Abstract
A variable degree of humoral immunodeficiency is a common feature in patients with B-cell chronic lymphocytic leukemia (B-CLL). The aim of this study was to explore the possibility that B-CLL cells play a direct role in this phenomenon. To this end, patients' bone marrow (BM) immunoglobulin (Ig)-secreting cells were cocultured with autologous purified B-CLL cells. The results show that tumoral cells inhibited the spontaneous IgG secretion by BM plasma cells, and this effect increased after PMA-induction of B-CLL cells. This inhibitory process was proportional to the number of B-CLL cells added and depended on cellular contact. Adhesion molecules did not appear to be involved in the cellular interaction, because the inclusion of blocking antibody to a variety of these proteins did not reverse the inhibitory phenomenon. However, the addition of monoclonal antibody that blocked the function of either CD95 or CD95L clearly reversed B-CLL cell inhibition on autologous BM plasma cells. These latter cells were shown to express CD95, and B-CLL cells contained detectable quantities of CD95L at the level of messenger RNA and protein. Annexin V–binding experiments revealed increased apoptosis of BM Ig-secreting cells when cocultured with autologous B-CLL cells. Finally, this inhibitory phenomenon might be operative in vivo because (a) there was a good correlation between the intensity of the inhibitory effect in vitro and the serum IgG level exhibited by every patient and (b) B-CLL cells also inhibited in vivo antigen-induced IgG-tetanus toxoid–secreting cells obtained from normal immunized subjects. Collectively, these data suggest that B-CLL cells inhibit autologous CD95-bearing Ig-secreting cells by the interaction with CD95L present on B-CLL cells and, hence, contribute to the state of humoral immunodeficiency that occurs in these patients.
Introduction
B-cell chronic lymphocytic leukemia (B-CLL) is the most frequent leukemia in the adult life of humans in western countries.1 This disease is caused by the expansion of monoclonal, slow-dividing CD5+ B lymphocytes.2These cells appear to be the tumoral counterpart of normal CD5+ B lymphocytes, a B-cell subset that predominates during fetal life and childhood, although only representing a minor population in adults.3 The physiologic role of normal CD5+ B lymphocytes remains uncertain, although evidence, mostly obtained in mice, implicates this cell subset in the production of the so-called polyreactive natural antibodies, which appear to recognize with low affinity widely expressed bacterial antigens as well as certain autoantigens.4 Most malignant B-CLL lymphocytes are arrested at the G0/G1 phase of the cell cycle.5 Therefore, the gradual accumulation of B-CLL cells in the patients' blood and organs cannot be explained by cell proliferation and seems to be the result of the prolonged survival shown by these cells, a process that has been connected with an increased capacity to resist normal apoptotic signals.6 7
A relevant clinical feature of CLL is the occurrence of a variable degree of humoral immunodeficiency in up to 70% of these patients, as evidenced by the appearance of hypogammaglobulinemia,8diminished humoral response to antigen immunization,9 and frequent infections, mainly by encapsulated bacteria.10Serum immunoglobulin (Ig)G, IgA, and IgM levels are similarly affected, and their decrease appears to occur in parallel with increased disease severity.11 The cause of this defect remains controversial, and excessive T-cell suppression12 as well as dilution of antibody-secreting cells by the tumoral cells13 have been proposed as possible explanations. It is well established that, in the human adult life, bone marrow (BM) becomes a major reservoir of terminally differentiated B cells (plasma cells).14,15 These cells express large quantities of CD38 molecules on their surface (CD38h)16,17 and can be functionally detected as cells capable of spontaneous and high-rate Ig secretion in vitro.18 19 The aim of the present study was to examine a possible direct role of B-CLL cells in the patients' capacity for Ig production. To this end, the effect of B-CLL cells on the Ig secretion by autologous BM plasma cells was tested. The results show that the tumoral cells inhibit in vitro production by autologous BM Ig-secreting cells by a mechanism dependent on cellular contact. Further studies revealed that the expression of CD95L by B-CLL cells was involved in the inhibition of CD95+ BM plasma cells present in these patients and that this molecular interaction induced the apoptosis of the latter cells. These findings suggest that B-CLL cells might have a direct inhibitory role in the patients' humoral response in vivo.
Patients, materials, and methods
Patients and samples
Thirty-six consecutive patients diagnosed with B-CLL were included in the study. In all the cases, the monoclonal B-cell expansion was CD5+. There were 25 male and 11 female patients, with ages ranging from 41 to 73 years (mean 59.2 years). B-CLL was diagnosed in all cases according to international CLL workshop criteria and staged according to the Rai system.20 At the time of the study, 2 patients had stage 0, 6 patients stage I, seventeen patients stage II, 9 patients stage III, and 2 patients stage IV disease. None of the patients had received chemotherapy for at least 1 month before the study. Patient blood samples were obtained by venopuncture, and BM specimens were obtained by iliac crest puncture. Healthy BM samples of 8 individuals were included in the study and were obtained as previously reported.19 Blood samples were also obtained from healthy volunteers immunized with tetanus toxoid (tet) 6 days earlier, as detailed elsewhere.21 All patients and controls were informed about the objectives and methods of the study and gave their consent.
Materials
Phorbol myristate acetate (PMA), ionomycin, actynomycin D, and cycloheximide were purchased from Sigma (St Louis, MO). Unconjugated and peroxidase-conjugated goat antihuman IgG, IgA, and IgM used for determining human Ig by enzyme-linked immunosorbent assay (ELISA) were provided by TAGO Immunologicals (Camarillo, CA). Highly purified tet used for IgG-tet ELISA testing was a generous gift from Llorente Laboratory (Madrid, Spain). Magnetic beads conjugated to goat antimouse IgG and the magnetic particle concentrator (MPC-6) were provided by Dynall (Oslo, Norway). Purified monoclonal antibodies (mAbs) used in immunomagnetic depletion (anti-CD5, -CD3, -CD56, and -CD14) and in flow cytometry (anti-CD19–fluorescein isothiocyanate [FITC], anti-CD5–phycoerythrin [PE], anti-CD3–PerCP, anti-CD38–PE, and the corresponding negative controls) were obtained from Becton Dickinson (San Jose, CA). Tri-color–conjugated mAb anti-CD5 and anti-CD38 were from Caltag (Burlingame, CA). Blocking mAb used in functional studies (anti-CD11a, -CD11c, -CD22, -CD50, and -CD58) were provided by Immunotech (Luminy, France). Blocking mAb anti-CD54 was obtained from Becton Dickinson. MAb anti-CD95 (ZB4 clone) was purchased from Biomol Research Laboratories (Plymouth Meeting, PA), and mAb anti-CD95L (NOK-1) was obtained from Pharmingen (San Diego, CA). The 24-well flat-bottomed culture plates were from Nunc (Roskilde, Denmark). Cell culture inserts were provided by Falcon (San Jose, CA). M-24 Dynatech microtiters plates used in ELISA were obtained from Dynatech Laboratories Ltd (Sussex, England). Polyclonal rabbit antibody against Fas-L (Q20) used in immunoblotting was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Peroxidase-coupled goat Ig antirabbit IgG used in immunoblotting was purchased from Promega (Madison, WI). The ECL system (Amersham, Buckingamshire, UK) was used for chemoluminescence analysis.
Preparation of purified B-CLL cells and CD5-depleted BM cells
Mononuclear cells were isolated from blood and BM samples by density centrifugation on Ficoll. All the CD5+ B-CLL cells used in this study were purified from patients' blood mononuclear cells by treatment with specific mAbs against T lymphocytes (anti-CD3), natural killers cells (anti-CD56), and monocytes (anti-CD14) and incubation with immunomagnetic beads conjugated with goat antimouse IgG according to manufacturers' instructions. CD5+ B-CLL cells were depleted from patients' BM by immunomagnetic selection using mAb against CD5. In brief, BM mononuclear cells at 2 × 107cells/mL were incubated with anti-CD5 mAb (2 μg/mL) for 30 minutes at 4°C, washed once, resuspended at 1 × 107 cells/mL, and incubated with immunomagnetic beads for 30 minutes at 4°C. The bead:cell ratio was 3:1. Cells that bound to beads were retained in the magnetic particle concentrator. Free cells were recovered, washed twice in Hank's balanced salt solution, and cultured. The purification obtained by these methods was verified by flow cytometry analysis. B-CLL cells contained more than 97% of CD19+CD5+ B-CLL cells. Fewer than 5% of CD5+ cells and fewer than 2% of CD3+ cells were present in the CD5-depleted BM cells, which in the remainder of this article will be referred to as patients' BM cells. Circulating IgG-tet–secreting cells were obtained from the blood of healthy volunteers who had been immunized with tet 6 days earlier, as reported.21
Cell culture
All the cultures were set up in 24-well flat-bottomed plates in a final volume of 1 mL per well. The cells were resuspended in a culture medium consisting of RPMI 1640 supplemented with 10% fetal calf serum, 10 mmol/L L-glutamine, and 0.05 mg/mL gentamycin. Culture plates were incubated at 37°C with 5% CO2. Patients' BM cells (106 cells/mL) were cultured alone and in combination with autologous B-CLL cells (106 cells/mL) for 7 days. In indicated experiments, purified B-CLL cells were induced with phorbol esters, as follows: B-CLL cells were cultured at a concentration of 5 × 106 cells/mL in the presence and in the absence of PMA (10 ng/mL). After 18 hours of culture, B-CLL cells were washed and used in cocultures with autologous BM cells, as above. In certain experiments, B-CLL cells and BM cells were cultured in the same well but separated by an insert membrane that prevented cellular contact. At the end of the culture period, cell-free culture supernatants were obtained, and the quantity of IgG, IgA, and IgM secretion was determined by ELISA as reported elsewhere.22 Additional experiments were carried out in which circulating IgG-tet–secreting cells (0.5 × 106 cells/mL) were cultured alone and in combination with increasing quantities of B-CLL cells for 7 days. IgG-tet secreted into the supernatant was measured by ELISA as previously reported.21 To assess the apoptosis of plasma cells, BM cells (106 cells/mL) were cultured alone or combined with autologous B-CLL cells (106 cells/mL) for 48 hours. After this period, the cells were recovered, washed twice in phosphate-buffered saline, and stained with FITC-labeled annexin V and PE-labeled anti-CD38 mAb using a previously described technique.23
Cell staining and flow cytometry
Cells (1 × 106/0.1 mL) were incubated with optimal concentrations of mAb for 20 minutes in the dark at 4°C in 1, 2, or 3 labeling experiments. Analysis was performed on a FACSCalibur cytometer (Becton Dickinson) equipped with an air-cooled argon ion laser emitting 15 mW at 480 nm. The cytometer was equipped with 3 fluorescence detector photomultiplier tubes, with green fluorescence (FITC) being collected through a 530/30-nm bandpass, orange-red (PE) through a 584/42-nm bandpass, and red (PerCP, TC) through a 650-nm long-pass filter. Cellular analysis was performed with Cellquest software (Becton Dickinson). Light scatter signals were recorded in linear scale and fluorescence signals in logarithmic mode. From 5000 to 10 000 events were acquired for every sample. Background fluorescence was determined with FITC-, PE-, PerCP-, and TC-conjugated control mAb of the appropriate isotype. The cursor was set so that fewer than 1% of the cells were positively stained in negative controls. BM plasma cells were defined as cells expressing high levels of surface CD38 molecules (CD38h cells).16,17 Apoptotic plasma cells were determined by flow cytometry as CD38h annexin V+ cells.23
CD95L reverse transcription–coupled polymerase chain reaction
Total RNA was prepared from 2 × 106 purified CD5+ B cells cultured overnight with and without PMA (10 ng/mL) and from 2 × 106 Jurkat cells that had been precultured with PMA (10 ng/mL) and ionomycin (5 μg/mL). RNA was isolated by the acid guanidine-phenol-chloroform method using the Tripure Isolation Reagent (Boehringer Mannheim, GmbH, Germany). Isolated RNA (10 μL) was transcribed to complementary DNA (cDNA) using 20 units of Moloney murine leukemia virus reverse transcriptase (Boehringer Mannhein) following the instruction of the manufacturers. Polymerase chain reaction (PCR) amplifications of FasL cDNA fragment was performed using the 5′-oligo 5′-CACCCCAGTCCACCCCCTGA-3′ and the 3′-oligo 5′-AGGGGCAGGTTGTTGCAAGA-3′ at a 400-nmol/L final concentration in a total reaction volume of 50 μL under the following conditions: 30 cycles of PCR with denaturation at 94°C for 1 minute, annealing at 60°C for 1 minute, and extension at 72°C for 1 minute. The reaction was performed using 1 unit of Taq polymerase (Boehringer Mannheim) on a DNA Thermal Cycler (Perkin Elmer Cetus). The expected products (225 base pairs) were resolved on an ethidium bromide–stained 1% agarose gel visualized under UV irradiation. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) primers were also used as control.
Analysis of CD95L protein by immunoblotting
Jurkat cells (106 cells/mL) were induced with PMA (10 ng/mL) and ionomycin (5 μg/mL) for 18 hours to express CD95L.24 Nonstimulated and PMA-induced B-CLL cells and induced Jurkat cells (106 cells) were resuspended in 1 mL of ice-cold lysis buffer consisting of 50 mmol/L HEPES, pH 7.4; 150 mmol/L NaCl; 10 mmol/L NaF; 10 mmol/L iodoacetamide; 200 μmol/L Na3VO4; and 1 mmol/L phenylmethylsulfonyl fluoride. After centrifugation at 15 000g for 20 seconds, the pellets were lysed in 100 μL of the same buffer supplemented with 1% Nonidet P-40. Proteins from cell lysates were resolved on 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred onto an Immobilon membrane (Millipore, Bedford, MA) by tank-blotting (100 mA, 50 minutes, 4°C). The blot was blocked with 5% milk powder in phosphate-buffered saline–0.1% Tween 20. The membrane was then incubated with the anti-CD95L antibody Q20 diluted to 1 μg/mL in the latter buffer. The blot was washed twice and developed with peroxidase-coupled antirabbit IgG as secondary antibody diluted at 1:5000, followed by enhanced chemoluminescence.
Statistical analysis
Results are generally expressed as the mean ± SEM or SD, as indicated. Significant differences were established by the Student t test. Correlation was estimated using the Pearson correlation test.
Results
Effect of B-CLL cells on autologous BM Ig-secreting cells
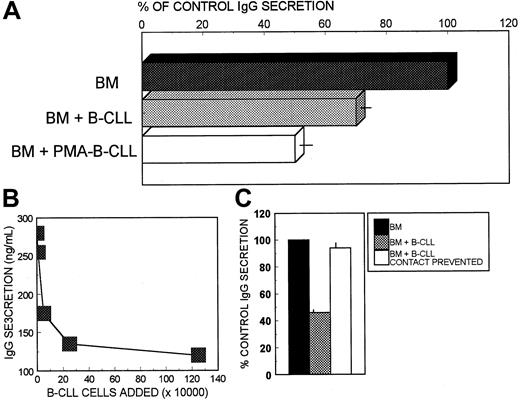
The aim of this study was to analyze the possibility that B-CLL cells played a direct role in the hypogammaglobulinemia observed in these patients. To this end, autologous BM preparations containing the cells capable of spontaneous and high-rate Ig secretion (BM Ig-secreting cells) were cultured in the absence and in the presence of purified B-CLL cells. Figure 1A summarizes the data obtained from studying 36 CLL patients. The addition of B-CLL cells inhibited an average of 30% of the IgG secretion by autologous BM cells (P < .00001). This effect increased up to about 50% inhibition of IgG secretion when the B-CLL cells had been preincubated with PMA (P < .00001) (Figure 1A). Spontaneous IgA and IgM secretion was also reduced in these experiments, reaching a mean percentage of inhibition of 24.6% ± 2% and 42% ± 2% for IgA and 21.5% ± 3% and 49.2% ± 3% for IgM when autologous secreting cells were cocultured with untreated and PMA-treated B-CLL cells, respectively (mean control production in these experiments was 207 ± 52 and 104 ± 35 ng/mL for IgA and IgM, respectively; mean ± SEM; n = 7;P < .0001 or lower). PMA induction of the inhibitory ability of B-CLL cells in this system did not depend on de novo RNA synthesis, because the addition of the specific inhibitor actinomycin D, along with the PMA, did not modify their inhibitory capacity (55% ± 9% and 54% ± 7% of control BM cell IgG production after the addition of B-CLL cells treated with and without actinomycin C, respectively; mean ± SD; n = 12). In contrast, the inclusion during the PMA pretreatment of cycloheximide (an inhibitor of protein synthesis) markedly reduced the PMA-inducing effect on the described inhibition (86% ± 8% and 54% ± 7% of control BM cell IgG production after the addition of B-CLL cells treated with and without cycloheximide, respectively; mean ± SD; n = 12). Figure 1B shows that the inhibition of BM cell Ig secretion was proportional to the number of B-CLL cells added to the culture, until a plateau was reached. To explore whether this inhibitory effect was mediated by soluble factors or required cellular interaction, cocultures of B-CLL cells and autologous BM cells were set up in which the cell contact was either allowed or prevented by the interposition between the 2 cell fractions of a membrane only permeable to soluble molecules. As shown in Figure 1C, the inhibition of Ig secretion was lost when the cellular contact between the BM Ig-secreting cells and the PMA-treated B-CLL cells was prevented. A similar effect was observed when untreated B-CLL cells were used as inhibitory cells in these cultures (mean percentage of inhibition of 30% ± 4% and 4.5% ± 1% for conventional and membrane-interposed coculture experiments, respectively; control IgG secretion in these experiments was 371 ± 95 ng/mL; mean ± SEM; n = 8). Thus, intimate interaction between both kinds of cells was required for the inhibitory effect to occur. Major histocompatibility complex proteins did not seem to be involved in this phenomenon, because autologous as well as allogeneic BM Ig-secreting cells were similarly inhibited by the B-CLL cells (data not shown). The possibilty that B-CLL cells exert an inhibitory effect on BM IgG-secreting cells obtained from healthy subjects23 was also explored. Neither untreated nor PMA-treated B-CLL cells modified the IgG production by normal BM IgG-secreting cells (7% ± 2% and 5% ± 2% of mean percentage of inhibition for untreated and PMA-treated B-CLL cells, respectively; control IgG production was 3149 ± 662 ng/mL; mean ± SEM; n = 8).
B-CLL cells inhibit Ig secretion by autologous BM cells.
(A) BM cells from patients with CLL (106 cells/mL) were cultured alone or in combination with autologous B-CLL cells (106 cells/mL), either nonstimulated or PMA-induced. At the end of the 7-day culture period, cell-free supernatants were collected and their content in IgG determined by ELISA. The values were expressed as the percentage of control IgG production (BM cells cultured alone). Results are expressed as the mean ± SEM of 36 different patients. Control IgG production in these experiments was 236 ± 72 ng/mL (mean ± SD; n = 36). Purified B-CLL cells cultured alone produced less than 5 ng/mL of either Ig isotype. (B) Effect of adding increasing numbers of PMA-induced B-CLL cells on the IgG-secreting capacity of autologous BM cells (106 cells/mL). Results of one representative experiment are shown. (C) Cellular mechanism involved in this phenomenon. BM cells were cultured alone and in combination with PMA-induced autologous B-CLL cells, either allowing or preventing the contact between the 2 kinds of cells. IgG secreted into the supernatants during 7 days of culture period was determined by ELISA. The values were expressed as the percentage of control IgG production (BM cells cultured alone). Results are expressed as the mean ± SEM of 12 different patients. Control IgG production in these experiments was 443 ± 68 ng/mL (mean ± SEM; n = 12).
B-CLL cells inhibit Ig secretion by autologous BM cells.
(A) BM cells from patients with CLL (106 cells/mL) were cultured alone or in combination with autologous B-CLL cells (106 cells/mL), either nonstimulated or PMA-induced. At the end of the 7-day culture period, cell-free supernatants were collected and their content in IgG determined by ELISA. The values were expressed as the percentage of control IgG production (BM cells cultured alone). Results are expressed as the mean ± SEM of 36 different patients. Control IgG production in these experiments was 236 ± 72 ng/mL (mean ± SD; n = 36). Purified B-CLL cells cultured alone produced less than 5 ng/mL of either Ig isotype. (B) Effect of adding increasing numbers of PMA-induced B-CLL cells on the IgG-secreting capacity of autologous BM cells (106 cells/mL). Results of one representative experiment are shown. (C) Cellular mechanism involved in this phenomenon. BM cells were cultured alone and in combination with PMA-induced autologous B-CLL cells, either allowing or preventing the contact between the 2 kinds of cells. IgG secreted into the supernatants during 7 days of culture period was determined by ELISA. The values were expressed as the percentage of control IgG production (BM cells cultured alone). Results are expressed as the mean ± SEM of 12 different patients. Control IgG production in these experiments was 443 ± 68 ng/mL (mean ± SEM; n = 12).
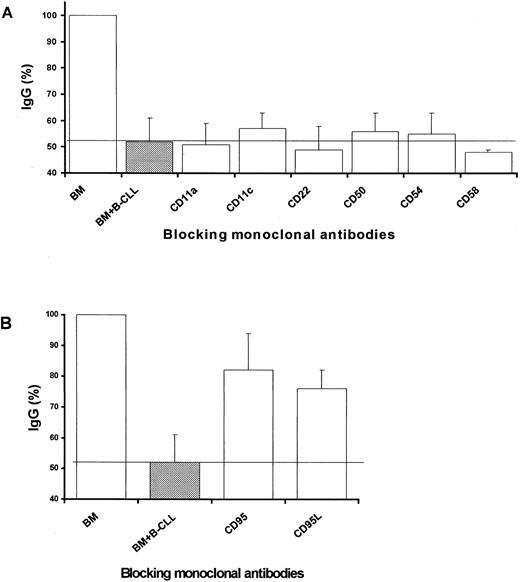
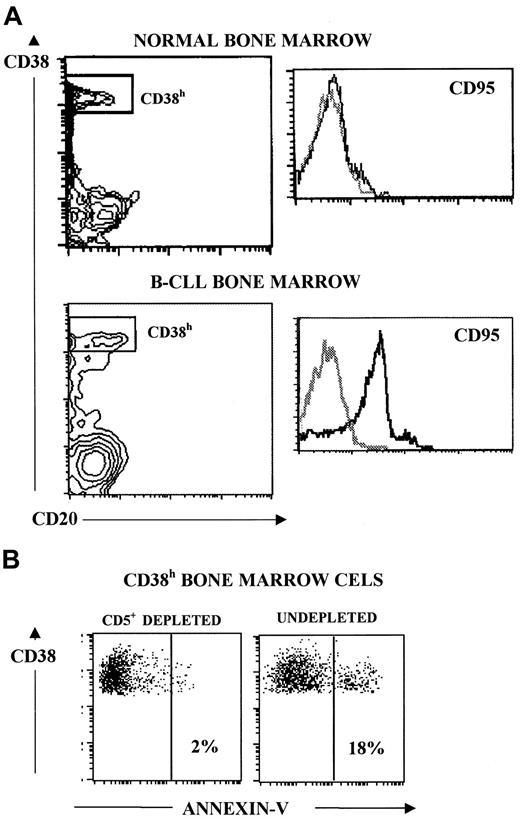
Analysis of the molecules implicated in the cellular interaction required for B-CLL cells to inhibit autologous BM Ig-secreting cells
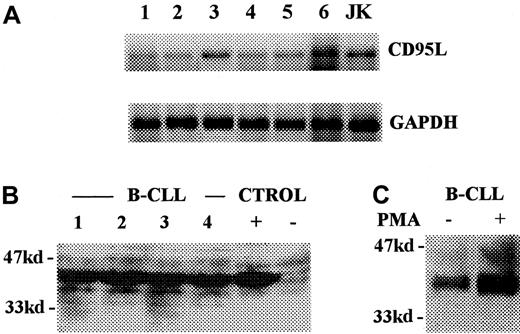
The molecular mechanism(s) by which the described inhibitory effect was exerted was investigated next. Adhesion molecules are commonly involved in processes requiring intercellular contact. Accordingly, the effect of adding blocking mAbs directed to several of these molecules was assessed on the described inhibitory event. As can be seen in Figure 2A, mAb to CD11a, CD11c, CD22, CD50, CD54, and CD58 added at 10 μg/mL did not modify the inhibition of BM cell Ig secretion by B-CLL cells. Alternatively, we looked for molecules present on the surface of patients' BM plasma cells that could be targets for inhibitory signals. In this regard, human plasma cells present in certain lymphoid territories can express CD95 (FAS, APO-1) and are susceptible to undergoing apoptosis via CD95 cross-linking.23 Figure 3A shows that patients' BM plasma cells, defined as CD38hcells, exhibited marked expression of CD95. Normal BM plasma cells, however, showed poor expression of this molecule (Figure 3A), and B-CLL cells did not express CD95 (data not shown). Accordingly, the possibility that the interaction between CD95 and CD95L was involved in the present system was also investigated. As can be seen in Figure 2B, the addition of blocking mAbs against these 2 interacting surface proteins significantly reversed the inhibitory effect of B-CLL cells on autologous BM Ig-secreting cells. These results suggest that B-CLL cells might bear CD95L. In fact, it has been reported that CD95L is expressed by B-CLL cells, as determined by recognition with specific mAb and fluorescence-activated cell sorter analysis.25 To confirm this point, the presence of CD95L messenger RNA (mRNA) was explored in B-CLL cells. As shown in Figure4A, message for this molecule was detectable in B-CLL cells by reverse transcription–PCR. The presence of CD95L at the protein level was also investigated by Western blot analysis of B-CLL cell lysates. Figure 4B shows that B-CLL cells isolated from patients (lanes 1-4) contained CD95L protein observed as a 40-kd band, similar to that detected in lysates of induced Jurkat cells that were used as control (lanes − and +). As shown in Figure 4C, the quantity of CD95L present in the lysates of nonstimulated B-CLL cells clearly increased on PMA stimulation.
Study of the molecules implicated in the inhibitory effect.
BM cells from patients with B-CLL (1 × 106) were cultured in the presence and the absence of PMA-induced autologous B-CLL cells (1 × 106). Blocking mAb against a variety of adhesion molecules (A) and against CD95 and CD95L (B) were added to the latter coculture (10 μg/mL) as indicated. Values were expressed as percentages of control B-CLL BM IgG secretion (BM cells cultured alone). Results represent the mean ± SD of 8 experiments. Control IgG production in these experiments was 251 ± 64 ng/mL (mean ± SEM; n = 8).The continuous line indicates the mean inhibition of IgG production by PMA-induced B-CLL cells in these experiments. Significant reversal of the inhibitory effect was only observed for mAb anti-CD95 and anti-CD95L, as determined by the Student t test (P < .001 and P < .005, respectively).
Study of the molecules implicated in the inhibitory effect.
BM cells from patients with B-CLL (1 × 106) were cultured in the presence and the absence of PMA-induced autologous B-CLL cells (1 × 106). Blocking mAb against a variety of adhesion molecules (A) and against CD95 and CD95L (B) were added to the latter coculture (10 μg/mL) as indicated. Values were expressed as percentages of control B-CLL BM IgG secretion (BM cells cultured alone). Results represent the mean ± SD of 8 experiments. Control IgG production in these experiments was 251 ± 64 ng/mL (mean ± SEM; n = 8).The continuous line indicates the mean inhibition of IgG production by PMA-induced B-CLL cells in these experiments. Significant reversal of the inhibitory effect was only observed for mAb anti-CD95 and anti-CD95L, as determined by the Student t test (P < .001 and P < .005, respectively).
CD95 expression and apoptotic induction in patients' BM plasma cells (CD38h cells).
(A) Three-color staining and flow cytometry analysis for CD95 expression by BM plasma cells from healthy subjects (upper panel) and patients with B-CLL (lower panel). Plasma cells were selected in a CD38 CD20 plot as cells CD38h CD20−. CD95 expression is shown in histograms. The results shown are representative of 8 and 5 experiments for patients with B-CLL and healthy BM samples, respectively. (B) BM cells (106 cells/mL) were cultured alone and in combination with untreated autologous B-CLL cells (106 cells/mL) for 48 hours. After this period, apoptotic BM plasma cells were determined in both kinds of culture as CD38h annexin V+ cells. Results of one representative experiment are shown.
CD95 expression and apoptotic induction in patients' BM plasma cells (CD38h cells).
(A) Three-color staining and flow cytometry analysis for CD95 expression by BM plasma cells from healthy subjects (upper panel) and patients with B-CLL (lower panel). Plasma cells were selected in a CD38 CD20 plot as cells CD38h CD20−. CD95 expression is shown in histograms. The results shown are representative of 8 and 5 experiments for patients with B-CLL and healthy BM samples, respectively. (B) BM cells (106 cells/mL) were cultured alone and in combination with untreated autologous B-CLL cells (106 cells/mL) for 48 hours. After this period, apoptotic BM plasma cells were determined in both kinds of culture as CD38h annexin V+ cells. Results of one representative experiment are shown.
Presence of CD95L in B-CLL cells.
(A) Reverse transcription–PCR detection of CD95L mRNA. Lanes 1 to 6 correspond to B-CLL cells from different patients. Lane 7 corresponds to Jurkat cells induced by PMA and ionomycin (positive control). GAPDH mRNA was used as mRNA quantity control. (B) Western blot analysis of CD95L expression in B-CLL lymphocytes (lanes 1 to 4). Lanes 5 and 6 correspond to Jurkat cells cultured in the presence (positive control: +) and in the absence (negative control: −) of PMA plus ionomycin. (C) Western blot analysis of the expression of CD95L proteins by nonstimulated (control) and PMA-induced B-CLL cells.
Presence of CD95L in B-CLL cells.
(A) Reverse transcription–PCR detection of CD95L mRNA. Lanes 1 to 6 correspond to B-CLL cells from different patients. Lane 7 corresponds to Jurkat cells induced by PMA and ionomycin (positive control). GAPDH mRNA was used as mRNA quantity control. (B) Western blot analysis of CD95L expression in B-CLL lymphocytes (lanes 1 to 4). Lanes 5 and 6 correspond to Jurkat cells cultured in the presence (positive control: +) and in the absence (negative control: −) of PMA plus ionomycin. (C) Western blot analysis of the expression of CD95L proteins by nonstimulated (control) and PMA-induced B-CLL cells.
The possibility that the cellular mechanism implicated in the described inhibitory effect was the B-CLL cell induction of patients' plasma cell apoptosis was examined next. To this end, BM cells were cultured either alone or in combination with untreated autologous B-CLL cells for 2 days, and apoptotic plasma cells were identified as CD38h annexin V+ cells.23 Figure3B shows a representative experiment. Apoptotic plasma cells increased from an average percentage of 6.8% ± 4% (BM cells cultured alone) to 35% ± 11% (BM cells cocultured with B-CLL cells) (mean ± SEM; n = 3).
Correlation between the in vitro inhibition of BM IgG-secreting cells by autologous B-CLL cells and the patients' level of serum IgG
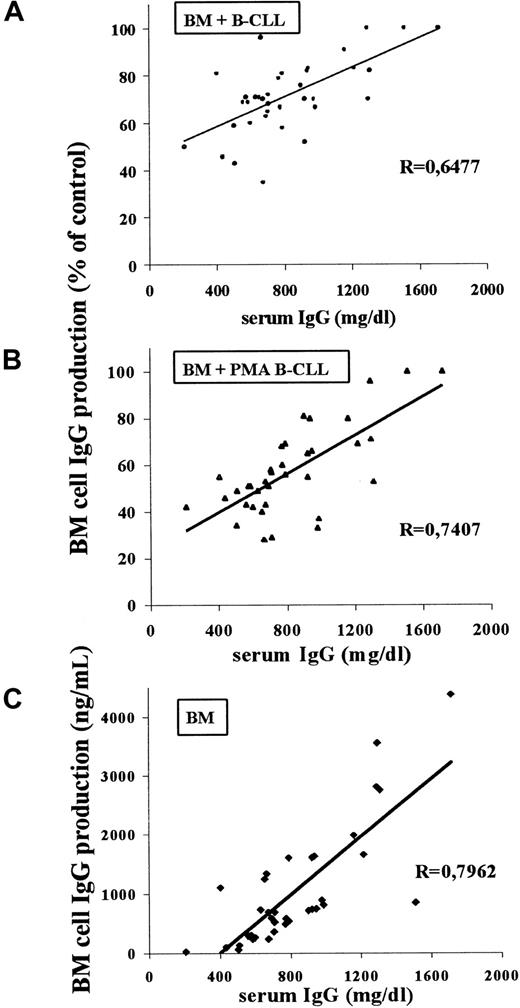
The relationship between the described inhibitory phenomenon and the gammaglobulinemia detected at the time of the study was explored in the 36 CLL patients. As seen in Figure 5, there was a significant correlation between these parameters because the higher the inhibition of in vitro BM cell Ig secretion by both nonstimulated (Figure 5A) and PMA-induced (Figure 5B) B-CLL cells, the lower the corresponding serum IgG levels. Moreover, a good correlation was also found between the patients' serum IgG level and the in vitro IgG production by the corresponding BM cells cultured alone (Figure 5C).
Scatter plot and regression line.
The plot and line were obtained in the analysis of the correlation between the in vivo degree of hypogammaglobulinemia (serum IgG level at the moment of the study) and the in vitro inhibition of BM cell IgG production by autologous B-CLL cells, both nonstimulated (A) or PMA-induced (B), as well as between the serum IgG level and the IgG secreted by cultured BM cells (C) for the 36 patients with CLL in this study. The correlation index (r) and the P values were calculated by the Pearson correlation test and were .000 01 for situations A, B, and C.
Scatter plot and regression line.
The plot and line were obtained in the analysis of the correlation between the in vivo degree of hypogammaglobulinemia (serum IgG level at the moment of the study) and the in vitro inhibition of BM cell IgG production by autologous B-CLL cells, both nonstimulated (A) or PMA-induced (B), as well as between the serum IgG level and the IgG secreted by cultured BM cells (C) for the 36 patients with CLL in this study. The correlation index (r) and the P values were calculated by the Pearson correlation test and were .000 01 for situations A, B, and C.
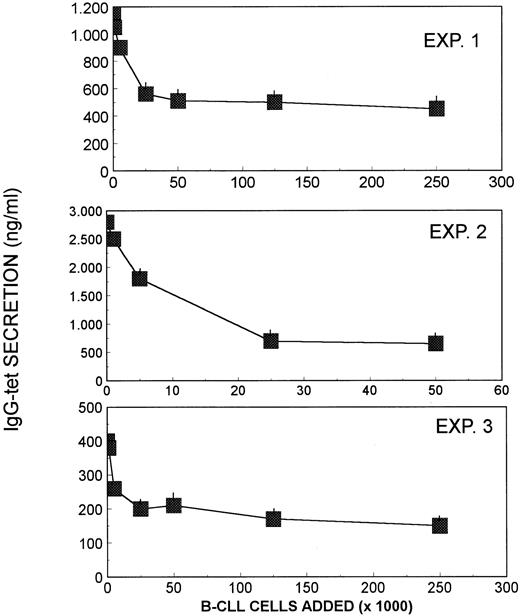
Effect of B-CLL cells on in vivo antigen-induced IgG-tet–secreting cells
Immunization of humans with antigens (ie, tet) leads, 5 to 7 days later, to the transient appearance of circulating B cells capable of spontaneous antibody (IgG-tet) secretion in culture.21These cells represent a model of in vivo antigen-induced plasma cells. In addition, it has been demonstrated that these cells are susceptible to inhibition by CD95 cross-linking.23 Therefore, the effect of B-CLL cells was also assessed on the IgG-tet–secreting cells obtained from healthy donors. Figure 6shows that B-CLL cells markedly inhibited IgG-tet secretion by in vivo tet-induced plasma cells, and this phenomenon was proportional to the number of B-CLL cells added.
Effect of B-CLL cells obtained from 3 different patients on in vivo antigen-induced B cells capable of IgG-tet secretion obtained from the blood of 3 different healthy volunteers who had been immunized with tet 6 days earlier.
Blood B cells (5 × 105 cells/mL) were cultured in the absence and in the presence of increasing numbers of B-CLL cells for 7 days. IgG-tet secreted into the culture supernatant was determined by ELISA.
Effect of B-CLL cells obtained from 3 different patients on in vivo antigen-induced B cells capable of IgG-tet secretion obtained from the blood of 3 different healthy volunteers who had been immunized with tet 6 days earlier.
Blood B cells (5 × 105 cells/mL) were cultured in the absence and in the presence of increasing numbers of B-CLL cells for 7 days. IgG-tet secreted into the culture supernatant was determined by ELISA.
Discussion
Humoral immunodeficiency is a frequent and occasionally severe complication in patients with CLL. This feature has been mostly explained as a result of abnormal T-cell regulation of the B-cell function in this disease.12 The present study examines the possibility that this defect is directly caused by the tumoral cells. Data presented here indicate that B-CLL cells are capable of inhibiting autologous BM Ig-secreting cells in vitro. This effect was carried out by purified, freshly isolated B-CLL cells and was enhanced when the tumoral cells were pretreated with the protein kinase C–activator PMA in a process that depended on de novo protein, but not RNA, synthesis. The inhibitory effect of purified B-CLL cells on BM Ig-secreting cells was detected in all but 2 of the patients. Therefore, B-CLL cells exhibit the capacity of inhibiting Ig secretion by terminally differentiated stages of B cells present in their own BM.
Experiments conducted to delineate the cellular mechanisms involved in the present phenomenon revealed that contact between B-CLL cells and BM Ig-secreting cells was an essential prerequisite for the inhibition to occur. Accordingly, the molecular mechanisms implicated in this process were investigated. The results show that adhesion molecules did not appear to be involved in this process. Instead, the recognition between the well-known death receptor CD95 26 and CD95L, its natural ligand,27 seemed to be critical in the present system. This was first indicated by the fact that the addition of mAb capable of preventing the triggering through CD95 28 and of blocking the CD95L signaling function29 markedly reversed the inhibitory effect of B-CLL cells on autologous BM Ig-secreting cells. The next step was to define the cell origin of each of these molecules. A previous report had shown the expression of CD95L on B-CLL cells by using a mAb and flow cytometry.25Present results confirm this observation, because mRNA for CD95L was present in freshly isolated B-CLL cell samples. In addition, CD95L protein was also detected in the B-CLL cell lysates by immunoblotting. The quantity of this protein increased with the PMA pretreatment, which probably explains the enhanced inhibitory effect exerted by PMA-induced B-CLL cells. In contrast, B-CLL cells did not express CD95. On the other hand, CLL patients' BM plasma cells, defined as CD38h cells,16 17 clearly expressed CD95 on their surface but lacked CD95L expression as determined by flow cytometry (data not shown). The reason CD95 expression by patients' BM CD38h cells was markedly higher than that exhibited by similar cells obtained from the BM of healthy individuals remains to be elucidated. In good agreement with the low CD95 expression shown by BM CD38h cells from healthy subjects, they were not susceptible to inhibition by B-CLL cells. Collectively, these data support the view that the interaction of CD95L present on the surface of B-CLL cells with CD95 expressed on patients' BM plasma cells is critical for the triggering of the inhibitory effect.
The cellular mechanism by which B-CLL cells inhibited autologous BM plasma cells is likely to be the induction of apoptosis in these latter cells. First, this is the effect normally produced in a CD95L-CD95 interaction.30 Second, it has been previously shown that freshly isolated B-CLL cells kill certain target cells by inducing their apoptosis in a process that is reversible by blocking the interaction between CD95 and CD95L.25 Third, it has been demonstrated that the ligation of CD95 on susceptible human plasma cells, either normal or present in reactive plasmocytosis, leads to apoptosis of these cells.23 31 Finally, present data indicate that patients' BM plasma cells undergo apoptosis on B-CLL cell contact, as determined by annexin V–binding assay.
The possible relevance in vivo of the described inhibitory phenomenon is first supported by the well-established fact that BM Ig-secreting plasma cells are responsible for most of the serum Ig and antibody formation in vivo in mice and humans14,15 and, in consequence, their inhibition could have a strong influence on humoral immune responses. Furthermore, a correlation was found between the intensity of the B-CLL cell inhibitory effect in vitro and the decrease of the IgG level in the patients' serum as well as between this latter variable and the IgG secreted by the corresponding BM cells in vitro, demonstrating that these parameters appeared closely related. In addition, circulating IgG-tet–secreting cells, a model of in vivo antigen-induced plasma cells in humans,21 were readily inhibited by B-CLL cells as well. In this regard, it is well documented that BM plasma cells are not generated in situ, but they are formed in distant lymphoid organs on antigen stimulation and travel through the circulation toward the BM, where they finally reside and perform their secretory capacity for prolonged periods.32 33 Therefore, it is conceivable that B-CLL cells, which become extremely frequent in the blood and can infiltrate lymphoid organs, including the BM, might have access to circulating and tissue plasma cells and cause their elimination by apoptosis. Taken together, these results strongly suggest that the B-CLL cell inhibitory mechanism on autologous plasma cells described here can help to explain the humoral immune-deficient state present in these patients.
Acknowledgments
The authors thank J. Muñoz and R. Ladines (Servicio de Hematologı́a, Hospital Universitario Puerta del Mar, Cádiz, Spain) for their cooperation in obtaining patients' samples, A. López-Rivas for his help with the Western blot analysis, and C. Hurtado and F. Nieto for their excellent technical assistance.
Supported by grant 98/1182 from the Fondo de Investigaciones Sanitarias of Spain and by grant 149/98 from the Servicio Andaluz de Salud of Spain.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
José A Brieva, Servicio de Inmunologı́a, Hospital Universitario Puerta del Mar, Avenida Ana de Viya 21, 11009 Cádiz, Spain; e-mail:jabrieva@hpm.sas.cica.es

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal