Abstract
Acute infectious mononucleosis (AIM) induced by Epstein-Barr virus (EBV) infection is characterized by extensive expansion of antigen-specific CD8+ T cells. One potential consequence of this considerable proliferative activity is telomere shortening, which predisposes the EBV-specific cells to replicative senescence. To investigate this, a method was developed that enables the simultaneous identification of EBV specificity of the CD8+ T cells, using major histocompatibility complex (MHC) class I/peptide complexes, together with telomere length, which is determined by fluorescence in situ hybridization. Despite the considerable expansion, CD8+ EBV-specific T cells in patients with AIM maintain their telomere length relative to CD8+ T cells in normal individuals and relative to CD4+ T cells within the patients themselves and this is associated with the induction of the enzyme telomerase. In 4 patients who were studied up to 12 months after resolution of AIM, telomere lengths of EBV-specific CD8+ T cells were unchanged in 3 but shortened in one individual, who was studied only 5 months after initial onset of infection. Substantial telomere shortening in EBV-specific CD8+ T cells was observed in 3 patients who were studied between 15 months and 14 years after recovery from AIM. Thus, although telomerase activation may preserve the replicative potential of EBV-specific cells in AIM and after initial stages of disease resolution, the capacity of these cells to up-regulate this enzyme after restimulation by the persisting virus may dictate the extent of telomere maintenance in the memory CD8+ T-cell pool over time.
Introduction
Primary infection with Epstein-Barr virus (EBV) leads to a syndrome known as acute infectious mononucleosis (AIM) characterized by a massive expansion of CD8+ T cells, which controls the spread of virus.1-3 This increase in CD8+ T-cell numbers is not unique to EBV infections; different viruses, including varicella zoster and influenza in humans4 and lymphocytic choriomeningitis virus (LCMV) in mice,5,6 all induce similar changes. Disease resolution is associated with a dramatic reduction in cell numbers to preinfection levels and this homeostasis is achieved by the induction of apoptosis in the expanded CD8+ T-cell pool.3,4,7,8However, for a memory CD8+ T-cell population to persist after the original immune challenge, some specific cells have to be retained and this involves mechanisms that prevent apoptosis and enable them to survive in vivo.5 9
The use of tetrameric major histocompatibility complex (MHC) class I/peptide complexes (tetramers) to identify antigen-specific CD8+ T cells has shown that in patients with acute EBV infection10 and also in mice with LCMV infections11 up to 50% of CD8+ T cells may be specific for a single viral epitope.2,10 It is estimated that the number of divisions required to reach such expansions would require the precursor populations to undergo at least 28 population doublings.12 This level of expansion would result in telomere shortening and be sufficient to induce replicative senescence.13 Telomeres are repeating hexameric sequences of DNA found at the ends of chromosomes, which are responsible for maintaining chromosomal stability and integrity.14With each cell division, between 50 and100 base pairs (bp) of telomeric DNA are lost due to the inability of DNA polymerase to fully replicate the ends of chromosomes. Critically short telomeres trigger cell cycle arrest and replicative senescence.15 This raises the possibility that virus-specific CD8+ T cells, which escape apoptosis and enter the memory pool, may have reduced replicative capacity,16 17 thus reducing the ability of these cells to proliferate on re-encounter with virus.
To assess the impact of telomere loss in antigen-specific CD8+ T cells during acute EBV infection, a method that combines the use of MHC class I tetramers with telomere analysis is required. Current techniques that are most commonly used for telomere length measurement would require the isolation of large numbers of tetramer-positive cells for Southern blot analysis of telomeric restriction fragments (TRFs).18 The labor-intensive isolation steps required for this18 and the substantial reduction of tetramer-binding cells in blood samples after resolution of the infection10 would preclude the routine analysis of large numbers of samples by this method. Another protocol for measuring telomere length involves fluorescence in situ hybridization (FISH) using fluorescencelabeled telomere probes on fixed cell suspensions, which are then analyzed for staining by flow cytometry (flow-FISH).19-21 However, in previous studies this method has only been used to measure telomere length in isolated subpopulations of cells, therefore once again requiring the purification of tetramer-positive CD8+ T cells.
In this study we describe a rapid way to identify simultaneously the telomere length and surface tetramer staining of individual EBV-specific CD8+ T cells by 2-color FISH and flow cytometry (2-color flow-FISH). Using this technique we demonstrate that despite the massive expansion of the EBV-specific CD8+ T cells during acute infection, they preserve their telomere length and that this is associated with up-regulation of the enzyme telomerase.
Patients, materials, and methods
Isolation of peripheral blood mononuclear cells
Heparinized peripheral blood was collected from patients with AIM and from the same patients at different times following resolution of acute infection as described.10 These samples were cryopreserved in liquid nitrogen and the phenotypes of acute and follow-up samples were analyzed simultaneously after thawing as described.2 Cryopreserved peripheral blood mononuclear cells (PBMCs) were isolated using Ficoll-Hypaque (Nycomed, Oslo, Norway) density centrifugation. PBMCs from healthy laboratory staff were used as controls. Purified CD8+ T cells were separated by negative selection using the VARIO MACS system (Miltenyi Biotec, Surrey, United Kingdom) according to the manufacturer's instructions. Briefly, PBMCs were incubated with CD16 (Immunotech, Luton, United Kingdom), CD14 (UCHM-1), CD19 (RFB9), and CD4 (RFT4) at pretitrated optimal levels for 30 minutes followed by incubation with goat-antimouse IgG microbeads (Miltenyi Biotec) for 20 minutes at 4°C. Cells were then washed and run through appropriate-sized depletion columns. The unbound cells were collected. Purity was over 95% as determined by flow cytometry. EBV-specific cells were enriched using positive selection. Purified CD8+ T cells were incubated with phycoerytheryn (PE)-conjugated MHC class I/peptide tetramers for 30 minutes at 4°C, washed, and then further incubated with anti-PE microbeads (Miltenyi Biotec). Cells were run through a positive selection column with the CD8+ tetramer-enriched population being collected from the bound fraction.
Cell staining for flow cytometric analysis
FACSCalibur (Becton Dickinson, Oxford, United Kingdom) analysis of T-cell phenotype was performed as previously described10using different combinations of the antibodies CD4, CD8, CD25, CD69, and MHC class II DR (all from Dako, Poole, United Kingdom). MHC class I peptide tetrameric complexes, specific for EBV epitopes, were linked to either streptavidin-PE (Sigma Chemical, Dorset, United Kingdom) as previously described10 or to streptavidin-Cy5 (Jackson Immunoresearch Laboratories, West Grove, PA). For 2-color flow-FISH we used Cy5 because it is heat stable, whereas PE is not. Two different EBV-specific MHC class I peptide tetramers were generated. The human leukocyte antigen (HLA) A2 peptide ligand was GLCTLVAML (from the EBV lytic protein BMLF1) and the HLA B8 peptide ligand was RAKFKQLL (from the EBV lytic protein BZLF1). These tetramers are referred to henceforth as A2-GLC and B8-RAK, respectively. Cells were incubated with either antibodies (10 minutes) or MHC class I/peptide tetramers (30 minutes) at 4°C, then washed and fixed in 2% paraformaldehyde in phosphate-buffered saline (PBS) prior to analysis. Data analysis was performed using Cell Quest software (Becton Dickinson).
Assessment of the mean TRF length using Southern blot
Genomic DNA was extracted from frozen samples using the WIZARD Genomic DNA purification Kit (Promega Ltd, Southampton, United Kingdom) and digested for 6 hours at 37°C with restriction enzymes MspI and RsaI (both from Pharmacia Biotech, Herts, United Kingdom). The digests were then electrophoresed in an 0.7% agarose gel for 48 hours at 20 V. The gel was then subjected to depurination, denaturation in an alkaline solution followed by neutralization. The DNA was transferred onto a nitrocellulose membrane (Hybond N+, Amersham Life Science, Amersham, United Kingdom) by Southern blotting and fixed by baking at 80°C. Prior to hybridization with the telomeric probe, the membranes were prehybridized with Rapid Hyb Buffer (Amersham Life Science). Hybridization was performed at 42°C for 1 hour with the telomeric specific probe (TTAGGG)3, which had previously been labeled with 32γP [ATP] using polynucleotide kinase (Pharmacia Biotech). Autoradiographs were obtained by exposing the autoradiography film (Hyperfilm MP, Amersham Life Science) to the hybridized membrane at −70°C for the appropriate amount of time. TRF length was calculated by densitometry of autoradiographs within the linear range using MultiAnalyst software (BioRad, Herts, United Kingdom). The mean TRF length was calculated as described.18
Assessment of telomere length using flow-FISH
Telomere length was determined using the flow-FISH technique as described recently.19 Briefly, PBMCs were washed in phosphate-buffered BSA (PBSA) (0.2% bovine serum albumin [BSA]) followed by permeabilzation using Ortho PermeaFix (Ortho Diagnostic Systems, Bucks, United Kingdom) for 30 minutes in the dark. Samples were then washed twice in PBSA followed by a wash in 1 mL hybridization buffer (70% deionized formamide [BDH, Poole, United Kingdom], 20 mM Tris pH 7, 1% BSA [both from Sigma Chemical]). Samples were then resuspended in 200-μL hybridization buffer and incubated with 0.3 μg/mL of the PNA telomeric (C3TA2)3 or the PNA control probe (alphoid sequences of the X chromosome, CCCATAACTAAACAC) both conjugated to fluorescein isothiocyanate (FITC), described previously.19-21 PNA probes were obtained from Perseptive Biosystems (Framingham, MA). Samples were incubated for 20 minutes in the dark followed by heating at 80°C for 10 minutes. Cells were rapidly cooled and further hybridized for 2 hours at room temperature in the dark. Samples were washed twice in posthybridization buffer (70% formamide [BDH], 10 mM Tris, 0.1% BSA, 0.1% Tween 20 [all from Sigma Chemical]) followed by 2 further washes in PBSA. Samples were then analyzed by flow cytometry (FACSCalibur cytometer, Becton Dickinson) using Cell Quest Software (Becton Dickinson). Polyfluorescent beads (Dako Fluospheres, Dako, Denmark) were used at the beginning of each experiment to standardize the cytometer. Following the last wash, samples were incubated with propidium iodide (5 μg/mL; Sigma Chemical) and RNAase (50 ng/mL; Sigma Chemical) for 15 minutes for the exclusion of doublets as determined by FL3-area versus FL3-width.
Assessment of telomere length in specific populations using 2-color flow-FISH
Cells were first stained with biotin-labeled anti-CD4 or CD8-biotin (Immunotech) followed by streptavidin-Cy5 (Southern Biotech Associates, Inc, supplied by Euro-Path, Cornwall, United Kingdom) or with A2-GLC–Cy5/B8-RAK–Cy5. These cells were then washed, fixed, and permeabilized using Ortho Permeafix (Ortho Diagnostic Systems, Raritan, NJ) and processed for flow-FISH as described above. Analysis was performed using the CellQuest software as described above.
Measurement of telomerase activity
A modified version of the telomeric repeat amplification protocol (TRAP) was used (Oncor Inc, Gaithersburg, MD) as previously described.22 Extracts from varying cell numbers were used for telomeric elongation, using a 33γP[ATP] (Amersham Life Science) end-labeled primer. These samples were used for polymerase chain reaction (PCR) amplification (Perkin-Elmer Cetus, Norwalk, CT), using 25 to 28 cycles of 30 seconds at 94°C and 30 seconds at 59°C. The PCR products were run on a 12% polyacrylamide gel (Amersham Life Science), which was vacuum dried before exposure to autoradiography film (Hyperfilm MP, Amersham Life Science). Extracts from the immortalized 293 cell line provided the positive control. The negative controls were obtained by heat inactivation of the RNA template for each cell extract used. In addition, lysis buffer was used in place of cell extract in one reaction tube. Telomerase activity was calculated according to the manufacturer's instructions using the optical density of the telomeric repeat bands, on samples and heat-inactivated controls, divided by the strength of the internal PCR control band (which also served to indicate the absence of Taq inhibitors).
Results
Characterization of 2-color flow-FISH method
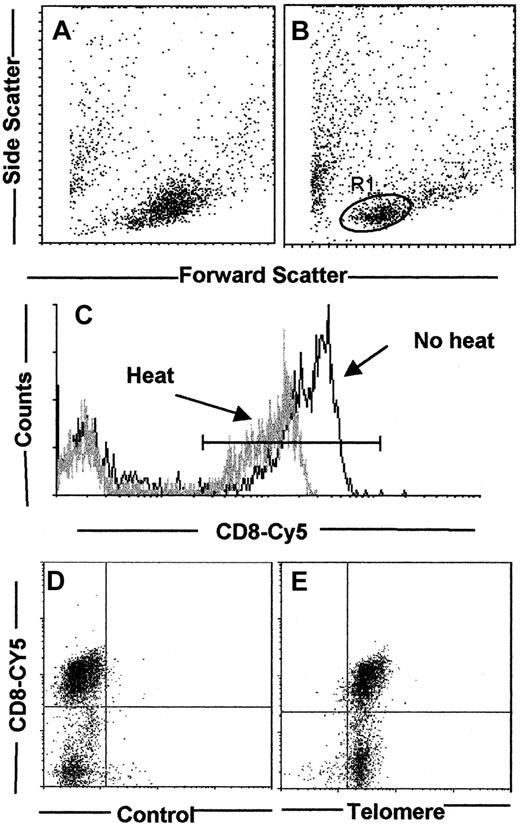
The first aim of the study was to develop a method for the analysis of telomere length in small subsets of cells without the need for their prior isolation. To do this, we adapted a technique that has been previously described,20,21 but we have included an initial surface staining step. The technique described by Rufer and colleagues19 involves heating cells in suspension for 10 minutes at 80°C to enable dissociation of DNA strands, which allows access to the FITC-labeled telomeric probe. This heating step did not substantially alter the forward scatter (FSC) and side scatter (SSC) characteristics of normal human T cells (Figure1A,B). Heated cells were stained with propidium iodide and analyzed on the flow cytometer. For single cells, the area and width were set using the gain adjustments for equal pulse heights. Doublets were excluded by their wider pulse area than the single cells of comparable DNA content. Using this method to exclude cellular aggregates from the analysis, single cells were then gated by their FSC and SSC characteristics. We only analyzed cells located within the R1 gate shown in Figure 1B, which only contains nonblastoid cells in the G0 or early G1 phase of the cell cycle (F.J.P. and A.N.A., unpublished observations, 2000). Complications that arise from the increased FISH signal in cycling cells compared to resting cells can therefore be ruled out.21 To characterize the telomere length in CD8+ T-cell subsets within a mixed population, surface labeling was performed first using biotinylated anti-CD8 antibody followed by addition of a Cy5-coupled streptavidin second layer, chosen because Cy5 is heat stable. The PBMCs were stained for CD8, then fixed, and percentage positivity determined by flow cytometry before and after heating for 10 minutes at 80°C (Figure 1C). Although heating induced a reduction in intensity of CD8 staining, the percentage of CD8+ cells remained relatively unchanged (75.3% before versus 69.5% after heating). We next investigated if we could detect telomeres by flow-FISH in surface-labeled cells. Cells were first labeled with anti-CD8 biotin and Cy5-labeled streptavidin. These cells were then fixed, permeabilized, and heated to 80°C as above. The cells were then stained with the FITC-labeled X-chromosome control probe (Figure 1D) or the FITC-labeled telomeric probe (Figure 1E) followed by flow cytometric analysis. The percentage of CD8+ T cells detected in cultures with the control or telomeric probe was 68% and 65%, respectively (Figure 1D,E). Although the control probe showed a low level of staining, all the CD8+ T cells were stained with the telomeric probe. These results demonstrated the feasibility of detecting telomere length in subsets of cells in a mixed population by 2-color flow-FISH.
Standardization of the 2-color flow-FISH technique.
The effect of heating on FSC and SSC profiles was investigated. Control PBMCs were fixed with 2% paraformaldehyde and analyzed before (A) and after (B) heating at 80°C for 10 minutes. Gates for telomere analysis were determined by the addition of propidium iodide for the exclusion of cell debris from dead/dying cells, and also doublets and blastoid cells (data not shown). Cells within the R1 gate were stained with anti-CD8 coupled to Cy5, fixed, and analyzed before and after heating to 80°C (C). Although the MFI for CD8-Cy5 decreased from 167 for the control to 78 in the heated sample, percentage of CD8+ T cells was relatively unaffected by the heating protocol (23.6% before, 19% after). PBMCs were stained with anti-CD8 biotin followed by streptavidin-Cy5. Cells were then fixed and permeabilized and processed for flow-FISH as described in “Patients, materials, and methods” with either the control X-chromosome FITC PNA probe (D) or the FITC-labeled PNA telomeric probe (E).
Standardization of the 2-color flow-FISH technique.
The effect of heating on FSC and SSC profiles was investigated. Control PBMCs were fixed with 2% paraformaldehyde and analyzed before (A) and after (B) heating at 80°C for 10 minutes. Gates for telomere analysis were determined by the addition of propidium iodide for the exclusion of cell debris from dead/dying cells, and also doublets and blastoid cells (data not shown). Cells within the R1 gate were stained with anti-CD8 coupled to Cy5, fixed, and analyzed before and after heating to 80°C (C). Although the MFI for CD8-Cy5 decreased from 167 for the control to 78 in the heated sample, percentage of CD8+ T cells was relatively unaffected by the heating protocol (23.6% before, 19% after). PBMCs were stained with anti-CD8 biotin followed by streptavidin-Cy5. Cells were then fixed and permeabilized and processed for flow-FISH as described in “Patients, materials, and methods” with either the control X-chromosome FITC PNA probe (D) or the FITC-labeled PNA telomeric probe (E).
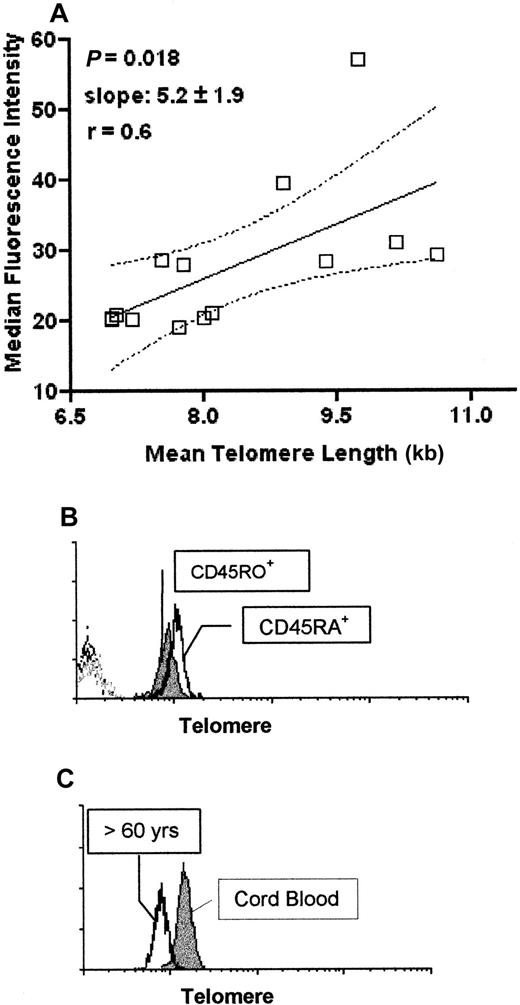
Analysis of telomere length by flow-FISH in healthy individuals
We compared telomere length changes obtained using the flow-FISH method with those obtained by measuring TRFs by Southern blot analysis. PBMCs isolated from 14 different healthy individuals of different ages were analyzed for telomere length using both methods (Figure 2A). We found a significant correlation between flow-FISH and TRF analysis (P = .018), confirming previous results.19,20 Using the flow-FISH method we also confirmed that isolated CD45RA+ T cells have longer telomeres than CD45RO+ T cells (Figure2B). We next compared the telomere lengths of mononuclear cell populations from 4 cord blood and 4 adult samples (individuals over 60 years old) using the flow-FISH method (representative experiment in Figure 2C). We found that the older adults showed significantly decreased telomeres as measured by flow-FISH (average median fluorescence intensity [MFI] 5.9 compared to 11.3 in the cord samples) as assessed by the Mann-Whitney test (P < .029). When TRF analysis was performed on the same samples, the cord samples also showed longer telomeres compared to the older individuals as assessed by the Mann-Whitney test (P = .0029; data not shown). Collectively these results indicate that the flow-FISH method of telomere analysis gives the same results as TRF analysis in human lymphocyte populations confirming previous reports.19-21
Comparison of telomere analysis by flow-FISH technique and Southern blotting.
A total of 14 PBMC samples were collected from individuals of different ages ranging from umbilical cord blood samples to individuals over 70 years old. Cells were cyropreserved in 10% DMSO for telomere length analysis by flow-FISH and snap frozen as pellets for Southern blotting. Cells analyzed for telomere length by flow-FISH were all processed and run on the same day and all samples for Southern blot analysis were also run simultaneously (A). Purified CD45RA+and CD45RO+ T lymphocytes were purified using the MACS separation protocols (see “Patients, materials, and methods”) and then processed for flow-FISH as described (B). The histogram profiles show CD45RA+ T cells (open histogram) and CD45RO+ T cells (closed histogram). Dotted histogram represents the control X-chromosome–FITC probe. Comparison of telomere length using the flow-FISH of umbilical cord blood PBMCs and PBMCs from an individual over 60 years of age is shown in panel C. The histogram shows cord blood PBMCs (closed histogram) versus PBMCs from an over 60 year old (open histogram). Dotted histogram represents the control PNA probe (X-chromosome–FITC). All cells analyzed for flow-FISH were gated on live, nonblastoid cells as described (Figure 1B). At least 50 000 live events were acquired.
Comparison of telomere analysis by flow-FISH technique and Southern blotting.
A total of 14 PBMC samples were collected from individuals of different ages ranging from umbilical cord blood samples to individuals over 70 years old. Cells were cyropreserved in 10% DMSO for telomere length analysis by flow-FISH and snap frozen as pellets for Southern blotting. Cells analyzed for telomere length by flow-FISH were all processed and run on the same day and all samples for Southern blot analysis were also run simultaneously (A). Purified CD45RA+and CD45RO+ T lymphocytes were purified using the MACS separation protocols (see “Patients, materials, and methods”) and then processed for flow-FISH as described (B). The histogram profiles show CD45RA+ T cells (open histogram) and CD45RO+ T cells (closed histogram). Dotted histogram represents the control X-chromosome–FITC probe. Comparison of telomere length using the flow-FISH of umbilical cord blood PBMCs and PBMCs from an individual over 60 years of age is shown in panel C. The histogram shows cord blood PBMCs (closed histogram) versus PBMCs from an over 60 year old (open histogram). Dotted histogram represents the control PNA probe (X-chromosome–FITC). All cells analyzed for flow-FISH were gated on live, nonblastoid cells as described (Figure 1B). At least 50 000 live events were acquired.
Analysis of telomere length in MHC class I tetramer–binding cells
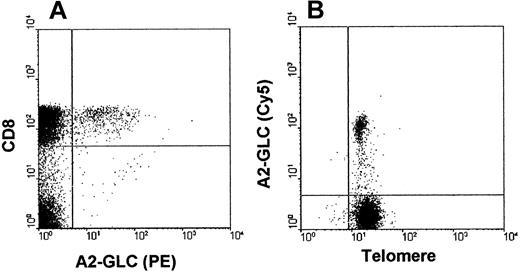
We next investigated if we could perform telomere staining in A2-GLC tetramer–positive cells, which was used to identify EBV-specific CD8+ T cells. The specificity of the A2-GLC tetramer staining has been described previously.10 A representative labeling profile is shown in Figure3A. In this study A2-GLC tetramer–positive cells composed between 2% and 11% of the CD8 pool of AIM patients. To identify telomere staining the tetramer was conjugated to Cy5 instead of PE because PE is degraded by heating. After staining with the Cy5-conjugated A2-GLC tetramer, the cells were fixed, permeabilized, and then heated to enable staining with the telomeric probe. Using this method we were able to detect telomere labeling in tetramer-specific cells (Figure 3B). We also showed that the telomere length detected by flow-FISH in A2-GLC–positive cells was almost identical when whole mononuclear cell populations were labeled (MFI 16) or when CD8+ T cells were first isolated from the mononuclear cell population before labeling (MFI 16.5).
Determination of telomere length in MHC class I/peptide tetramer staining on PBMCs isolated from AIM patients.
(A) PBMCs from an AIM patient stained with CD8 RPE-Cy5 and HLA-A2 GLC-PE tetramer. Cells were gated to exclude doublets and dead/dying cells as determined by propidium iodide (Figure 1B). At least 50 000 live events were acquired on the flow cytometer. (B) Two-color flow-FISH analysis of telomere staining (FITC) of A2-GLC tetramer–positive cells (A2-GLC–Cy5) in an acute AIM patient. At least 50 000 live events were acquired.
Determination of telomere length in MHC class I/peptide tetramer staining on PBMCs isolated from AIM patients.
(A) PBMCs from an AIM patient stained with CD8 RPE-Cy5 and HLA-A2 GLC-PE tetramer. Cells were gated to exclude doublets and dead/dying cells as determined by propidium iodide (Figure 1B). At least 50 000 live events were acquired on the flow cytometer. (B) Two-color flow-FISH analysis of telomere staining (FITC) of A2-GLC tetramer–positive cells (A2-GLC–Cy5) in an acute AIM patient. At least 50 000 live events were acquired.
Comparison of telomere length in T-cell subsets of normal and EBV patients
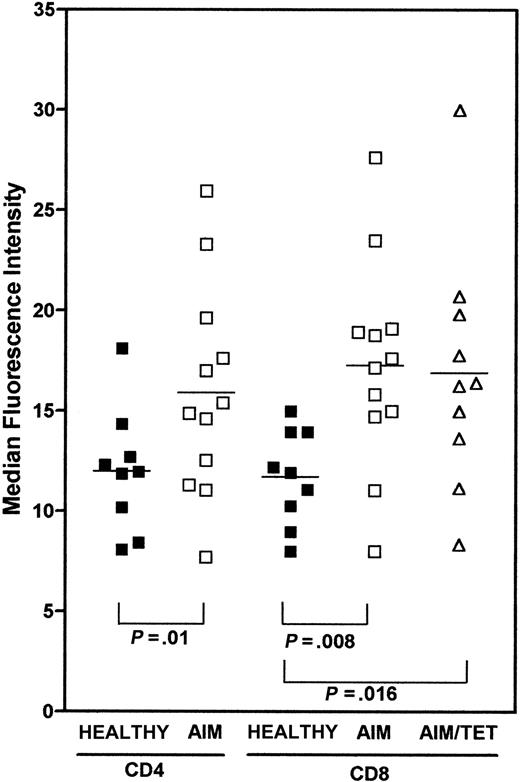
Using the 2-color flow-FISH method, we investigated telomere length in CD4+ and CD8+ T cells in 9 healthy controls and compared these to the CD4+, CD8+, and A2-GLC tetramer–positive cells of 12 AIM patients (Figure4). In these studies, control samples were always processed at the same time as AIM patient samples. Reproducible results were also obtained when the same samples were run on separate occasions. In addition, telomere length in different T-cell subsets were compared within the same patients themselves, allowing them to act as their own controls. Although most CD8+ T cells from these patients expressed HLA class II, indicating that they were activated (Figure 6A) and contained oligoclonal expansions generated by repeated cell division,23 24 they had significantly longer telomeres than CD8+ T cells from normal individuals (Figure 4;P = .008). The A2-GLC tetramer–positive cells in AIM patients were also activated, as shown by elevated class II expression (Figure 6A). Like the total CD8+ T-cell pool, these expanded A2-GLC tetramer–positive cells also had significantly longer telomeres than CD8+ T cells from normals (P < .016). It was of interest that the mean telomere length of CD4+ T cells from AIM patients was also longer than that of normals (P < .01), suggesting that this subset was involved in the cellular response in AIM and that mechanisms responsible for telomere maintenance were also engaged in this population.
Comparison of telomere length in CD4, CD8, and A2-GLC tetramer–positive cells in healthy controls and AIM patients.
Frozen PBMCs from 9 healthy controls or 12 AIM patients were stained with either CD4-biotin or CD8-biotin followed by strep-Cy5. A2-GLC-Cy5 tetramer staining was determined in 10 of the AIM patients. These cells were fixed with 2% paraformaldehyde and then processed for flow-FISH using the telomeric PNA-FITC probes as described above. Samples from patients and healthy controls were stained and analyzed on the same day. At least 50 000 live cells were acquired and gates were determined as described in Figure 1B. Values represent the MFI. TheP values represent the significance as assessed by the Mann-Whitney test.
Comparison of telomere length in CD4, CD8, and A2-GLC tetramer–positive cells in healthy controls and AIM patients.
Frozen PBMCs from 9 healthy controls or 12 AIM patients were stained with either CD4-biotin or CD8-biotin followed by strep-Cy5. A2-GLC-Cy5 tetramer staining was determined in 10 of the AIM patients. These cells were fixed with 2% paraformaldehyde and then processed for flow-FISH using the telomeric PNA-FITC probes as described above. Samples from patients and healthy controls were stained and analyzed on the same day. At least 50 000 live cells were acquired and gates were determined as described in Figure 1B. Values represent the MFI. TheP values represent the significance as assessed by the Mann-Whitney test.
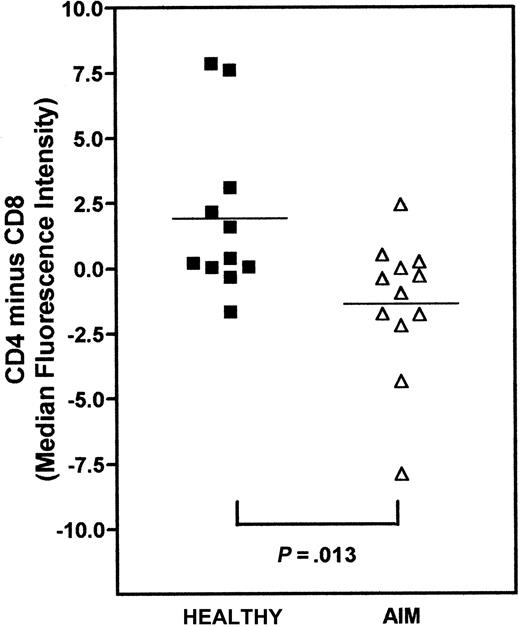
We next investigated the relative differences in telomere lengths between CD4+ and CD8+ populations within each individual AIM patient and in the controls (Figure5). We subtracted the median telomere length of CD8+ T cells from CD4+ T cells. Although values above zero reflect longer telomeres in CD4 compared to CD8 cells, values below zero indicate longer telomeres in the CD8 subset. We found that, on average, CD4+ T cells had longer telomeres than the CD8+ T cells in healthy controls, whereas in AIM patients, the CD8+ T cells had longer telomeres than the CD4+ cells (Figure 5). When the differences between CD4 and CD8 subsets in controls and AIM patients were compared, there was a significant difference between the 2 groups (P < .013; Mann-Whitney test). This indicates that despite the greater proliferative activity within the CD8 relative to the CD4 subset in AIM,1,12 the CD8+ cells had preserved their telomere length relative to the CD4+ cells in the same individuals. These results confirm our previous observations using Southern blot analysis for telomere length.12
Relative difference in telomere length between CD4+ and CD8+ within healthy controls and AIM patients.
PBMCs were stained with CD4-biotin or CD8-biotin followed by strep-Cy5, fixed, and then processed for flow-FISH using the telomeric PNA-FITC probes. Telomere length in these populations was determined by measuring the MFI of the telomere probe and the cells gated as described in Figure 1B. To assess the differences in telomere length between CD4 and CD8 lymphocytes within healthy controls and patients, MFI of telomere staining of the CD8+ T cells was subtracted from that of the CD4+ population. A positive value indicates that CD4 cells have longer telomeres compared to CD8 T cells and vice versa. Each patient sample was processed simultaneously with a healthy control sample. Squares represent healthy controls; triangles represent AIM patients. The horizontal lines show the mean difference in relative telomere length between CD4+ and CD8+ T cells for each group. The P value indicates the statistical significance as determined by the Mann-Whitney test.
Relative difference in telomere length between CD4+ and CD8+ within healthy controls and AIM patients.
PBMCs were stained with CD4-biotin or CD8-biotin followed by strep-Cy5, fixed, and then processed for flow-FISH using the telomeric PNA-FITC probes. Telomere length in these populations was determined by measuring the MFI of the telomere probe and the cells gated as described in Figure 1B. To assess the differences in telomere length between CD4 and CD8 lymphocytes within healthy controls and patients, MFI of telomere staining of the CD8+ T cells was subtracted from that of the CD4+ population. A positive value indicates that CD4 cells have longer telomeres compared to CD8 T cells and vice versa. Each patient sample was processed simultaneously with a healthy control sample. Squares represent healthy controls; triangles represent AIM patients. The horizontal lines show the mean difference in relative telomere length between CD4+ and CD8+ T cells for each group. The P value indicates the statistical significance as determined by the Mann-Whitney test.
Telomere length in EBV-specific CD8+ T cells in patients before and after resolution of AIM
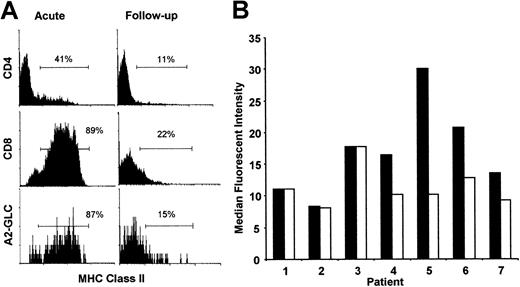
The absolute numbers of both total CD8+ and A2-GLC tetramer–positive CD8+ T cells decrease substantially after resolution of AIM, as described previously.3,10Although MHC class II is not expressed in nonactivated CD8+T cells,25 the majority of the CD8+ T-cell pool and A2-GLC tetramer–positive CD8+ T cells expressed high levels of class II during AIM suggesting intense activation (Figure 6A). In this analysis we gated on nonblastoid cells (gate RI in Figure 1) as described above. MHC class II expression was substantially reduced on total CD8+ and A2-GLC tetramer–positive T cells in the follow-up samples (Figure 6A). Nevertheless, the expression of MHC class II remained elevated on some tetramer-positive cells suggesting continued activation on a proportion of these cells after resolution of the acute infection. Although there also was increased MHC class II expression in CD4+ T cells during AIM, this level of activation was much lower than in the CD8+ T cells as reported previously.25
Changes in activation status and telomere length after resolution of AIM.
The activation status of CD4+, CD8+, and tetramer (A2-GLC)–positive lymphocyte subsets in acute AIM patients and 12 months after infection (A). Isolated PBMCs from these patients with acute AIM and from the same patients 12 months after infection were stained for CD8-RPE-Cy5 and A2-GLC-PE or CD4-RPE-Cy5 in combination with MHC class II–FITC. The cells were gated as described in Figure 1B, which excludes blast cells. Percentages represent cells found in the gates set according to isotype-matched control antibodies. The results are representative of MHC class II expression in 4 different patients studied during AIM and after resolution of the infection. Telomere length in tetramer–positive lymphocytes in 7 AIM patients and between 5 months and 14 years after follow-up were investigated by 2-color flow-FISH (B). Patients 1 and 2 were investigated 12 months after AIM, patient 3 was investigated after 8 months, patient 4 was investigated after 5 months, patient 5 was investigated after 15 months, and patients 6 and 7 were investigated 14 years after the infection. Isolated PBMCs were stained for A2-GLC-CY5 and telomeric PNA-FITC probe. The results are expressed as MFI for the FITC channel, obtained by viable cell gating and tetramer positivity. Acute patients, ▪; follow-ups, ■. For individual patients, each acute and follow-up sample was assessed at the same time.
Changes in activation status and telomere length after resolution of AIM.
The activation status of CD4+, CD8+, and tetramer (A2-GLC)–positive lymphocyte subsets in acute AIM patients and 12 months after infection (A). Isolated PBMCs from these patients with acute AIM and from the same patients 12 months after infection were stained for CD8-RPE-Cy5 and A2-GLC-PE or CD4-RPE-Cy5 in combination with MHC class II–FITC. The cells were gated as described in Figure 1B, which excludes blast cells. Percentages represent cells found in the gates set according to isotype-matched control antibodies. The results are representative of MHC class II expression in 4 different patients studied during AIM and after resolution of the infection. Telomere length in tetramer–positive lymphocytes in 7 AIM patients and between 5 months and 14 years after follow-up were investigated by 2-color flow-FISH (B). Patients 1 and 2 were investigated 12 months after AIM, patient 3 was investigated after 8 months, patient 4 was investigated after 5 months, patient 5 was investigated after 15 months, and patients 6 and 7 were investigated 14 years after the infection. Isolated PBMCs were stained for A2-GLC-CY5 and telomeric PNA-FITC probe. The results are expressed as MFI for the FITC channel, obtained by viable cell gating and tetramer positivity. Acute patients, ▪; follow-ups, ■. For individual patients, each acute and follow-up sample was assessed at the same time.
Using the 2-color flow-FISH technology we analyzed telomere length of tetramer-positive cells during and after resolution of AIM in the same patients. PBMCs from 7 AIM patients and the same individuals between 5 months and 14 years after the resolution of the infection were investigated (Figure 6B). The acute and follow-up samples were cryopreserved immediately after isolation, thawed, and analyzed simultaneously. We found that telomere lengths were identical in the acute and follow-up samples in 3 patients tested 8 to 12 months after AIM (patients 1-3). In patient 4, who was tested after 5 months, however, telomere shortening was observed. Patients who were tested at longer times after AIM, ranging from 15 months (patient 5) to 14 years (patients 6 and 7), all showed decreased telomere lengths in the A2-GLC tetramer–positive cells (Figure 6B). These results suggest that although telomere length is maintained in the EBV-specific cells during the acute phase of infection and up to 1 year after resolution in the majority of patients, telomere shortening occurs after longer periods of time in vivo. There may be individual variation in the capacity to maintain telomere shortening of EBV-specific cells after disease resolution (eg, patient 4) and we are currently investigating the impact of telomere length in EBV-specific CD8+ T cells and subsequent functional responses to the virus by post-AIM patients in vitro.
Telomerase activity in EBV-specific CD8+ T cells during acute infection and after follow-up
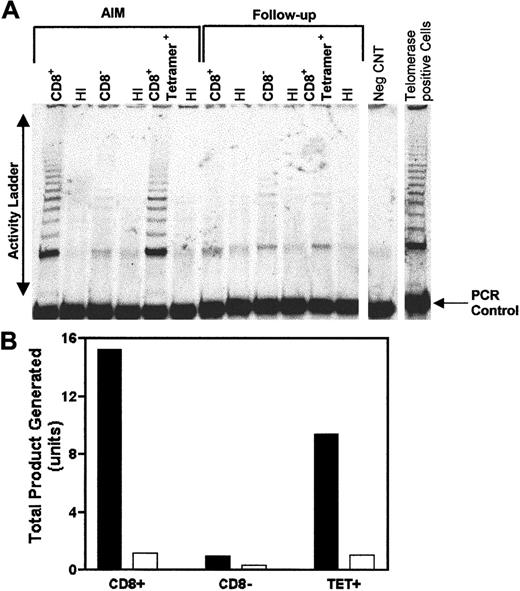
One crucial mechanism for the maintenance of telomere length involves the induction of the enzyme telomerase, which can extend telomeres in cells.17,26,27 We therefore investigated if the telomere maintenance that is seen in cells from AIM patients was related to their up-regulation of telomerase activity. We first isolated CD8+ T cells from AIM patients by negative selection. We next enriched for tetramer-binding cells within the CD8+ fraction by positive selection (see “Patients, materials, and methods”). The CD8+ T-cell, the tetramer-enriched, and the CD8-depleted cell fractions were all analyzed for telomerase activity (Figure7). Both the CD8+ and the tetramer-enriched fractions showed high telomerase activity during the acute phase of infection. Low but detectable levels of telomerase activity were found in the CD8-depleted subset, which confirms our previous observations that telomerase is activated in CD4+T cells in AIM.12
EBV-specific CD8+ cells express high telomerase activity during AIM.
CD8+ T cells were first isolated from a PBMC population from an AIM patient. B8-RAK tetramer–positive cells were then isolated from the CD8+ population using anti-PE MACS beads. Telomerase activity was determined using the TRAP assay autoradiography performed on CD8+, CD8−, and tetramer–enriched T-cell fractions from the patient during AIM and 12 months after follow-up (A). The TRAP-eze protocol, in which telomerase activity is amplified by PCR, gives rise to a ladder of products with 6-bp increments, starting at 50 nucleotides. Heat-inactivated (HI) cell extracts provided a telomerase negative control for each sample. The negative control (−CNT) contains the PCR mix without the cell extract. In addition, amplification of the internal control oligonucleotides together with the TS primer give rise to the internal PCR control (36 bp). Telomerase activity in acute and follow-up AIM patient activity was expressed as total product generated calculated according to manufacturer's instructions (B). Each unit of total product generated corresponds to the number of TS primers extended with at least 4 telomeric repeats by telomerase in the extract in a 10-minute incubation at 30°C. Telomerase activity in acute AIM patients, ▪; follow-up patient samples, ■. In a total of 4 patients studied, high telomerase activity was found in CD8+ T cells during AIM, which was substantially reduced in the follow-up samples taken after 12 months or longer after resolution of the acute infection.
EBV-specific CD8+ cells express high telomerase activity during AIM.
CD8+ T cells were first isolated from a PBMC population from an AIM patient. B8-RAK tetramer–positive cells were then isolated from the CD8+ population using anti-PE MACS beads. Telomerase activity was determined using the TRAP assay autoradiography performed on CD8+, CD8−, and tetramer–enriched T-cell fractions from the patient during AIM and 12 months after follow-up (A). The TRAP-eze protocol, in which telomerase activity is amplified by PCR, gives rise to a ladder of products with 6-bp increments, starting at 50 nucleotides. Heat-inactivated (HI) cell extracts provided a telomerase negative control for each sample. The negative control (−CNT) contains the PCR mix without the cell extract. In addition, amplification of the internal control oligonucleotides together with the TS primer give rise to the internal PCR control (36 bp). Telomerase activity in acute and follow-up AIM patient activity was expressed as total product generated calculated according to manufacturer's instructions (B). Each unit of total product generated corresponds to the number of TS primers extended with at least 4 telomeric repeats by telomerase in the extract in a 10-minute incubation at 30°C. Telomerase activity in acute AIM patients, ▪; follow-up patient samples, ■. In a total of 4 patients studied, high telomerase activity was found in CD8+ T cells during AIM, which was substantially reduced in the follow-up samples taken after 12 months or longer after resolution of the acute infection.
We next investigated if the telomerase activity in the CD8+ T cells from AIM patients was maintained after resolution of the infection. We therefore investigated telomerase activity in isolated CD8+ and tetramer-enriched cells from the same patient after resolution of AIM. We found that on follow-up a marked reduction of telomerase activity in both the CD8+and tetramer-positive cells compared to the same cells in AIM (Figure7). There is, however, low but detectable telomerase activity in both these subsets suggestive of a residual level of activation in these cells. In 5 patients studied to date, CD8+ T cells that were isolated during AIM all had high telomerase activity relative to CD8+ T cells from the same patient after follow-up (data not shown). This suggests that the ability to maintain telomeres of EBV-specific CD8+ T cells is not due to the ability to up-regulate telomerase during the acute phase of disease. However, because telomerase activity decreases after AIM, telomere maintenance or shortening of EBV-specific CD8+ T cells will depend on the capacity of these cells to up-regulate telomerase on subsequent challenge with virus after longer periods in vivo (Figure 6B). Longitudinal studies are now in progress to address this point.
Discussion
We have modified a method for detecting telomere length by flow cytometry to enable us to investigate the relative replicative senescence of EBV-specific CD8+ T cells in patients during AIM and after resolution of the infection. We show that despite the massive activation and proliferation of EBV-specific CD8+ T cells during AIM,2,3 these cells maintain their telomeres relative to the CD8+ T cells from normal individuals and also their own CD4+ T cells. However, our results suggest that the CD4+ T cells pool in these patients have also increased their telomeres suggesting that they have also been activated and have a role in the acute phase of this infection. We only analyzed cells within the nonblastoid gate, which excludes the possibility that any differences between the subsets analyzed arose from the increased FISH signal resulting from telomere replication in cycling cells.21 Although the cells from AIM patients that fell in this gate were not blasts, the majority expressed HLA-DR suggesting that they had been previously activated and were likely to be recently activated postmitotic cells rather than resting cells. The increased telomere length of CD8+ relative to CD4+ T cells in AIM patients detected by flow-FISH analysis confirms our previous results using DNA analysis by Southern blot.12The maintenance of telomere length of EBV-specific CD8+ T cells in AIM is associated with induction of telomerase activity and suggests that the CD8+ T cells that escape apoptosis during resolution of AIM enter the memory pool with relatively preserved replicative capacity.
Using the 2-color flow-FISH technology, we were able to analyze the relative changes of telomere length of the EBV-specific CD8+ T cells during the acute phase and in the same patients after the infection resolved. We found that in 3 of 4 individuals, telomere length remained virtually identical up to 1 year after disease resolution. In patients who were studied after longer periods after AIM, there was consistent telomere shortening. The ability to maintain telomere length is not related to the capacity of different individuals to up-regulate telomerase activity in the acute phase of disease because all the patients we have studied during AIM have up-regulated telomerase activity in the CD8+ T-cell population. However, because EBV causes a persistent infection1 there will be episodes of viral replication followed by some reactivation and expansion of specific CD8+ T cells. It is likely, therefore, that the ability of EBV-specific CD8+ T cells from different individuals to up-regulate telomerase activity on subsequent challenge and also the extent of reactivation of the cells by virus over time determines residual telomere length. Previous studies have shown that although initial antigenic challenge of T cells leads to highly increased telomerase activity, subsequent challenges induce progressively less enzymic activity.27 The observation that EBV epitope-specific primary responses may be dominated by just a few highly expanded clonotypes and that with persistent viral challenge, these dominant T-cell clonotypes may be lost and replaced by others in memory provides indirect support for the loss of reactive clones by telomere shortening.28 The 2-color flow-FISH technology described here will enable further detailed investigation of the heterogeneity of telomere length changes in patients at multiple time points after acute infection. This information is clearly crucial for the clarification of the role of replicative senescence in the regulation of CD8+ T-cell memory.
The identification of signals that may regulate telomerase activity is undoubtedly of importance because these may be manipulated to enhance immunity after vaccination, especially in the aged. It has been shown previously that both T-cell receptor and costimulation via CD28 are required for optimal telomerase induction in antigen-specific T cells.27 However, CD28 expression by CD8+ T cells in humans is decreased considerably after activation both in vitro and in vivo and most of the CD8+ T cells that are generated during acute viral infection in humans and simians are CD28−.16,29-31 Indeed, a substantial proportion of EBV-specific CD8+ T cells in patients who have recovered from AIM are also CD28−.10This would suggest that the activated and expanded CD8+ T cells in AIM patients may have constraints on their subsequent ability to up-regulate telomerase. This may be one reason why telomere lengths in CD8+CD28+ T cells are longer than those in the CD8+CD28− T cells in humans.32 It is also possible that other signals apart from CD28 may regulate telomerase activity and these may also have a role in the maintenance of replicative capacity within the human memory CD8+ T-cell pool. It is of interest that TCR-activated CD8+ T cells can be also costimulated by the CD11a/CD18 pathway30 and this may be one alternative way to up-regulate telomerase activity in these cells.
Although there are many similarities in mechanisms that may regulate the CD8+ T-cell pool in humans and mice, there are also some striking differences. Similarities include the ability of cytokines such as those that signal via the γ-chain of the interleukin-2 receptor (IL-2R) and the type I interferons (IFNs) to prevent apoptosis of activated CD8+ T cells after acute antigenic challenge and thus allow them to enter the memory pool.33-38 These cytokines also appear to prevent apoptosis by similar mechanisms in humans and mice, and although the IL-2R γ-chain signaling cytokines induce Bcl-2 and Bcl-xLexpression relative to Bax in the rescued cells,4,35,39the type I IFNs only induce Bcl-xL but not Bcl-2.34,36,38,40 There are, however, 2 main differences in the regulation of CD8+ T cells in humans and mice. First, although telomere lengths in humans are in the 12- to 16-kb range, they are much longer (90-100 kb) in mice.41 This suggests that telomere shortening after activation is likely to have more impact on the regulation of CD8+ T-cell memory in humans, especially when the much longer life span of humans relative to mice is considered. The second difference is the loss of CD28 expression by the CD8 pool as described above, which may not occur in mice.42 The loss of this molecule may affect the replicative senescence as well as the reactivation of human CD8+ memory T cells.
These observations have important implications for the maintenance of CD8 memory in different species. In mice, memory CD8+ T cells have been shown to undergo slow but continuous cycling, induced by cytokines such as IL-15 and this may enable the maintenance of the memory pool.33,43 Although this has not yet been studied in detail, the maintenance of CD8 memory in humans by continuous bystander proliferation by cytokines would potentially lead to excessive telomere shortening and loss of the cells through replicative senescence. The fact that we can see telomere shortening in patients after long-term follow-up and that certain clones of EBV-specific T-cell activities are lost with time,28provides indirect support for this possibility. Also, accelerated shortening of telomeres in leukocytes in human bone marrow transplant recipients may be due to extensive expansion of these cells to repopulate the host.44 Other additional mechanisms that enable maintenance of memory CD8+ T cells without inducing proliferation may therefore have to operate in humans. One such mechanism may be the maintenance of cells by type I IFNs, which prevent apoptosis of activated T cells without inducing proliferation.45 This would enable survival without overt proliferative activity. This would not preclude the possibility of episodes of activation and proliferation in memory CD8+ T cells, which is induced by antigen or cytokines, which are then interspersed with periods of proliferative quiescence induced by the type I IFNs.46
In conclusion, the use of MHC class I tetramer technology together with the ability to analyze changes in telomere length in these cells by 2-color flow-FISH now permits the study ofreplicative senescence in memory CD8+ T cells in humans. This will allow the investigation of changes within cells that are reactive to different epitopes of viruses in response to vaccination and will clarify if replicative senescence may be the reason why immunity to some vaccines offer lifelong protection, whereas others are only effective for a limited period. This technology will also enable us to investigate antigen-specific CD8+ T-cell responses in other viral infections such human immunodeficiency virus, and will enable us to determine reasons for the compromised antigen-specific T-cell responses in the elderly.47
Acknowledgments
We would like to thank Dr Paul Travers and Professor P. C. L. Beverley for discussions. We would also like to thank Dr Caroline Sabin for advice on statistical analysis.
Supported by grant 77/SAG1002 from the BBSRC SAGE initiative to F.J.P. and grant BD/9254/96 from Programa PRAXIS XXI to M.V.D.S.
F.J.P., M.V.D.S., and N.A. contributed equally to this work.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Arne N. Akbar, Department of Clinical Immunology, Royal Free and University College Medical School, Pond St, Hampstead, London NW3 2QG, United Kingdom; e-mail: akbar@rfhsm.ac.uk.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal