such as CTL against mHag H-Y, a ubiquitously expressed mHag. It is expected that most recipient hematopoietic cells will be replaced by donor cells by day 50 to 60 after SCT. Thus, early modulation of the HA-1 immune response would benefit the patient.
We thank Dr G. Vriend (EMBL, Heidelberg, Germany) for the sequence analogy search and Dr A. Geluk for productive discussions.
Supported by grants from the Dutch Organization for Scientific Research (NWO 901-09-201) (J.d.H.) and the J. A. Cohen Institute for Radiopathology and Radiation Protection (E.A.J.M.G.).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
E. A. J. M. Goulmy, Dept of Immunohematology and Blood Transfusion, Leiden University Medical Center, Albinusdreef 2, 2333 ZA Leiden, The Netherlands;e.a.j.m.goulmy@lumc.nl.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal