CD4+, CD56+ DC2 malignancies constitute a novel disease entity, which has recently been shown to arise from a transformed lymphoid-related plasmacytoid dendritic cell (DC2). Diagnosis is primarily based on a particular immunophenotype with tumor cells expressing CD4 and CD56 antigens in the absence of common lymphoid or myeloid lineage markers. Little is currently known about the cytogenetic features of this disease entity. In this setting, the Groupe Français de Cytogénétique Hématologique (GFCH) initiated a cytogenetic study of 18 adults and 3 children with CD4+, CD56+ DC2 acute leukemia using conventional and fluorescence in situ hybridization/24-color karyotyping. Clonal, mostly complex chromosome aberrations were found in 14 patients (66%). Six major recurrent chromosomal targets were defined. These were 5q, 12p, 13q, 6q, 15q, and 9, which were involved in 72% (5q), 64% (12p and 13q), 50% (6q), 43% (15q), and 28% (monosomy 9) of cases, respectively. Cytogenetic features can be summarized as follows: (1) gross genomic imbalances (mostly losses) predominate, (2) no single anomaly can be considered as specific, whereas their combination/accumulation is, and (3) both lymphoid and myeloid lineage–associated rearrangements are observed in unusual combinations in the same cell. This is suggestive of complex multistep tumorigenic mechanisms and is supportive of the hypothesis that CD4+, CD56+ DC2 acute leukemia may arise from an undifferentiated nonmyeloid nonlymphoid progenitor cell. In conclusion, the present study documents for the first time the existence of a characteristic cytogenetic profile for this novel disease entity.
Introduction
The CD4+, CD56+malignancies, initially described by Brody et al1 and further characterized by others,2-4 constitute a novel disease entity based on clinical, morphologic-cytologic, and immunologic studies.5 This disease has recently been shown to arise from a transformed lymphoid-related plasmacytoid dendritic cell type 2 (DC2).6 Primarily defined through their particular immunophenotypic profile (CD4+, CD56+ cells without common lymphoid- or myeloid-lineage markers), patients characteristically present with primary cutaneous nodules (83%), lymph node and spleen enlargement (61%), and cytopenia and bone marrow infiltration (87%), as extensively described by Feuillard et al.5 Despite an initial good response to chemotherapy (78% complete remission), patients will eventually relapse within a few months (median, 9 months) and overall survival at 24 months is low (25%). From a morphologic viewpoint, bone marrow is usually massively infiltrated by malignant cells with a blastic aspect, a large cytoplasm with (micro)vacuoles often adopting a pearl necklace aspect beneath the nuclear membrane and pseudopodialike cytoplasmic expansions. However, the cytogenetic features of this novel disease entity are currently poorly characterized.
In this setting, we have analyzed tumor cells from 21 patients with CD4+, CD56+ DC2 acute leukemia using conventional and fluorescence in situ hybridization (FISH)/24-color karyotyping. We here present cytogenetic evidence that the CD4+, CD56+ DC2 acute leukemia represents a distinct clinicopathologic entity, with respect to the World Health Organization (WHO) classification.7
Patients, materials, and methods
Selection of patients
In the setting of a multicenter study conducted by the French GEIL and GFHC groups (Groupe d'Etude Immunologique des Leucémies, Groupe Français d'Hématologie Cellulaire) on CD4+, CD56+ hematologic malignancies, the GFCH group (Groupe Français de Cytogénétique Hématologique) initiated a cytogenetic study that included and reviewed all patients selected for the GEIL/GFHC study for whom a cytogenetic analysis was available. Unique patient numbers (UPNs) were assigned to each case and are consistent with those of Feuillard et al.5 Briefly, in the latter study, patient selection was based on the expression of CD4 and CD56 in the absence of other lineage conventional markers (CD13, CD33, MPO, CD3, CD5, CD19, CD20). Detailed clinical, cytologic, and immunophenotypic data are provided in Feuillard et al.5
Of the 23 patients enrolled in the GEIL/GFHC study, 3 were excluded from the chromosomal analysis. No karyotypic analysis was performed in patients 5 and 23. Forty normal metaphases were observed in the bone marrow of patient 22 at the time of diagnosis of so called non-Hodgkin lymphoma (NHL), natural killer (NK) subtype. However, at that time the patient's bone marrow was found to be normal (0% blasts) and as a consequence, this normal karyotype was considered uninformative. Another patient (no. 24), excluded from the GEIL/GFHC study because of lack of bone marrow smears for cytologic review, was included in the chromosomal study. The immunologic profile of this patient had been extensively described by Chaperot et al (patient GEN).6Data on patient 3 have been previously published.4
In total, this study includes 21 patients (18 adults, 3 children), whose ages ranged from 5 to 86 years (median, 61.8 years); 5 are female and 16 male.
Cytogenetic analysis
Chromosome analysis was performed on bone marrow in 17 cases, blood in 3 cases, and a lymph node biopsy in 1 case. Seventeen samples were studied at the time of diagnosis and 4 during the course of medullar dissemination.
Cell suspensions were cultured for 24 to 72 hours without stimulation. Samples were collected according to the appropriate institutional IRB-approved protocols (CCPPRB-CHU de Grenoble). Metaphases were prepared according to standard procedures and analyzed with R banding. At least 20 metaphases were required to assess a normal karyotype. Karyotype descriptions conform to the International System for Human Cytogenetic nomenclature.8 All karyotypes were reviewed by members of the GFCH.
FISH analysis
Complementary metaphase or interphase FISH studies were performed in 13 cases using standard procedures. Probes used included whole chromosome paintings (wcps) as well as centromeric, telomeric, or locus-specific probes (5q31/EGR1,12p13/ETV6 or 13q14/D13S319 or D13S25). The 24-color FISH karyotyping (M-FISH or SKY)9,10 was carried out on 9 cases using Vysis probes (7 cases; Vysis, Voisins le Bretonneux, France), Metasystems probes (1 case; Metasystems, Altlussheim, Germany), or noncommercial probes (1 case).11 Protocols were as recommended by manufacturers or as previously described.11-13
Results
CD4+, CD56+DC2 leukemia shows clonal karyotypic changes involving 6 major chromosome targets
Molecular cytogenetic data for the 21 patients are detailed in Table 1. Twenty of the patients had successful karyotype analyses. Clonal chromosome aberrations were found in 14 patients (66%); 12 of these 14 presented a fraction of residual normal metaphases (mean percentage of cells with normal karyotype, 25%).
Cytogenetic/FISH investigations in the 21 patients with CD4+, CD56+ DC2 acute leukemia
| UPN . | Sex/age . | Source of cells/culture (h) . | % blasts BM . | FISH/probes . | Karyotype according to R-bands and as modified after 24-color karyotyping . |
|---|---|---|---|---|---|
| 1* | M/76 | BM/24 | 63 | — | 46,XY[40] |
| 2* | M/8 | BM/24 | 70 | M-FISH | 44,X,-Y,del(1)(q21),der(3),del(5)(q3?3q3?5),add(6)(q2?1),-13[7]]/46,XY[8] |
| Ish wcp M-FISH 44,X,-Y,der(1)t(Y;1)(q12;q?21),der(3)t(1;3)(p11;q?),del(5)(q3?3q3?5), der(6)(6pter->6q22::1q?::8q?),der(12)t(1;12)(?;p11),r(13)[3] | |||||
| 3 | F/86 | BM/24 | 87 | M-FISH | 46,XX,del(5)(q13q33)[2]/42,idem,add(1)(q41),add(2)(q37),-9,-12,-13,-15,-20,-21,add (21)(q22),+2mar[20]/46XX[3] |
| ish wcp M-FISH der(1)t(1;8)(q4?3;?),dup(2)(?q35q37),-9,der(12)(?),-13,-15,der(20;21) (?p11;?q22),der(21)(21pter->21qter::12::21q11->21qter),-21[3] | |||||
| 4 | F/81 | BM/24 & 48 | 44 | — | 84-87,XX,del(X)(q24qter)x2[11],add(2)(q3?)[11],der(2)(?)[11],+del(4)(q23qter)[9], +del(5)(q14qter)[11],+del(5)(q14q23)[11],add(6)(q?),-9[11],-11[6],del(12)(p12){3],-13[11],-13[9],-14[11],-15x2[11],-17[11],-17[5],-18[3],+r(?)[11],+mar1[11},+mar2[4], +mar3[3][cp11]/46,XX[4] |
| 6 | M/68 | BM/24 | 40 | M-FISH | 46,XY[21]. ish wcp M-FISH[30] |
| 7 | M/75 | BM/24 | 74 | M-FISH | 45,XY,-5,add(7)(p22)[3]/46,XY[10]. |
| Ish wcp M-FISH del(5)(q3?1q3?5),der(7)(12qter->12q1?1::7q10->7qter),-12[3] | |||||
| 8 | M/79 | LN/17 | 74 | M-FISH | 45,XY,-5,add(6)(q2?2),del(11)(q14q23),-13,-15,+2mar[4]/46,XY[2]. |
| ish wcp M FISH 45,XY,+5,der(5)t(5;11)(p10;?)x2,add(6)(q2?2),del(11)(q14q23),-13, -15[4] | |||||
| 9 | F/6 | BM/24 & 48 | 90 | — | 44,XX,del(3)(p21),-5,-12,-13,add(17)(p11),-18,-19,+3mar[27]/46,XX[2] |
| 10 | M/55 | BM/24 | 80 | 13q14 | 46,XY[25].Ish 13q14(D13S25)x2)[100%] |
| 11* | M/79 | BM/24 | 0 | Wcp5/6 | 48,XY,+6,add(6)(q10)x2,add(9)(p24),der(12)t(1;12)(q22;p13),+21[2]/46,XY[13]. |
| ish 5(wcp5x2),add(6)(wcp6+) | |||||
| 12 | M/14 | BM/17 | 96 | SKY wcp3, 19 Cen 7/8, Tel3p,3q,6p,6q | 43,XY,add(3p2?3),add(6)(q?21),-7,-9,add(12)(p11),-13,-19,+mar[7]/86,idemx2[3].ish SKY 43,XY,t(3;6)(p25;q2?3),der(7)t(7;19)(7qter->7p21::19q10->19qter),-9, der(12)t(5;12)(?;p11),-13,-19[10] |
| 13* | M/74 | PB/24 | 0 | M-FISH | 46,XY,del(2)(p2?1),add(8)(q2?4),del(13)(q1?3q2?1)[31]/47,idem,+mar[10]. ish wcp M-FISH +Y(wcpY+)[10] |
| 14 | F/79 | BM/24 | 74 | TEL/AML1 | 46,XX,del(12)(p12p13)[9]/46,XX[2]. |
| 5q31, 13q14 | ish/nuc ish12p13(TEL/ETV6x1),21q22(AML1x2)[78%], 5q31(248FSx2), 13q14(D13S319x2)[100%] | ||||
| 15 | M/67 | BM/24 | 60 | TEL/AML1 | 46,XY[20]. nuc ish 12p13(TEL/ETV6x2),21q22(AML1x2) |
| 16 | F/75 | BM/48 | 90 | — | 45,XX,del(5)(q13q34),?inv(11)(p11q21),der(15)(15qter->15q10::?::18q10->18qter), -18[15]/44,idem,-22,dmin[2]/88,idemx2,del(6)(q16)[2]/46,XX[1] |
| 17 | M/60 | BM/24 | 100 | — | Failure |
| 18 | M/82 | PB/24 | 92 | M-FISH | 42,X,-Y,-2,del(5)(p13),del(5)(q13q33),del(6)(q24),-11,-13,add(13)(p11),-14,add(14) (p11),-15,add(18)(q21),add(19)(q13),-21,-22,+4mar[18]/46,XY[3] |
| ish wcp M-FISH 42,X,-Y,der(2)(6qter-6q1?1::2p10->2p?::5?),del(5)(p13),der(5) (5pter->5q21::13q?),der(6)(6pter->6q1?1::2q?14->2qter),t(6;18)(q2?2;q22), der(11),-13,der(13)t(13;21)(q10;q10),-14,der(14)t(Y;14)(q11;p11),r(15),der(19) (19pter->19q13::3p21->3pter),-21,i(22)(q10)[3] | |||||
| 19 | M/56 | BM/72 | 52 | — | 46,XY[21] |
| 20 | M/77 | BM/96 | 21 | — | 46,XY[40] |
| 21 | M/70 | BM/24 | 48 | — | 44,XY,-9,-13[8]/88,idemx2[18]/46,XY[5] |
| 24 | M/74 | PB/24 | 95 | M-FISH wcp 6, 8, 15, 16 | 49,XY,+add(6)(q21),-8,+2mar,+r[6]/49,idem,t(15;16)(?q21;?q21)[6]/49,idem,t(3;5) (?q21;?q31)[5] |
| ish wcp M-FISH 49,XY,+6,t(6;8)(p21;q24),+r(12),+20/49,idem,inv(15)(q1?4q2?3), t(16;16)(q?;q?)/49,idem,t(3;5)(q?21;q?31)[13] |
| UPN . | Sex/age . | Source of cells/culture (h) . | % blasts BM . | FISH/probes . | Karyotype according to R-bands and as modified after 24-color karyotyping . |
|---|---|---|---|---|---|
| 1* | M/76 | BM/24 | 63 | — | 46,XY[40] |
| 2* | M/8 | BM/24 | 70 | M-FISH | 44,X,-Y,del(1)(q21),der(3),del(5)(q3?3q3?5),add(6)(q2?1),-13[7]]/46,XY[8] |
| Ish wcp M-FISH 44,X,-Y,der(1)t(Y;1)(q12;q?21),der(3)t(1;3)(p11;q?),del(5)(q3?3q3?5), der(6)(6pter->6q22::1q?::8q?),der(12)t(1;12)(?;p11),r(13)[3] | |||||
| 3 | F/86 | BM/24 | 87 | M-FISH | 46,XX,del(5)(q13q33)[2]/42,idem,add(1)(q41),add(2)(q37),-9,-12,-13,-15,-20,-21,add (21)(q22),+2mar[20]/46XX[3] |
| ish wcp M-FISH der(1)t(1;8)(q4?3;?),dup(2)(?q35q37),-9,der(12)(?),-13,-15,der(20;21) (?p11;?q22),der(21)(21pter->21qter::12::21q11->21qter),-21[3] | |||||
| 4 | F/81 | BM/24 & 48 | 44 | — | 84-87,XX,del(X)(q24qter)x2[11],add(2)(q3?)[11],der(2)(?)[11],+del(4)(q23qter)[9], +del(5)(q14qter)[11],+del(5)(q14q23)[11],add(6)(q?),-9[11],-11[6],del(12)(p12){3],-13[11],-13[9],-14[11],-15x2[11],-17[11],-17[5],-18[3],+r(?)[11],+mar1[11},+mar2[4], +mar3[3][cp11]/46,XX[4] |
| 6 | M/68 | BM/24 | 40 | M-FISH | 46,XY[21]. ish wcp M-FISH[30] |
| 7 | M/75 | BM/24 | 74 | M-FISH | 45,XY,-5,add(7)(p22)[3]/46,XY[10]. |
| Ish wcp M-FISH del(5)(q3?1q3?5),der(7)(12qter->12q1?1::7q10->7qter),-12[3] | |||||
| 8 | M/79 | LN/17 | 74 | M-FISH | 45,XY,-5,add(6)(q2?2),del(11)(q14q23),-13,-15,+2mar[4]/46,XY[2]. |
| ish wcp M FISH 45,XY,+5,der(5)t(5;11)(p10;?)x2,add(6)(q2?2),del(11)(q14q23),-13, -15[4] | |||||
| 9 | F/6 | BM/24 & 48 | 90 | — | 44,XX,del(3)(p21),-5,-12,-13,add(17)(p11),-18,-19,+3mar[27]/46,XX[2] |
| 10 | M/55 | BM/24 | 80 | 13q14 | 46,XY[25].Ish 13q14(D13S25)x2)[100%] |
| 11* | M/79 | BM/24 | 0 | Wcp5/6 | 48,XY,+6,add(6)(q10)x2,add(9)(p24),der(12)t(1;12)(q22;p13),+21[2]/46,XY[13]. |
| ish 5(wcp5x2),add(6)(wcp6+) | |||||
| 12 | M/14 | BM/17 | 96 | SKY wcp3, 19 Cen 7/8, Tel3p,3q,6p,6q | 43,XY,add(3p2?3),add(6)(q?21),-7,-9,add(12)(p11),-13,-19,+mar[7]/86,idemx2[3].ish SKY 43,XY,t(3;6)(p25;q2?3),der(7)t(7;19)(7qter->7p21::19q10->19qter),-9, der(12)t(5;12)(?;p11),-13,-19[10] |
| 13* | M/74 | PB/24 | 0 | M-FISH | 46,XY,del(2)(p2?1),add(8)(q2?4),del(13)(q1?3q2?1)[31]/47,idem,+mar[10]. ish wcp M-FISH +Y(wcpY+)[10] |
| 14 | F/79 | BM/24 | 74 | TEL/AML1 | 46,XX,del(12)(p12p13)[9]/46,XX[2]. |
| 5q31, 13q14 | ish/nuc ish12p13(TEL/ETV6x1),21q22(AML1x2)[78%], 5q31(248FSx2), 13q14(D13S319x2)[100%] | ||||
| 15 | M/67 | BM/24 | 60 | TEL/AML1 | 46,XY[20]. nuc ish 12p13(TEL/ETV6x2),21q22(AML1x2) |
| 16 | F/75 | BM/48 | 90 | — | 45,XX,del(5)(q13q34),?inv(11)(p11q21),der(15)(15qter->15q10::?::18q10->18qter), -18[15]/44,idem,-22,dmin[2]/88,idemx2,del(6)(q16)[2]/46,XX[1] |
| 17 | M/60 | BM/24 | 100 | — | Failure |
| 18 | M/82 | PB/24 | 92 | M-FISH | 42,X,-Y,-2,del(5)(p13),del(5)(q13q33),del(6)(q24),-11,-13,add(13)(p11),-14,add(14) (p11),-15,add(18)(q21),add(19)(q13),-21,-22,+4mar[18]/46,XY[3] |
| ish wcp M-FISH 42,X,-Y,der(2)(6qter-6q1?1::2p10->2p?::5?),del(5)(p13),der(5) (5pter->5q21::13q?),der(6)(6pter->6q1?1::2q?14->2qter),t(6;18)(q2?2;q22), der(11),-13,der(13)t(13;21)(q10;q10),-14,der(14)t(Y;14)(q11;p11),r(15),der(19) (19pter->19q13::3p21->3pter),-21,i(22)(q10)[3] | |||||
| 19 | M/56 | BM/72 | 52 | — | 46,XY[21] |
| 20 | M/77 | BM/96 | 21 | — | 46,XY[40] |
| 21 | M/70 | BM/24 | 48 | — | 44,XY,-9,-13[8]/88,idemx2[18]/46,XY[5] |
| 24 | M/74 | PB/24 | 95 | M-FISH wcp 6, 8, 15, 16 | 49,XY,+add(6)(q21),-8,+2mar,+r[6]/49,idem,t(15;16)(?q21;?q21)[6]/49,idem,t(3;5) (?q21;?q31)[5] |
| ish wcp M-FISH 49,XY,+6,t(6;8)(p21;q24),+r(12),+20/49,idem,inv(15)(q1?4q2?3), t(16;16)(q?;q?)/49,idem,t(3;5)(q?21;q?31)[13] |
BM indicates bone marrow; PB, peripheral blood; LN, lymph node; Cen, centromeric probe; Tel, telomeric probe.
Cytogenetic data obtained in the course of the secondary medullary dissemination.
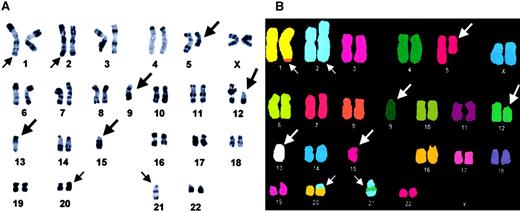
Nine of the 14 patients with clonal aberrations (64%) exhibited a hypodiploid-hypotetraploid karyotype (42-45 chromosomes), 3 were pseudodiploid (46 chromosomes), and 2 hyperdiploid (48-49 chromosomes). With the exception of 3 patients (2 with a single anomaly and 1 with 2 aberrations), all 11 other patients were characterized by complex chromosome clones (3 or more anomalies/clone; Figure1A). The number of alterations in individual clones varied between 1 and 16 (mean, 6.8 aberrations). In this setting, analysis by 24-color karyotyping was particularly useful for resolving complex rearrangements such as ring chromosomes and derivative chromosomes, for example (Figure 1B).
Karyotype analysis in CD4+, CD56+ DC2 acute leukemia patient UPN3.
Representative R-banded (A) and M-FISH-Metasystems probe (B) karyotypes. Arrows indicate the presence of chromosomal abnormalities; large arrows identify recurrent anomalies in DC2 acute leukemia; small arrows identify other chromosomal changes. Modal number is 42. M-FISH allows to interpret add(1)(q41) as a der(1)t(1;8), add(2) as a dup(2), der(20) as a der(20)t(20;21), and add(21)(q22) as a der(21)(21p13 → 21q22::12::21q11→21qter). M-FISH also confirms the complete loss of chromosomes 9, 13, and 15 as well as partial 5q13q33 monosomy.
Karyotype analysis in CD4+, CD56+ DC2 acute leukemia patient UPN3.
Representative R-banded (A) and M-FISH-Metasystems probe (B) karyotypes. Arrows indicate the presence of chromosomal abnormalities; large arrows identify recurrent anomalies in DC2 acute leukemia; small arrows identify other chromosomal changes. Modal number is 42. M-FISH allows to interpret add(1)(q41) as a der(1)t(1;8), add(2) as a dup(2), der(20) as a der(20)t(20;21), and add(21)(q22) as a der(21)(21p13 → 21q22::12::21q11→21qter). M-FISH also confirms the complete loss of chromosomes 9, 13, and 15 as well as partial 5q13q33 monosomy.
Despite considerable karyotype complexity, chromosomal alterations clearly concentrated on 6 major chromosomal targets: 5q (10 of 14, 72%), 12p and 13q (9 cases each, 64%), 6q (7 of 14, 50%), 15q (6 of 14, 43%), and 9 (4 of 14, 28%). Only del(5q) and del(12p) were observed as isolated aberrations in one case each and could thus be considered as “primary” events. However, in the patient with del(5q) as the sole aberration, cells with del(5q) alone were part of the minor stemline, whereas the major deriving subclone contained as many as 9 additional aberrations. One patient had a simple hypodiploid karyotype with monosomy 9 and 13. All other patients displayed these aberrations in varying combinations, except one (UPN13) who presented a complex karyotype with del(13q) without any other aberration belonging to this major group of 6. Six patients showed evidence of karyotype progression as indicated by clonal evolution (UPN 3, 12, 13, 16, and 24) or the presence of a composite karyotype (UPN 4). Interestingly, in 3 of these 6 patients, del(12p), del(6q), and 5q rearrangements could be seen to occur as secondary events (one case each, Table 1).
No clear-cut association was observed between cytogenetic profiles (presence or absence of aberrations, degree of complexity, clonal evolution) and clinical presentation/outcome or cytologic or immunophenotypic patterns.
Recurrent cytogenetic anomalies in CD4+, CD56+ DC2 leukemia target–specific chromosomal regions
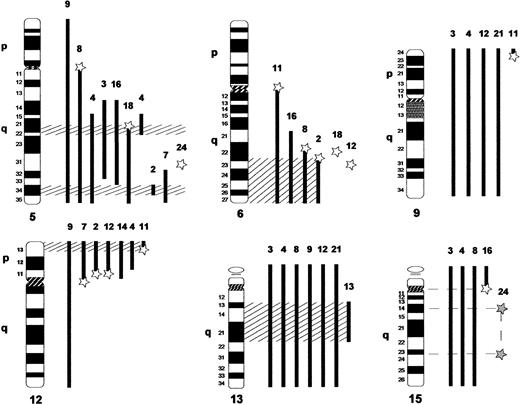
The cytogenetic and FISH data obtained for the 14 CD4+, CD56+ DC2 acute leukemia patients with clonal abnormalities in this study were further analyzed with the aim of determining chromosomal regions of potential pathologic significance in this disease. For the 6 major chromosomal rearrangements, clustering of deletion and translocation breakpoints into specific chromosomal bands was evident in most cases (Figure2).
Schematic representation of the chromosomal regions targeted by recurrent cytogenetic anomalies in CD4+, CD56+ DC2 acute leukemia.
Numbers correspond to patient UPN. Vertical solid black bars to the right of the chromosomes indicate the extent of deletion as determined by cytogenetics (R-banding and M-FISH) for each patient. Stars represent chromosomal breakpoints; open stars identify translocation breakpoints and filled stars identify inversion breakpoints. Stars combined with solid black bars indicate deletions resulting from unbalanced chromosomal rearrangements. Hatched lines delimit the minimal regions of deletion.
Schematic representation of the chromosomal regions targeted by recurrent cytogenetic anomalies in CD4+, CD56+ DC2 acute leukemia.
Numbers correspond to patient UPN. Vertical solid black bars to the right of the chromosomes indicate the extent of deletion as determined by cytogenetics (R-banding and M-FISH) for each patient. Stars represent chromosomal breakpoints; open stars identify translocation breakpoints and filled stars identify inversion breakpoints. Stars combined with solid black bars indicate deletions resulting from unbalanced chromosomal rearrangements. Hatched lines delimit the minimal regions of deletion.
Cytogenetic 5q aberrations included 7 total/partial 5q deletions and 3 translocations (2 unbalanced, 1 balanced). It is noteworthy that a single patient showed a 5q balanced translocation breakpoint in band 5q31. Two distinct regions of deletion localizing to bands 5q21 and 5q34, respectively, were evident. Codeletion of these 5q intervals was observed in 4 of 9 patients.
A total of 8 breaks (7 patients) was found in chromosome 6. One der(6) (UPN 4) and a complex interchromosomal rearrangement identified by M-FISH (UPN 18) could not be specified further. Bands 6q22-23 were affected by balanced or unbalanced translocations in 4 cases: no recurrent partner chromosome was observed (3p25, 18q22, 1q, and one addition of material of unknown origin). Combined with 2 other cases with larger 6q deletions, this defines region 6q23-qter as the common region of deletion.
Anomalies of chromosome 12 usually resulted in nearly complete 12p deletion with the exception of one case with a terminal 12p13 deletion, thus delineating band 12p13 as the critical region of deletion. Four deletions resulted from unbalanced translocations with variable chromosome partners (chromosome 1 in 2 cases, chromosome 5 and 7 in 1 case each).
Chromosome 13 was involved in 9 cases: 6 complete deletions, one small interstitial deletion 13q13-21, one ring, and one derivative 5 chromosome composed of part of 13q without further precision (UPN18). These results delineate bands 13q13-21 as the minimal region of deletion.
Finally, chromosome 9 and 15 were mostly involved in complete deletions as confirmed by 24-color karyotyping (Figure 1), except for a 9p24 addition and a 15-paracentric inversion (breaks in 15q14 and 15q23), which occurred as a secondary event in one patient.
Discussion
The present study constitutes the largest cytogenetic investigation available to date on CD4+, CD56+DC2 acute leukemia and documents for the first time the existence of a unique cytogenetic profile for this novel disease entity. Following conventional cytogenetic and 24-color FISH analyses in 14 clonally rearranged cases, 6 major recurrent chromosomal aberrations were defined. These were 5q, 12p, 13q, 6q, 15q, and 9, which were involved in 72% (5q), 64% (12p and 13q), 50% (6q), 43% (15q), and 28% (monosomy 9) of cases, respectively. No evidence for the existence of cytogenetic/clinical subgroups was apparent in this series. In particular, adults and children showed equally complex karyotype changes and the same recurring chromosomal aberrations were observed in both cases. No correlation with clinical presentation, cytology, or immunology was found.
To date, only 4 reports totaling 11 patients have dealt with chromosomal aberrations in CD4+, CD56+hematologic neoplasms1-4 (Table2). Five of these 11 patients showed an abnormal karyotype. Of these, one is also included in our series (case 1 in Petrella et al4 equals case 3, present study). Brody et al1 described a 63-year-old man with acute agranular CD4+ NK lymphoma for whom 4 serial cytogenetic analyses were performed during a 56-day period after admission. An identical hypodiploid complex karyotype that showed del(5q), monosomy 13 and 15, as well as a derivative 13q was observed at the first 3 analyses. Clonal evolution with appearance of a minor subclone with an additional del(17) (p13) appeared at the last control (56 days after initial diagnosis). All 3 further studies in the literature noted the complexity of these karyotypes, which, as in our study, tend to be hypodiploid with a net loss of material from chromosomes 5, 6, 9, 12, 13, and 15 (Table 2). Also, lineage-specific, balanced translocations/inversions typically observed in acute leukemia are markedly absent. Instead, the recurring chromosomal aberrations characterizing CD4+, CD56+ DC2 acute leukemia are shared by numerous hematologic malignancies.
Review of the literature on CD4+, CD56+ hematologic malignancies (cases with cytogenetic studies)
| Reference/ (case number) . | Sex/age . | Source of cells . | Status* . | Karyotype . |
|---|---|---|---|---|
| Brody et al1 | M/63 | BM | D | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[1]/46,XY[4] |
| PB | 23 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[20] | ||
| BM | 40 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[9]/46,XY[11] | ||
| PB | 56 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15/44,idem,del(17)(p13)[10%] | ||
| DiGuiseppe et al2(3) | M/79 | BM | D | ?,der(1)t(1;1)(q42;q11),t(7;12)(p13;p13)inc[5]/46,XY[64] |
| ? | 30 d | 44,XY,der(1)t(1;1)(q42;q11),t(7;12)(p13;p13),-13,-17,der(19)t(17;19)(q21;p13)[8]/46,XY[11] | ||
| DiGuiseppe et al2 (1) | F/81 | ? | D | 46,XX[?] |
| Kameoka et al3 (1) | F/21 | BM | D | 46,XX[?] |
| Kameoka et al3(2) | M/81 | BM | D | 45,XY,der(7)t(1;7)(q11;p22),der(12;15)(q10;q10),add(13)(q12)[7]/44,idem,-9[4]/44,idem,del(3)(p25),-9[3]/46,XY[6] |
| Petrella et al4 (1)† | F/86 | BM | D of AL | 46,XX,del(5)(q13q33)[2]/42,idem,-9,-12,-13,-15,-21,+3mar[20]/46,XY{2] |
| Petrella et al4 (3) | F/67 | Skin | R | Failure |
| Petrella et al4(5) | F/65 | LN | D | 43,XX,der(1)t(1;15)(p22;q14),der(2)t(2;6;9)(p23;?;?),del(5)(q12q34),del(6)(q21),-9,-12,-13.ish der(15)t(1;15)(p?;q21)(wcp1+)[12]/46,XX{17] |
| Petrella et al4 (4) | M/84 | BM | D of AL | 45,X,-Y[2]/46,XY[36] |
| Petrella et al4(7) | M/37 | Skin | R | 46,XY[10] |
| Reference/ (case number) . | Sex/age . | Source of cells . | Status* . | Karyotype . |
|---|---|---|---|---|
| Brody et al1 | M/63 | BM | D | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[1]/46,XY[4] |
| PB | 23 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[20] | ||
| BM | 40 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15[9]/46,XY[11] | ||
| PB | 56 d | 44,XY,del(5)(q13),-13,der(13)t(11;13)(q12;q32),-15/44,idem,del(17)(p13)[10%] | ||
| DiGuiseppe et al2(3) | M/79 | BM | D | ?,der(1)t(1;1)(q42;q11),t(7;12)(p13;p13)inc[5]/46,XY[64] |
| ? | 30 d | 44,XY,der(1)t(1;1)(q42;q11),t(7;12)(p13;p13),-13,-17,der(19)t(17;19)(q21;p13)[8]/46,XY[11] | ||
| DiGuiseppe et al2 (1) | F/81 | ? | D | 46,XX[?] |
| Kameoka et al3 (1) | F/21 | BM | D | 46,XX[?] |
| Kameoka et al3(2) | M/81 | BM | D | 45,XY,der(7)t(1;7)(q11;p22),der(12;15)(q10;q10),add(13)(q12)[7]/44,idem,-9[4]/44,idem,del(3)(p25),-9[3]/46,XY[6] |
| Petrella et al4 (1)† | F/86 | BM | D of AL | 46,XX,del(5)(q13q33)[2]/42,idem,-9,-12,-13,-15,-21,+3mar[20]/46,XY{2] |
| Petrella et al4 (3) | F/67 | Skin | R | Failure |
| Petrella et al4(5) | F/65 | LN | D | 43,XX,der(1)t(1;15)(p22;q14),der(2)t(2;6;9)(p23;?;?),del(5)(q12q34),del(6)(q21),-9,-12,-13.ish der(15)t(1;15)(p?;q21)(wcp1+)[12]/46,XX{17] |
| Petrella et al4 (4) | M/84 | BM | D of AL | 45,X,-Y[2]/46,XY[36] |
| Petrella et al4(7) | M/37 | Skin | R | 46,XY[10] |
BM indicates bone marrow; PB, peripheral blood; LN, lymph node; D, diagnosis; d, day; D of AL, diagnosis of acute leukemia.
Status at the time of sample.
This case is also included in our study (UPN3).
Deletion 5q is the most frequent chromosomal rearrangement observed in our series of CD4+, CD56+, DC2 acute leukemia (10 of 14 patients). Deletion 5q is the primary aberration in the 5q− syndrome but is also one of the hallmarks of secondary myelodysplastic syndromes (MDSs)/acute myeloid leukemia where it generally accompanies partial or complete monosomy 7q, trisomy 8 or 21, and deletion 12p.14 In our cases, however, del(5q) was not accompanied by any of these additional aberrations except for deletion 12p. We looked for the presence of MDS features through a careful review of medical records as well as of bone marrow aspirates and peripheral blood films in 20 of 21 patients included in this series.5 Five patients presented significant dysplastic features of one or more lineages (UPN 1, 4, 14, 15, and 20). Two patients (UPN 1 and 15) had a prior history of MDS. However, they presented a normal karyotype at the time of both MDS and CD4+, CD56+ DC2 acute leukemia diagnosis. In fact, 3 of 5 patients with MDS features at diagnosis had a normal karyotype, 1 had an isolated deletion 12p, and only 1 had a complex karyotype with del(5q). Conversely, of 10 patients with del(5q), only 1 had morphologic signs of MDS, ruling out a direct link between 5q−, MDS, and CD4+, CD56+DC2 acute leukemia.
Two different critical regions of deletion, 5q31 and 5q32, have been specified for acute myeloid leukemia (AML)/some MDS and for the 5q− syndrome, respectively.15 16 In our series of CD4+, CD56+ DC2 acute leukemia, 6 of 9 and 7 of 9 5q deletions also involved band 5q31 and 5q32, respectively. Although a number of candidate tumor suppressor genes have been mapped to a minimal deletion interval at 5q31, no single tumor suppressor gene has as yet been identified. It is of interest that 2 of our patients showed small distinct 5q deletions (Figure 2). These centered on bands 5q21 and 5q34, respectively, thus suggesting the involvement of other tumor suppressor loci in this disease.
Noteworthy, deletion 5q was observed as an isolated abnormality in the stemline clone in one patient. Indeed, combined immunophenotyping and FISH studies in MDS patients have demonstrated the presence of del(5q) not only in granulocytic, monocytic, and erythroid lineages but also in B cells (but not T cells).17,18However, use of interphase FISH analysis on purified cell populations has shown that early lymphomyeloid hematopoietic stem cells are the primary target for 5q deletions in MDS. Extensive functional analyses indicate that these 5q deleted stem cells are inefficient at reconstituting hematopoiesis.18
Deletion 12p was observed in 9 of 14 of our DC2 acute leukemia patients. Deletion/breaks at 12p13 are frequent changes in acute lymphoid as well as myeloid disorders. Also, a recurrent t(6;12)(q15;p13) has been identified in chronic lymphoproliferative disorders.19 Molecular studies have identifiedTEL/ETV6 as the target gene involved in most of these chromosomal rearrangements. In one DC2 leukemia case tested here, FISH analysis confirmed heterozygous loss of the TELgene. Other genes may, however, be targeted by 12p rearrangements in hematologic malignancies. Notably, cloning of a t(12;22)(p13;q11) in one case of chronic lymphocytic leukemia (CLL) at relapse, identified a breakpoint within the negative regulatory element of the cyclin D2 promoter.20
Half of our patients with DC2 acute leukemia showed del(6q). This aberration is generally observed as an isolated anomaly, in lymphoid disorders, chronic lymphocytic lymphoma/leukemia, as well as acute lymphoblastic leukemia particularly of T-cell lineage. Deletion 6q is, however, also observed as a frequent secondary aberration in association with Ig or T-cell receptor loci translocations in NHL of B- or T-cell subtypes.21 22 In marked contrast to the situation in chronic lymphoid proliferations, no translocations involving B- or T-cell–receptor gene loci were associated with the 6q− in our cases.
Deletion 6q is characterized by considerable breakpoint heterogeneity, and a correlation between del(6q) breakpoint site and disease subtype is evident. For example, bands 6q15, 6q21, 6q23-24, and 6q25-27 represent frequent deletion sites in lymphoma.23,24 Deletion 6q21q23 is specifically associated with a subset of B-cell CLL (B-CLL),25 whereas del(6)(q21) is associated with high-grade lymphoma.26 Deletion of bands 6q22-23 was recurrent in this series of DC2 leukemia patients. It is noteworthy that del(6q), with recurrent loss of bands 6q22-23 is also one of the most frequent aberrations in NK cell lymphoma/leukemia.27 Extensive FISH/loss of heterozygosity studies in large series of both low- and high-grade NHL of T- and B-cell subtype and acute lymphocytic leukemia (ALL) have led to the identification of several commonly deleted regions in 6q21, some of which were shown to overlap.26
Deletion 13q was observed in 9 of 14 patients with abnormal karyotype in the series (64%). Specific deletion of bands 13q14-21 was observed in one case. Deletion 13q involving region 13q14-21 is found in a wide variety of chronic lymphoid and acute/chronic myeloid disorders. Deletion 13q14 is the most frequent chromosome aberration in CLL (55% of CLL patients28). When present as an isolated aberration (66% of patients with del 13q14, del 13q14 is correlated with a prolonged treatment-free interval. This is at variance with multiple myeloma where del 13q14 is encountered in up to 86% of FISH-studied cases29 and is of adverse prognosis.30 Deletion 13q is also a common secondary aberration in lymphomas.31 It is considered as a primary aberration, although at a lower frequency in chronic as well as acute myeloid disorders. Finally, del(13q) has been reported in NK leukemia/lymphoma.27 The respective minimal regions of 13q14 deletion have been shown to overlap in myeloid and lymphoid disorders.32 However, despite considerable molecular cloning and sequencing efforts, all 3 transcriptional units identified in the 300-kb interval encompassing the 13q14 minimal region of deletion of B-CLL were discarded as candidate tumor suppressor genes, as far as not fulfilling a classical 2-hit inactivation mechanism.33 The identity of the tumor suppressor gene(s) targeted by these rearrangements thus remains elusive.
Finally deletion/rearrangement of chromosome 15 and monosomy 9 were observed in, respectively, 6 of 14 and 4 of 14 CD4+, CD56+ DC2 acute leukemia patients in this series. One further patient showed addition of unknown material to 9p24. These chromosomal aberrations are generally rare in hematologic malignancies but 15q anomalies have been reported in 4 of 5 previously published DC2 leukemia cases with abnormal karyotypes (Table 2).1-4Chromosome 9 rearrangements generally involve loss of 9p21 (possibly targeting the p16 gene) or 9q [mostly acute myeloid leukemia with or without t(8;21)] or trisomy 9 in chronic myeloproliferative disorders or multiple myeloma.
CD4+, CD56+ tumors are referred to as blastic NK lymphoma in the WHO classification and were previously reported as plasmacytoid T-cell lymphoma.34-37 Three of the 5 previous cases34-37 presented with acute or chronic myelomonocytic leukemia (CMML) thus suggesting a potential monocytic origin for these neoplastic cells. Isolated 12p aberrations described in previous series of CMML38 are also encountered in DC2 acute leukemias. In a few CMML cases, molecular studies have demonstratedTEL gene involvement, notably in the t(5;12)(q33;p13).39 However, the high frequency of 6q deletions combined with absence of 7q deletion or trisomy 8 is not consistent with a myelomonocytic origin for this disease. Indeed, it is now known that CD4+, CD56+ tumors are derived from the recently characterized plasmacytoid dendritic cell (pDC).6 40
This detailed cytogenetic analysis of CD4+, CD56+ leukemia confirms the clonal nature of this novel disease entity. The cytogenetic features can be summarized as follows: (1) gross genomic imbalances (mostly losses) tend to predominate over gene-specific rearrangements (recurrent chromosomal translocations, for example), (2) no single anomaly may be considered as specific, whereas their combination/accumulation is, and (3) both lymphoid- and myeloid lineage–associated rearrangements are observed in unusual combinations in the same cell. This is suggestive of complex multistep tumorigenic mechanisms that potentially involve loss of function or gene dosage anomalies at multiple target loci. Finally the coexistence of myeloid, T- and B-cell, and NK lymphoid–associated chromosomal abnormalities in CD4+, CD56+ leukemic cells would tend to support the hypothesis that these cells originate from more undifferentiated progenitors than lymphoid-restricted progenitors as previously suggested.41
We wish to express our gratitude to the clinicians and biologists who provided patients and data for our study. We deeply thank our technical staff for their unfailing dedication and high level of professionalism. We are particularly grateful to Dr Roland Berger for his encouragement and helpful discussions. We thank Dr Patricia Le Baccon for help with the bibliography searches and Michel Leroux for expert design of the figures. We also thank Cécile Meyrieux for excellent secretarial assistance.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Dominique Leroux, Lymphoma Research Group and Laboratory of Cellular and Molecular Hematology, Department of Cell Biology and Pathology, Grenoble University Hospital, BP217, 38043 Grenoble cedex 9, France; e-mail:dleroux@chu-grenoble.fr.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal