Adhesive interactions with stromal cells and the extracellular matrix are essential for the differentiation and migration of hematopoietic progenitors. In the erythrocytic lineage, a number of adhesion molecules are expressed in the developing erythrocytes and are thought to play a role in the homing and maturation of erythrocytic progenitors. However, many of these molecules are lost during the final developmental stages leading to mature erythrocytes. One of the adhesion molecules that remains expressed in mature, circulating erythrocytes is CD147. This study shows that blockade of this molecule on the cell surface by treatment with F(ab′)2 fragments of anti-CD147 monoclonal antibody disrupts the circulation of erythrocytes, leading to their selective trapping in the spleen. Consequently, mice develop an anemia, and de novo, erythropoietin-mediated erythropoiesis in the spleen. In contrast, these changes were not seen in mice similarly treated with another antierythrocyte monoclonal antibody with a different specificity. These results suggest that the CD147 expressed on erythrocytes likely plays a critical role in the recirculation of mature erythrocytes from the spleen into the general circulation.
Introduction
The development and maturation of hematopoietic cells are highly dependent on their interactions with stromal cells and extracellular matrix, and adhesion molecules play an important role in these interactions.1,2 Several adhesion molecule/ligand pairs, such as the very late antigen-4/vascular cell adhesion molecule (VLA-4/VCAM), lymphocyte function-associated 1/intercellular adhesion molecule 1 (LFA-1/ICAM-1), and VLA-5/fibronectin, have been implicated in cell-to-cell contact between hematopoietic progenitors and stroma.3-7 The significance of such interactions is illustrated by the central role played by VLA-4 in the differentiation and homing of hematopoietic progenitors in vivo.8 9
Erythroid progenitors express a wide range of surface molecules, several of which, such as ICAM-1, -2, -3, and -4, CD44, LFA-3, basal cell adhesion molecule/Lutheran protein (B-CAM/LU), and VLA-4, have adhesive properties.10 However, expression of some of these molecules is lost from the cell surface during the final stage of erythroid development leading to mature erythrocytes.10 Interestingly, in certain pathologic conditions adhesion molecules are de novo expressed or up-regulated on mature erythrocytes. For instance, in sickle cell disease the adhesion molecule B-CAM/LU is up-regulated on erythrocytes,11whereas advanced glycation end-products are found on diabetic erythrocytes, leading to increased erythrocyte adhesion to vascular endothelium and migration defects such as vascular occlusion and splenomegaly.12
CD147 is a member of the immunoglobulin superfamily and is a highly glycosylated cell surface protein.13 CD147 is considered an adhesion molecule and has been shown to induce matrix metalloprotease expression by neighboring epithelial cells.14 It is expressed in various tissues, including brain, leukocytes, endothelial cells, and most tumor cell lines. In addition, CD147 is expressed on erythrocyte lineage cells throughout erythroid development, including in mature erythrocytes,8and has been shown to be the carrier molecule for the blood group antigen Oka.15 To investigate whether this expression has a functional significance, we determined the effect of the in vivo treatment with a CD147-specific monoclonal antibody (mAb) on the development and trafficking of erythrocytes in mice. We show here that CD147 blockade disrupts the migration of erythrocytes out of the spleen, leading to transient splenomegaly and anemia, and consequent de novo erythropoiesis in this organ.
Materials and methods
Mice and treatment
Four- to 8-week-old DBA/2 mice were obtained from Iffa Credo (Lyon, France). F(ab′)2 fragments of 2 rat IgG mAbs, RL7316 and Ter11917 (a kind gift of Dr Tatsuo Kina) were prepared using the ImmunoPure F(ab′)2preparation kit (Pierce, Rockford, IL) and were injected intraperitoneally at 100 μg/mouse. Where indicated, 100 μg rabbit antihuman erythropoietin (EPO) or normal rabbit serum (R&D Systems, Minneapolis, MN) were administered intraperitoneally.
Antibodies and flow cytometry
The following commercial mAbs were used: fluorescein isothiocyanate (FITC)-labeled anti-VLA-4, phycoerythrin (PE)–labeled anti-Ter119, Cy-chrome-labeled anti-CD8, anti-B220 (BD Pharmingen, San Diego, CA) and anti-CD4 (Caltag, Burlingame, CA). FITC-labeled anti-GRI and anti-CD11b, and PE-labeled anti-γδ T-cell receptor (TCR) were purchased from BD Pharmingen. Stained cells were analyzed on a FACScan (Becton Dickinson, San Jose, CA)
Cell-cycle analysis
Sorting of lymphocytes (CD4+, CD8+, or B220+) and erythroblasts (Ter119+VLA-4−) was performed on FACS Vantage (Becton Dickinson) and cell cycle performed as described.18 Briefly, cells were fixed with 70% ethanol for at least 30 minutes and the DNA was partially denatured in 3 N HCl, then neutralized with 0.1 M Na2B4O7. The cells were subsequently resuspended in 100 μL phosphate-buffered saline (PBS) containing 2.5% fetal calf serum (FCS) and added to 200 μL PBS/FCS containing 3 μg/mL propidium iodide, 50 μg/mL RNAse A (Sigma, St Louis, MO), 50 mM Tris base, 50 mM NaCl, and 5 mM EDTA, pH 7.5. Cells were then incubated 5 minutes at 37°C and analyzed on a FACScan equipped with a doublet discrimination module.
Treatment of erythrocytes
The DBA/2 erythrocytes were untreated or treated with RL73 F(ab′)2 or Ter119 F(ab′)2, 1 hour at room temperature, and washed 3 times with PBS 1 ×. Erythrocytes were then injected intravenously into DBA/2 mice, and spleen weight and hematocrit were measured.
Hematocrit measurement
Peripheral blood was drawn from the retro-orbital sinus and aspirated into a capillary tube, the bottom of which was then sealed with Play Dough. After centrifugation for 30 minutes at 900 rpm, the height of packed cells was measured and expressed as a percentage of total height.
Microscopy
The DBA/2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 or Ter119 F(ab′)2, or left untreated, and killed on day 3. Spleens, livers, kidneys, and lungs were obtained, processed for histologic examination, and stained with hematoxylin and eosin.
Results
Selective erythrocyte trapping in spleen of mice treated with anti-CD147 mAb
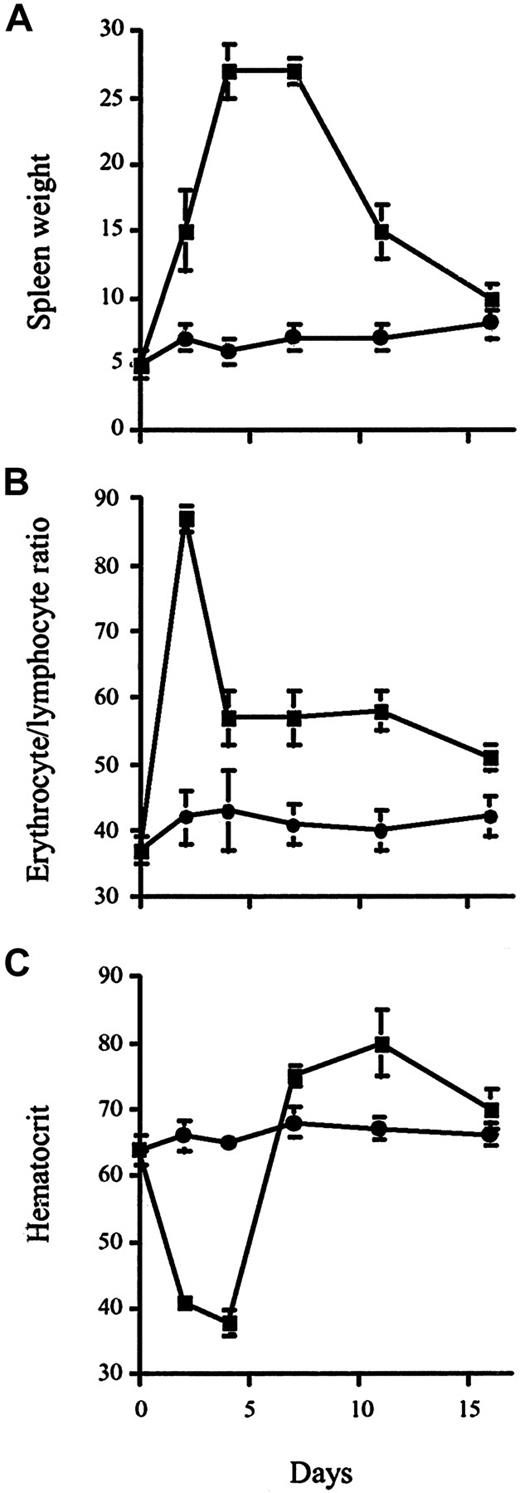
We have recently identified the molecule recognized by mAb RL73 as being CD147 (manuscript submitted). Because CD147 is expressed on erythroblasts and mature erythrocytes10 (and our own unpublished observations, February 2000), the effect of an in vivo administration of RL73 on the generation or trafficking of erythrocytes was assessed. As a control, we used another mAb (Ter119), which reacts with erythroid cells at differentiation stages from early proerythroblast to mature erythrocyte, and recognizes a 52-kd protein associated with glycophorin A.17 To avoid any possible effect of interaction of IgG mAb with Fc receptor (FcR)-bearing cells such as macrophages, F(ab′)2 fragments of RL73 and Ter119 were used in this study. Both F(ab′)2 preparations bind erythrocytes to the same extent (data not shown). Figure1A shows a transient increase of spleen weight, peaking between 4 to 7 days and persisting for 11 days after intraperitoneal injection of 100 μg RL73 F(ab′)2, whereas this was not seen in mice similarly treated with Ter119 F(ab′)2. In a shorter time course, we observed enlarged spleens as early as 6 hours after injection (data not shown).
Increase in erythrocyte number in spleen of anti-CD147–treated mice coincides with decrease in hematocrit.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 (▪)or Ter119 F(ab′)2 (●) and mice were killed on days 2, 4, 7, 11, and 16. Spleen cells were weighed (A), and analyzed by FACS (B). The erythrocyte/lymphocyte ratio reflects the percentage of Ter119+VLA-4− erythrocytes divided by the combined percentage of CD4+, CD8+, and B220+ cells. Hematocrit (C) was measured by standard methods at the indicated time points following RL73 F(ab′)2 or Ter119 F(ab′)2 treatment.
Increase in erythrocyte number in spleen of anti-CD147–treated mice coincides with decrease in hematocrit.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 (▪)or Ter119 F(ab′)2 (●) and mice were killed on days 2, 4, 7, 11, and 16. Spleen cells were weighed (A), and analyzed by FACS (B). The erythrocyte/lymphocyte ratio reflects the percentage of Ter119+VLA-4− erythrocytes divided by the combined percentage of CD4+, CD8+, and B220+ cells. Hematocrit (C) was measured by standard methods at the indicated time points following RL73 F(ab′)2 or Ter119 F(ab′)2 treatment.
To identify the cell type(s) that increase(s) in number after administration of RL73 F(ab′)2 anti-CD147, multiparameter fluorescence-activated cell sorter (FACS) analysis was performed on spleen cells. We observed that there was a marked increase (86% ± 3% on day 2) in low forward scatter (FSClow) cells in RL73-injected mice, as compared with Ter119-injected mice (28% ± 1%), and that the accumulating cells were Ter119+ (an erythrocytic lineage marker) and VLA-4−(α4β1-integrin, expressed on erythrocyte precursors but not on mature erythrocytes) (not shown), indicating they are mature erythrocytes. Dead lymphocytes, which normally are also FSClow, were excluded by propidium iodide. Calculation of the erythrocyte/lymphocyte ratio in the spleen revealed a dramatic increase in the percentage of erythrocytes 2 days after injection with RL73 F(ab′)2, which persisted as late as 16 days following the mAb treatment (Figure 1B). Because the number of lymphocytes in the spleen was unaffected by RL73 F(ab′)2anti-CD147 treatment (not shown), this indicates that the absolute number of erythrocytes in the spleen was markedly augmented (5.4 × 108 versus 5.1 × 107 on day 2). This increase in erythrocytes was accompanied by a highly significant drop in hematocrit (Figure 1C). These data suggest a retention of erythrocytes in the spleen, resulting in depletion of this population from the circulating blood pool. The specificity of the effect of RL73 F(ab′)2 was supported by the lack of effect of another erythrocyte-specific antibody, Ter119 F(ab′)2, on the accumulation of erythrocytes in spleen (Figure 1).
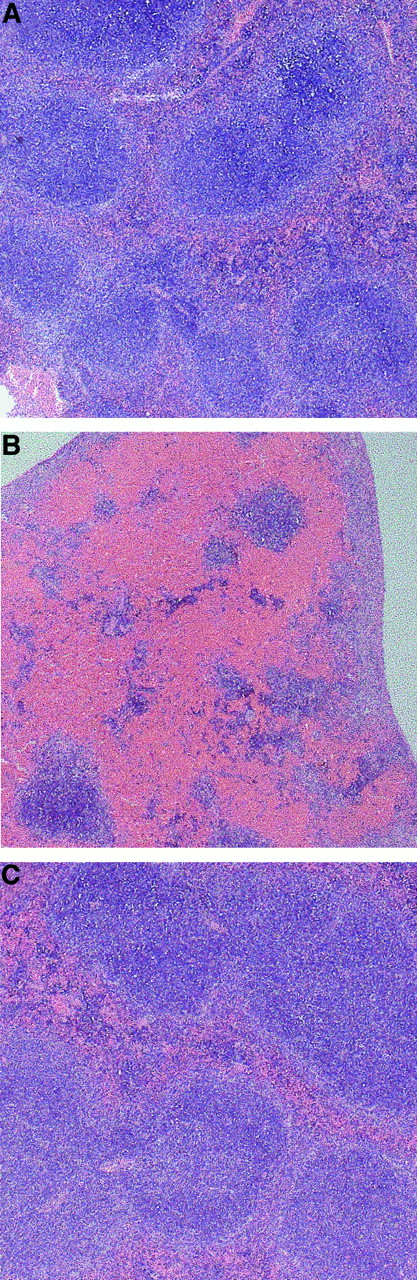
Erythrocyte accumulation in the spleen was further confirmed by light microscopic analysis of spleen sections. As shown in Figure2B, a massive accumulation of erythrocytes occurred in the red pulp of spleens from RL73 F(ab′)2-treated mice, whereas spleens from Ter119 F(ab′)2-treated mice (Figure 2C) were indistinguishable from those of unmanipulated mice (Figure 2A). Strikingly, no erythrocyte accumulation was observed in other major organs studied, such as liver, kidney, or lung from any of the mice analyzed (data not shown), suggesting selectivity of erythrocyte trapping in the spleen following CD147 blockade.
Erythrocytes accumulate in the red pulp of spleens from anti-CD147–treated mice.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 (B) or Ter119 F(ab′)2 (C), or left untreated (A) and killed on day 3. Spleens were removed and fixed in 10% formaldehyde. Microtome sections were then stained with hematoxylin and eosin and analyzed by light microscopy (original magnification × 25).
Erythrocytes accumulate in the red pulp of spleens from anti-CD147–treated mice.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 (B) or Ter119 F(ab′)2 (C), or left untreated (A) and killed on day 3. Spleens were removed and fixed in 10% formaldehyde. Microtome sections were then stained with hematoxylin and eosin and analyzed by light microscopy (original magnification × 25).
To investigate whether anti-CD147 has a direct effect on erythrocytes or acts through other cells (endothelial cells, for instance, which are CD147+),19 DBA/2 mice were bled, erythrocytes were untreated or treated with RL73 F(ab′)2 or Ter119 F(ab′)2, washed extensively to remove free Ab, then reinjected intravenously into syngeneic recipient mice. One day later, spleen weight and hematocrit were measured. As expected, mice that were perfused with untreated or Ter119 F(ab′)2-treated erythrocytes exhibited an increased hematocrit (P < .001) but no splenomegaly (Table 1). By contrast, spleen weight was significantly increased in mice that received RL73 F(ab′)2-treated erythrocytes (P < .005), with only a slight increase in their hematocrit values (P < .05). Taken together, these data strongly suggested that blockade of CD147 on erythrocytes was directly responsible for their trapping in the spleen.
Spleen weight and hematocrit in syngeneic recipient mice
| . | Control . | Hypertransfused . | ||
|---|---|---|---|---|
| Untreated . | RL73 F(ab′)2 . | Ter119 F(ab′)2 . | ||
| Spleen weight (g) | 0.05 ± 0.01 | 0.07 ± 0.01 | 0.12 ± 0.01 | 0.05 ± 0.01 |
| Hematocrit (%) | 62 ± 2 | 84 ± 1 | 70 ± 3 | 85 ± 3 |
| . | Control . | Hypertransfused . | ||
|---|---|---|---|---|
| Untreated . | RL73 F(ab′)2 . | Ter119 F(ab′)2 . | ||
| Spleen weight (g) | 0.05 ± 0.01 | 0.07 ± 0.01 | 0.12 ± 0.01 | 0.05 ± 0.01 |
| Hematocrit (%) | 62 ± 2 | 84 ± 1 | 70 ± 3 | 85 ± 3 |
Erythrocytes from peripheral blood were untreated or treated with RL73 F(ab′)2 or Ter119 F(ab′)2, then reinjected intravenously into mice. One day later, spleen weight and hematocrit were measured. Control, unmanipulated mice.
Induction of erythropoiesis in spleen of mice treated with anti-CD147 mAb
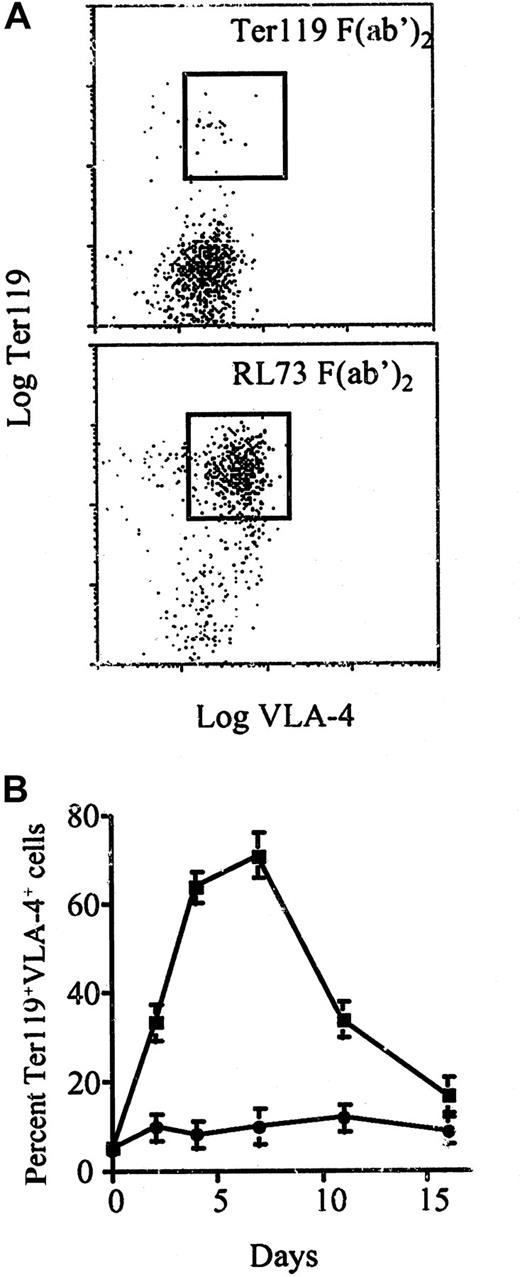
Two days after injection of mice with RL73 F(ab′)2, a FSChighCD4−CD8−B220−population started appearing in the spleen (not shown). These cells were also negative for markers of granulocytes, monocytes, and γδ T cells (not shown). By contrast, virtually all nonlymphocyte FSChigh spleen cells stained strongly with Ter119 and were VLA-4+ (Figure 3A). This phenotype, coupled with their large size, strongly suggested that these cells were erythroblasts. Histologic analysis further confirmed the presence of multiple erythroblast-containing erythropoietic foci in the spleen of these mice (data not shown). The percentage of FSChighTer119+VLA-4+ cells increased sharply starting 48 hours after injection, reaching peak levels at day 7, then returning to normal levels by day 16 (Figure 3B). An increase in FSChighTer119+VLA-4+cells was also observed in bone marrow, albeit with slightly delayed kinetics (not shown). The observation that these cells appeared in the spleen earlier than in the bone marrow of RL73-treated mice argues against the possibility that their increased numbers in the spleen is only due to import from the bone marrow.
De novo erythropoiesis occurs in the spleen of anti-CD147–treated mice.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 or Ter119 F(ab′)2 and killed at the indicated time points. (A) Spleen cells were stained for expression of Ter119 and VLA-4 and analyzed by FACS on day 3. (B) Kinetics of the increase in percentage of Ter119+, VLA-4+ cells in spleen of mice treated with 100 μg RL73 F(ab′)2 (▪) or Ter119 F(ab′)2 (●).
De novo erythropoiesis occurs in the spleen of anti-CD147–treated mice.
DBA2 mice were injected intraperitoneally with 100 μg RL73 F(ab′)2 or Ter119 F(ab′)2 and killed at the indicated time points. (A) Spleen cells were stained for expression of Ter119 and VLA-4 and analyzed by FACS on day 3. (B) Kinetics of the increase in percentage of Ter119+, VLA-4+ cells in spleen of mice treated with 100 μg RL73 F(ab′)2 (▪) or Ter119 F(ab′)2 (●).
If FSChighTer119+VLA-4+ cells are indeed erythroblasts, they should be cycling. Cell-cycle analysis was therefore performed on these cells sorted from spleen 4 days after the RL73 F(ab′)2 treatment. As expected, a majority of Ter119+VLA-4+ cells were cycling (> 40% in S phase and G2/M), further confirming their identity as erythroblasts. These cycling cells most likely are responsible for the amplification of the mature erythrocyte pool reflected by the increase above baseline in hematocrit observed at days 7 to 16 (Figure 1C).
Induction of erythropoiesis in spleen of phlebotomized mice
Collectively, these results indicate that de novo erythropoiesis occurred in the spleen following anti-CD147–mediated erythrocyte trapping in this organ. At least 2 scenarios could be invoked to explain this sequence of events. First, because CD147 is expressed on both mature erythrocytes and their precursors, it is possible that administration of RL73 F(ab′)2 masks CD147 on mature erythrocytes, leading to their sequestration in the spleen, and simultaneously delivers a signal to erythrocytic precursors through ligation of CD147, leading to their expansion. In this case, erythrocyte trapping and erythropoiesis in the spleen would be unrelated events. Alternatively, anti-CD147 may only interfere with the migration of mature erythrocytes from the spleen. This, in turn, would decrease the number of circulating erythrocytes, leading to a state of hypoxia. In this context, the de novo generation of erythrocytes from precursors is a compensatory feedback mechanism due to the lack of oxygen available to the tissues. To test this hypothesis, we removed 400 μL peripheral blood by phlebotomy and looked for the appearance of Ter119+VLA-4+ cells in the spleen 3 days later. This treatment led to a significant increase in the percentage of these cells (17% ± 3%) as compared with the control mice (3% ± 1%). Moreover, we observed that a simultaneous treatment with neutralizing anti-EPO antibodies completely inhibited the expansion of Ter119+VLA-4+ erythroblasts in the spleen from anti-RL73-injected mice (not shown), which is consistent with the demonstration that such a feedback erythropoiesis was mediated almost exclusively by EPO.20
Discussion
In the present study, we have demonstrated that the injection of RL73 anti-CD147 mAb, but not Ter119 mAb having a different specificity, induced a selective accumulation of circulating erythrocytes in red pulp of spleen. Because the possible consequences of complement activation and interaction with FcR-expressing cells were excluded because of the use of F(ab′)2 fragments of the mAb, the sequestration of erythrocytes in spleen is likely to be a result of the masking of CD147 on the surface of circulating erythrocytes, thereby blocking their migration out of the spleen. Thus, our data suggest that the CD147 molecule expressed on erythrocytes acts as an adhesion molecule, which apparently plays a critical role in the recirculation of mature erythrocytes from the spleen into the general circulation.
It has been previously reported that in vivo injection of antierythrocyte autoantibodies derived from autoimmune NZB mice into nonautoimmune mice resulted in anemia that was due to either sequestration of agglutinated erythrocytes in spleen and liver or Fc receptor-mediated erythrophagocytosis by Kupffer cells in liver.21 In these experiments, it was clearly demonstrated that a massive agglutination and sequestration of erythrocytes was observed not only in spleen but also in liver, and that this occurred only with antierythrocyte mAb that failed to induce significant FcR-mediated erythrophagocytosis in vivo.21-24 Because we used F(ab′)2 fragments of RL73 anti-CD147, one may argue that the observed massive accumulation of erythrocytes in spleen from RL73 anti-CD147–treated mice could be a result of the lack of efficient elimination of FcR-mediated erythrophagocytosis by Kupffer cells. However, this possibility is excluded because we did not see any sign of agglutinated erythrocytes in hepatic sinusoids in RL73-injected mice, unlike mice injected with agglutinating antierythrocyte monoclonal autoantibodies, which failed to interact efficiently with FcR.21-24 In addition, our results rule out the possible agglutination of erythrocytes resulting from the binding of highly opsonized erythrocytes through FcR or complement receptors on splenic macrophages.
Instead, our data suggest that masking CD147 on the surface of circulating erythrocytes is likely to be responsible for the trapping of these cells in the spleen. This was further supported by the complete lack of erythrocyte sequestration in spleen from mice treated with F(ab′)2 fragments of Ter119 mAb with a different specificity.17 CD147 has been described as a potential adhesion molecule,25 which binds to endothelial cells and fibroblasts.19 The adhesion molecule α4β1-integrin (VLA-4) has been previously shown to play a major role in the trafficking of hematopoietic precursors and their homing to the spleen and liver.9,26CD147 is expressed (presumably through its unique transmembrane domain27) in association with α3β1- and α6β1-integrins on the surface of a human fibrosarcoma cell line.28 Although mature erythrocytes lack expression of integrin-β1,29 it remains possible that CD147 plays a role in erythrocyte trafficking via an associated adhesion molecule yet to be identified. However, in the absence of evidence for such an association, it is not unreasonable to propose that CD147 on erythrocytes interacts with a putative ligand on endothelial cells and that this receptor/ligand interaction mediates the peripheralization of erythrocytes.
In summary, we show here that masking of CD147 on the surface of erythrocytes leads to their selective trapping in the spleen, and induces an anemia, and consequently, de novo EPO-mediated erythropoiesis in this organ and in the bone marrow. Apparently, the interaction of CD147 on erythrocytes and its potential ligand expressed on splenic vasculature is likely to be critical in the recirculation of mature erythrocytes from spleen into the general circulation. We postulate that the role of CD147 in erythrocyte circulation may be in its capacity as an inducer of matrix-degrading metalloproteases, thus actively participating in the exit of circulating erythrocytes from the spleen. Indeed, it was shown that CD147 increases the expression of interstitial collagenase, stromelysin, and gelatinase in stromal fibroblasts.14 However, other explanations cannot be ruled out. For instance, CD147, like CD47, may be part of the recognition of erythrocytes as self,30 and that blockade of this molecule may prevent erythrocytes from escaping the splenic reticuloendothelial system. Alternatively, it is possible that ligation of CD147 alters erythrocyte deformability, leading to their mechanical trapping in the spleen. A further possibility is that ligation of CD147 may alter erythrocyte adhesion through an intracellular signaling mechanism. The identification of the ligand(s) for CD147 and the cell type expressing it should help elucidate mechanisms involved in CD147-mediated erythrocyte trafficking.
S.I. is supported by a grant from the Swiss National Foundation for Scientific Research.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Isabelle Coste, Centre d'Immunologie Pierre Fabre 5, Avenue Napoleon III, 74160 Saint-Julien en Genevois, France; e-mail:isabelle.invernizzi@pierre-fabre.com.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal