Abstract
CD64 (FcγRI) receptors represent highly potent trigger molecules for activated polymorphonuclear cells (PMN) and mediate lysis of a range of tumors in the presence of appropriate monoclonal antibodies. An huCD64 transgenic mouse model designed to analyze the therapeutic activity of a panel of bispecific F(ab')2(BsAb) in retargeting granulocyte–colony-stimulating factor (G-CSF)–activated PMN against syngeneic B-cell lymphomas is reported. This model allows careful analysis of the individual elements of the therapeutic process. BsAb were directed against immunoglobulin-idiotype (Id), major histocompatibility class II (MHC II), or CD19 on the tumors and huCD64 on the effectors. In vitro cytotoxicity assays and in vivo tumor tracking showed that, provided effectors were activated with G-CSF, all 3 derivatives destroyed and cleared lymphoma cells, with (huCD64 × MHC II) proving by far the most cytotoxic in vitro. However, though all derivatives delivered some survival advantage, only the [huCD64 × Id] BsAb provided long-term protection to tumor-bearing animals. These results demonstrate that CD64-recruited cytotoxic effectors operate in vivo but that the (huCD64 × Id) conferred an additional anti-tumor function essential for long-term protection. T-cell depletion studies demonstrated that this extra therapeutic activity with [huCD64 × Id] was totally dependent on CD4 and CD8 T cells and that mice, once “cured” with BsAb, were resistant to tumor rechallenge. These findings indicate that CD64 is an effective trigger molecule for delivering cytokine-activated PMN against tumor in vivo and that, provided tumor targets are selected appropriately, CD64-based BsAb can establish long-term T-cell immunity.
Introduction
Recent clinical trials have generated considerable optimism for the use of monoclonal antibodies (mAb) in a range of neoplastic diseases.1 Two reagents in particular are behind the current enthusiasm, chimeric anti-CD20 (Rituximab), used in relapsed non-Hodgkin lymphoma,2 and humanized anti-Her-2 (Herceptin), which reacts with the proto-oncogene erbB2 and is used in metastatic breast cancer.3 Both products are relatively nontoxic and provide benefits to patients. Perhaps the most interesting aspect of this work arises from recent evidence showing that when mAb are used in combination with certain cytotoxic drugs or radiotherapy, the therapeutic efficacy, but not the toxicity, of the 2 moieties are additive or even synergistic.4
Despite such successes we are still surprisingly ignorant of how mAb operate in vivo.2,4-8 Direct transmembrane signaling to the tumor cell, recruitment of cellular effectors, and activation of complement-dependent lysis have all been implicated as possible mechanisms. Recent in vivo evidence underlines the importance of FcR in mediating mAb function and shows that not only are FcR essential for mAb anticancer activity, but that they can act as inhibitory molecules on certain effector cells and significantly diminish killing activity.9 10 Thus, under normal circumstances, the therapeutic activity of any particular mAb will be determined by a fine balance of inhibitory and stimulatory signals, controlled by different FcR and dependent on the mAb and its isotype.
In the current study we have investigated the potential of a panel of BsAb to retarget polymorphonuclear cells (PMN) against lymphoma through their FcγRI/CD64 trigger molecules. The BsAb exhibit dual specificity for the target and effector cells and are F(ab')2derivatives that recruit the effectors with an antibody binding site rather than an Fc region.11 BsAb are an attractive alternative to conventional IgG mAb and can be designed to recruit a range of cellular effectors, including T cells, natural killer cells, and FcR-bearing myeloid cells. Their potential benefits include the ability to recruit effectors through trigger molecules (eg, CD64 for neutrophils), the lack of an Fc region that will avoid inhibitory FcR, and the possibility of binding to a trigger molecule outside its natural ligand-binding site and avoiding blocking by the natural ligand.12 To date the most promising FcR for therapeutic applications with BsAb are FcγRIII (CD16), FcαRI (CD89), and FcγRI (CD64),11,12 all of which trigger cellular cytotoxicity or phagocytosis of tumor cells. CD64 is constitutively expressed on monocytes/macrophages and some dendritic cells, and it is induced on neutrophils by IFN-γ or granulocyte–colony-stimulating factor (G-CSF).9 G-CSF–induced expression of CD64 renders neutrophils a major effector-cell population for recruitment by BsAb,13 and phase I clinical trials have been initiated to test CD64-directed BsAb in combination with G-CSF for the treatment of metastatic breast cancer.14 15
Because there are no mAb available for mouse CD64, until now it has not been possible to test CD64 BsAb in syngeneic tumor models. The generation of CD64 transgenic (Tg) mice, expressing the human FcR as a transgene under the control of the endogenous regulatory elements, has overcome this limitation.16 17 Here we show for the first time direct evidence that CD64-directed BsAb are highly therapeutic in vivo, are more active than IgG mAb, and that certain BsAb can provide long-term protection to tumor-bearing animals.
Materials and methods
Animals and cell lines
BALB/c and CBA/H mice were supplied by Harlan UK (Blackthorn, UK). Mice (FVB/n) expressing the human CD64 transgene16were crossed with BALB/c and CBA/H to produce F1 mice that were compatible for tumor growth. BCL118 and A3119 B lymphoma lines were maintained by in vivo (intraperitoneal) passage in BALB/c and CBA/H mice, respectively.20 The πBCL1 cell line is a variant of the BCL1 tumor that can be maintained both in vivo and in culture.21
All cell cultures were grown in supplemented Dulbecco modified Eagle medium (DMEM) (Life Technologies, Paisley, UK) containing glutamine (2 mmol/L), pyruvate (1 mmol/L), penicillin and streptomycin (100 IU/mL), Fungizone (2 μg/mL), and 5% to 10% fetal calf serum (FCS; Myoclone; Life Technologies), except πBCL1, which was maintained in supplemented RPMI (Life Technologies), plus 50 μmol/L 2-ME (BDH, Poole, UK) and 20% FCS (Life Technologies).
Monoclonal and bispecific antibodies
The mAb used in this study were Mc10-6A5 (rat anti-BCL1 idiotype)22; Mc39-16 (rat anti-A31 idiotype)7; ID3 (rat antimouse CD19)23; N22 (hamster anti-mouse MHC class II) (ATCC, Rockville, MD); M22 (mouse anti-CD64)24; YTS 169 (rat antimouse CD8)25; and GK1.5 (rat anti-mouse CD4).25 Hybridoma cells were expanded in stationary culture using 5% supplemented DMEM. To purify the IgG mAb, supernatants were concentrated by membrane filtration (Amicon, Beverly, MA), precipitated with saturated ammonium sulfate, and dialyzed and fractionated on protein G (Pharmacia, Piscataway, NJ).
F(ab')2 fragments were prepared from IgG by limited proteolysis with pepsin at pH 4.1 to 4.2 in 0.1 mol/L sodium acetate.26 Bispecific F(ab')2 heterodimers were prepared using o-phenylenedimaleimide (o-PDM; Sigma, UK).26 27 The constructs were size fractionated and passed through an anti-Fcγ immunosorbent column to remove any traces of intact IgG contaminating the preparation.
Flow cytometry to determine huCD64 expression
Whole blood obtained from tail bleeds of 6-week-old mice was analyzed by direct immunofluorescence staining using a FACSCalibur (Becton Dickinson, San Jose, CA). Samples were incubated with fluorescein isothiocyanate (FITC)-labeled M22 (50 μg/mL) for 20 minutes on ice, treated once with FACS-lysing solution (Becton Dickinson) for 1 minute, then washed with phosphate-buffered saline (PBS) containing 1% bovine serum albumin, and analyzed for expression of huCD64 by gating on the PMN population as determined by FSC/SSC profiles.
Redirected cellular cytotoxicity assay
In vitro cytotoxicity was measured using a standard chromium51Cr-release assay.7 Briefly,51Cr-labeled target cells (A31 or BCL1) were prepared from spleens of terminal animals and resuspended at 105/mL in supplemented RPMI. The effectors were murine PMN from whole blood separated on a discontinuous Percoll (Sigma) gradient.28 To mobilize PMN, mice received 2 μg/d recombinant murine G-CSF (Amgen, Thousand Oaks, CA) subcutaneously for 4 days before PMN isolation. After separation PMN were washed once and resuspended in supplemented RPMI. For the assay, 50 μL aliquots of target cells (5 × 103) and BsAb were mixed in wells of a 96-well U-bottomed plate (Life Technologies) and left on ice for 15 minutes. Aliquots (100 μL) of effector cells were then added at an effector:target (E:T) ratio of 50:1. Plates were centrifuged at 200g for 5 minutes at room temperature, incubated for 4 hours at 37°C, and recentrifuged at 500g for 5 minutes before 100 μL of supernatant was harvested from each well to estimate the released 51Cr.
Determinations were in triplicate, and maximal release of51Cr was obtained using target cells to which 150 μL of 1% Nonidet P-40 had been added. The percentage specific51Cr release was calculated using the formula: [(sample release − background release)/(maximum release − background release)] × 100.
Immunotherapy
Groups of age-matched F1 mice were injected intraperitoneally with 105 BCL1 (BALB/c × FVB/n) or 2 × 105 A31 (CBA × FVB/n) lymphoma cells. If appropriate, mice also received 2 μg/d murine G-CSF subcutaneously to mobilize PMN, administered daily for 10 days from 4 days before to 5 days after the tumor inoculum. For most experiments mice were treated with BsAb (intraperitoneally) twice daily on days 1 to 5 after the tumor. Both tumors develop primarily in the spleen, with a leukemic overspill toward the end of the disease.18,19Lymphoma-bearing animals surviving for more than 100 days after treatment with BsAb were rechallenged, along with age matched naive mice, with 104 fresh tumor cells. Survival was monitored daily, and the results were analyzed using the χ2 test of Peto.29 Animal immunotherapy was cleared through the local ethical committee and was performed under Home Office license.
Tracking tumor cells in vivo
To investigate the activity of BsAb early in the course of tumor development, groups of mice were inoculated with 107BCL1 tumor cells intraperitoneally on day 0 and then treated with 50 μg BsAb intraperitoneally twice daily, on days 1 to 5 (500 μg/mouse in total). If necessary mice also received murine G-CSF (2 μg/d) as before. Mice were killed on consecutive days throughout the course of therapy, and peritoneal cells were removed by lavage with PBS. Cells were washed once by centrifugation and resuspended in supplemented RPMI at 106/mL.
Tumor cells in the exudate were detected by dual-fluorescence staining using FITC-NIMR6 (rat anti-mouse CD22)30 and phycoerythrin (PE)-Mc10-6A5 (rat anti-BCL1 Id). Cells were stained with mAb (50 μg/mL) on ice for 15 minutes. In the case of mice treated with anti-Id mAb, which caused blocking or modulation of the surface Id, cells were stained with FITC-NIMR6 (rat anti-mouse CD22) and PE-ID3 (rat anti-mouse CD19) instead. After staining, cells were treated with FACS lysis solution to lyse red blood cells and then washed with PBS containing 1% bovine serum albumin
To detect the presence of tumor cells in the spleen, homogenates were washed by centrifugation, and cells were resuspended in supplemented RPMI at a concentration of 106/mL. Cells then were stained using either FITC-NIMR6 versus PE-Mc10-6A5 to detect BCL1cells or FITC-M22 (anti-human CD64) and PE-7/4 (rat anti-mouse neutrophil) (VH Bio, Newcastle Upon Tyne, UK) to detect PMN.
Measurement of intracellular free calcium
Intracellular free calcium concentration was measured using the cell-permeable fluorescent probe Indo-1-am(Sigma). Indo-1-am gives a shift in fluorescence emission from 482 nm to 398 nm on calcium binding, and an increase in the ratio of the 398 nm/482 nm signal is indicative of increased intracellular free calcium. Cultured πBCL1 cells (107/mL) were washed twice in serum-free media and loaded with Indo-1-am at 5 μmol/L for 30 minutes at 37°C. Cells were then washed twice by centrifugation in serum-free RPMI to remove excess dye and were resuspended in supplemented RPMI at 5 × 106 cell/mL. Labeled cells were stored in the dark at room temperature until used. For the assay, 250 μL of this cell suspension was warmed to 37°C for 5 minutes, an equal volume of mAb solution added at a final concentration of 2 μg/mL, and immediately analyzed using flow cytometry on a FACS Vantage (Becton Dickinson). Where appropriate, hyper-cross-linking sheep anti-rat IgG or rabbit antihamster IgG was added at a final concentration of 20 μg/mL immediately before analysis. Intracellular calcium mobilization was estimated by determining the mean fluorescence ratio of Indo-1 bound (398 nm, FL5) to free calcium (482 nm, FL4) against time. The response was measured over 300 seconds and recorded using the Cell Quest program (Becton Dickinson).
Results
Expression of human FcγRI/CD64 in transgenic FVB/n mice
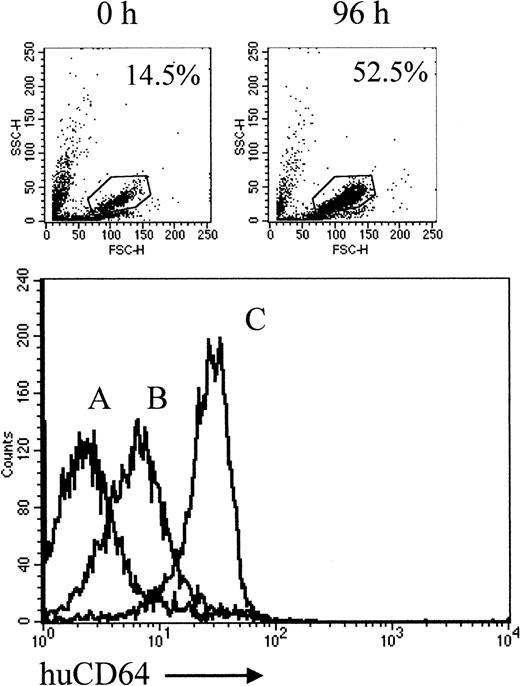
Transgenic FVB/n mice expressing human FcγRI/CD64 were generated with its transgene under the control of endogenous regulatory elements.16 This results in normal CD64 expression patterns on monocytes/macrophages, dendritic cells, and PMN, and responsiveness to stimulation with G-CSF. F1 mice (FVB/n × BALB/c and FVB/n × CBA), compatible for the growth of BCL1(BALB/c) and A31 (CBA) lymphoma, were screened for the expression of huCD64 on circulating PMN by flow cytometry. The 50% of animals expressing the transgene were selected for immunotherapy, and non-Tg littermates were controls. To confirm that huCD64 expression on PMN was responsive to G-CSF in the F1 mice, we examined levels before and after 4 days of treatment with cytokine. Figure1 shows expression in a non-Tg mouse (A), an untreated huCD64 Tg mouse (B), and a G-CSF–primed Tg mouse (C). Treating mice with G-CSF increased in the level of huCD64 expression on PMN. Although the expression of huCD64 could be increased with low levels of G-CSF (eg, 0.5 μg/d), the total number of circulating PMN was increased (from approximately 14% to 50%) only when mice were given 2 μg/d (Figure 1; inset). For this reason the treatment protocol included 2 μg G-CSF/d for all subsequent experiments.
Levels of huCD64 expression on PMN.
HuCD64+ mice were treated with murine G-CSF, 2 μg/d, subcutaneously, for 4 days. Levels of huCD64 on PMN were determined by FACS analysis of whole blood incubated with FITC-labeled M22 (anti-huCD64). The comparative expression levels on nontransgenic (A), unprimed (B), and primed (C) transgenic mice are shown. The level of staining given by a control irrelevant FITC-labeled IgG on PMN from Tg and nonTg was the same as that shown by curve A. The inset shows the scatter profiles, with the percentages of PMN (gated) in the total leukocyte population before and after the course of G-CSF.
Levels of huCD64 expression on PMN.
HuCD64+ mice were treated with murine G-CSF, 2 μg/d, subcutaneously, for 4 days. Levels of huCD64 on PMN were determined by FACS analysis of whole blood incubated with FITC-labeled M22 (anti-huCD64). The comparative expression levels on nontransgenic (A), unprimed (B), and primed (C) transgenic mice are shown. The level of staining given by a control irrelevant FITC-labeled IgG on PMN from Tg and nonTg was the same as that shown by curve A. The inset shows the scatter profiles, with the percentages of PMN (gated) in the total leukocyte population before and after the course of G-CSF.
Redirected cellular cytotoxicity of A31 and zcaseBCL1
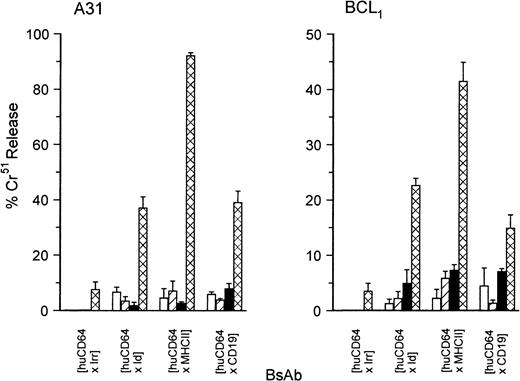
Considerable evidence suggests that, at least in human systems, CD64 is a potent trigger for PMN.31 To determine the cytotoxic potency of in vivo–activated mouse PMN we performed redirected cellular cytotoxicity (RCC) assays using huCD64-specific BsAb directed at 3 tumor targets, [huCD64 × Id], [huCD64 × CD19], and [huCD64 × MHC II]. Figure2 shows that activated PMN were able to lyse both A31 and BCL1 tumor cells in the presence of all 3 BsAb. However, MHC II–directed PMN clearly gave the highest levels of lysis, in particular against A31, when RCC resulted in more than 90% specific 51Cr-release. Tumor cells were not destroyed in the presence of PMN from non-Tg animals (huCD64-negative littermates) that were either unprimed or given G-CSF. This indicates that BsAb were indeed functioning by binding huCD64 on PMN. Importantly, tumor cells were also not lysed by PMN taken from huCD64 Tg mice that had not received G-CSF, presumably because these PMN were not activated and FcγRI levels were relatively low.
RCC of A31 and BCL1 using isolated murine PMN.
Chromium-labeled target cells were mixed with BsAb of varying specificity (as indicated) at 1 μg/mL, and then murine PMN were added at an E:T ratio of 50:1. PMN isolated from huCD64 Tg animals either without (solid bars) or with G-CSF priming (hatched bars) were compared to PMN from huCD64-non Tg littermates, again either unprimed (open bars) or G-CSF treated (lined bars). BsAb were of the specificity indicated. Control BsAb contained the alternative, nonbinding anti-Id Fab' arm for each tumor (ie, anti-BCL1 for A31 and anti-A31 for BCL1) and were ineffective. Similar results were obtained in 2 separate experiments. All determinations were performed in triplicate. Error bars represent the SD of the triplicates. ■, nonTg −G-CSF; ▨, nonTg +G-CSF; ▪, Tg −G-CSF; ⊠, Tg +G-CSF.
RCC of A31 and BCL1 using isolated murine PMN.
Chromium-labeled target cells were mixed with BsAb of varying specificity (as indicated) at 1 μg/mL, and then murine PMN were added at an E:T ratio of 50:1. PMN isolated from huCD64 Tg animals either without (solid bars) or with G-CSF priming (hatched bars) were compared to PMN from huCD64-non Tg littermates, again either unprimed (open bars) or G-CSF treated (lined bars). BsAb were of the specificity indicated. Control BsAb contained the alternative, nonbinding anti-Id Fab' arm for each tumor (ie, anti-BCL1 for A31 and anti-A31 for BCL1) and were ineffective. Similar results were obtained in 2 separate experiments. All determinations were performed in triplicate. Error bars represent the SD of the triplicates. ■, nonTg −G-CSF; ▨, nonTg +G-CSF; ▪, Tg −G-CSF; ⊠, Tg +G-CSF.
Immunotherapy of A31- and BCL1-bearing mice
Having established that our BsAb could mediate tumor killing in the presence of G-CSF–primed PMN, especially when targeted through MHC II, we next determined their efficacy in vivo. The therapeutic model was based on previous work from this32 and other33 laboratories, giving twice-daily treatments with BsAb to compensate for the short half-life of F(ab')2derivatives. Figure 3 illustrates an initial set of immunotherapy experiments obtained when lymphoma-bearing (A31 or BCL1) mice were treated with G-CSF and anti-Id–based derivatives. Interestingly, the [huCD64 × Id] BsAb provided complete tumor protection for almost all mice, even when used at a dose of just 50 μg/mouse. This result shows that CD64 is an effective trigger molecule for BsAb-based therapy in vivo. By contrast, neither anti-Id F(ab')2 nor a mixture of anti-Id and anti-huCD64 F(ab')2 provided any protection, demonstrating that only when the 2 Ab specificities are linked to form a bispecific unit does effector retargeting occur effectively.
Immunotherapy of A31- or BCL1-bearing mice with BsAb directed against huCD64 on the effector cells and Id on the tumors.
Groups of 5 huCD64+ age-matched mice received 2 μg/d murine G-CSF subcutaneously, 4 days before tumor inoculation, and throughout the course of therapy (10 days in total). Mice were inoculated with either 105 (BCL1) or 2 × 105 (A31) tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). (This treatment schedule is shown in the inset.) Treatment was as indicated: PBS (●), anti-huCD64 F(ab')2 (⋄), anti-Id F(ab')2 (■), a mixture of anti-Id and anti-huCD64 F(ab')2 (♦), [huCD64 × Id] (○). Symbol labeling on plots shows antibody specificity. Anti-Id Ab preparations were selected for reaction with A31 or BCL1 Id Ig, as appropriate. Survival was recorded daily. Only cohorts treated with [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P < .02).
Immunotherapy of A31- or BCL1-bearing mice with BsAb directed against huCD64 on the effector cells and Id on the tumors.
Groups of 5 huCD64+ age-matched mice received 2 μg/d murine G-CSF subcutaneously, 4 days before tumor inoculation, and throughout the course of therapy (10 days in total). Mice were inoculated with either 105 (BCL1) or 2 × 105 (A31) tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). (This treatment schedule is shown in the inset.) Treatment was as indicated: PBS (●), anti-huCD64 F(ab')2 (⋄), anti-Id F(ab')2 (■), a mixture of anti-Id and anti-huCD64 F(ab')2 (♦), [huCD64 × Id] (○). Symbol labeling on plots shows antibody specificity. Anti-Id Ab preparations were selected for reaction with A31 or BCL1 Id Ig, as appropriate. Survival was recorded daily. Only cohorts treated with [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P < .02).
We next asked whether the nontumor-specific derivatives ([huCD64 × CD19] or [huCD64 × MHC II] BsAb) were also active in vivo. Figure 4 shows that these derivatives conferred a small survival advantage over control cohorts that was statistically significant in the BCL1 model (P < .02) but not in A31. However, the degree of protection observed was considerably less than that given by [huCD64 × Id] BsAb and, consequently, did not correlate with the derivatives' in vitro activity. In particular, the [huCD64 × MHC II] BsAb, which was exceptionally potent in vitro, showed surprisingly little activity in these therapeutic models. This relative lack of in vivo efficacy did not result from giving insufficient [huCD64 × MHC II] BsAb because protection was not improved even when the dose was increased to 1 mg/mouse (not shown). The possibility that [huCD64 × MHC II] BsAb was eradicating an important population of MHC II–expressing effector cells was also investigated using a mixture of [huCD64 × MHC II] and [huCD64 × Id] BsAb. In these experiments the Id-directed BsAb still displayed full therapeutic activity, suggesting that this was not occurring (not shown).
Immunotherapy of lymphoma-bearing mice with BsAb directed to Id, MHC II, or CD19 on the tumors.
Groups of 5 huCD64+ age-matched mice received a treatment schedule as shown in Figure 3. Treatment was as indicated: PBS (●); [huCD64 × CD19] (▵); [huCD64 × MHC II] (■); [huCD64 × Id] (○). Survival was recorded daily. In the A31 model all treatment modalities resulted in a statistically significant increase in survival over PBS-treated controls (P < .02), though only [huCD64 × Id] BsAb–treated cohorts showed long-term survival. Similar results were obtained in at least 3 similar experiments.
Immunotherapy of lymphoma-bearing mice with BsAb directed to Id, MHC II, or CD19 on the tumors.
Groups of 5 huCD64+ age-matched mice received a treatment schedule as shown in Figure 3. Treatment was as indicated: PBS (●); [huCD64 × CD19] (▵); [huCD64 × MHC II] (■); [huCD64 × Id] (○). Survival was recorded daily. In the A31 model all treatment modalities resulted in a statistically significant increase in survival over PBS-treated controls (P < .02), though only [huCD64 × Id] BsAb–treated cohorts showed long-term survival. Similar results were obtained in at least 3 similar experiments.
As expected no therapeutic activity was observed with any of the BsAb in non-Tg littermates (with or without G-CSF) or in Tg mice that had not received cytokine treatment. Figure 5shows an example of these controls for the BCL1 model, with the data from Tg and non-Tg mice treated with or without G-CSF and either IgG or BsAb anti-Id. At the antibody level used (50 μg/mouse total) the IgG anti-Id was not therapeutic in any of the mice, but the BsAb protected huCD64 Tg mice given G-CSF. These results confirm the importance of huCD64 expression to BsAb activity and, interestingly, show for the first time that the BsAb is more active in vivo than IgG mAb.
Only huCD64 Tg mice treated with G-CSF are protected by [huCD64 × Id] BsAb.
Groups of 5 age-matched mice received a treatment schedule as shown in Figure 3 using BCL1 lymphoma cells. However, mice were either huCD64 Tg (c, d) or nonTg littermates (a, b) that were (b, d) or were not (a, c) given 10 days of recombinant G-CSF. Antibody-treatment was as indicated: PBS (●), anti-Id IgG (▵), [huCD64 × Id] (○). Survival was recorded daily. Similar results were obtained with the A31 model, but in this case the IgG anti-Id gave approximately 50 days protection in all groups (not shown).
Only huCD64 Tg mice treated with G-CSF are protected by [huCD64 × Id] BsAb.
Groups of 5 age-matched mice received a treatment schedule as shown in Figure 3 using BCL1 lymphoma cells. However, mice were either huCD64 Tg (c, d) or nonTg littermates (a, b) that were (b, d) or were not (a, c) given 10 days of recombinant G-CSF. Antibody-treatment was as indicated: PBS (●), anti-Id IgG (▵), [huCD64 × Id] (○). Survival was recorded daily. Similar results were obtained with the A31 model, but in this case the IgG anti-Id gave approximately 50 days protection in all groups (not shown).
To investigate this latter observation further, we compared the efficacy of BsAb and IgG anti-Id mAb in the BCL1 model using BsAb and IgG anti-Id at a range of doses. Figure6 shows the results for cohorts of huCD64-Tg mice given G-CSF, 105 BCL1, and then treated with 1 μg to 1000 μg anti-Id IgG or [huCD64 × Id] BsAb. These results underscore the superior activity of BsAb in mice. This is most clearly seen with 1000 μg (Figure 6D) where, though the anti-Id IgG provided approximately 25 days of protection, treatment with the BsAb resulted in long-term survival for all animals. Similar observations were found in the A31 model (data not shown).
Comparison of the immunotherapy achieved against BCL1 with anti-Id IgG and BsAb.
Groups of 5 huCD64+ age-matched mice received the treatment schedule shown in Figure 3. However, mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of (a) 0.1, (b) 1, (c) 10, or (d) 100 μg mAb or BsAb intraperitoneally on days 1 to 5 (either 1, 10, 100, or 1000 μg in total/mouse). Treatment was as indicated: PBS (●), anti-Id IgG (▵), or [huCD64 × Id] BsAb (○). Symbols on plots show antibody specificity. Survival was recorded daily, and similar results were obtained in at least 3 similar experiments.
Comparison of the immunotherapy achieved against BCL1 with anti-Id IgG and BsAb.
Groups of 5 huCD64+ age-matched mice received the treatment schedule shown in Figure 3. However, mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of (a) 0.1, (b) 1, (c) 10, or (d) 100 μg mAb or BsAb intraperitoneally on days 1 to 5 (either 1, 10, 100, or 1000 μg in total/mouse). Treatment was as indicated: PBS (●), anti-Id IgG (▵), or [huCD64 × Id] BsAb (○). Symbols on plots show antibody specificity. Survival was recorded daily, and similar results were obtained in at least 3 similar experiments.
Tumor tracking in vivo
Because there were discrepancies between the cytotoxic activity of BsAb in vitro compared with their in vivo therapeutic effect, we next investigated their in vivo activity more closely. Tumor tracking experiments were set up to measure the early and late activity of each derivative in the peritoneal cavity and spleen. To monitor the tumor cells, it was necessary to inoculate mice with 107BCL1 cells/animal, and consequently the dose of BsAb was increased (500 μg/mouse from days 1-5). Early therapeutic activity in the peritoneal cavity was measured by detecting tumor cells in PBS lavage using flow cytometry. Figure 7represents a typical set of observations. Interestingly, in the first 24- to 48-hour period after treatment, all 3 BsAb ([huCD64 × Id], [huCD64 × CD19], and [huCD64 × MHC II]) were able to dramatically deplete the peritoneal tumor load to less than 1% of that in controls. The CD19-specific BsAb appeared the least active, requiring 48 hours to deplete the peritoneal cavity. Tumor cell depletion was not observed in non-Tg littermates (data not shown), again indicating the requirement for huCD64. Both the anti-Id and anti-MHC II derivatives were particularly potent, showing maximal reduced tumor volume by 24 hours.
In vivo tracking of BCL1 tumor in peritoneal cavity.
HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and throughout the course of therapy. Mice were then inoculated with 107BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse total). One animal per group was killed each day of therapy, and the percentage tumor cells in the peritoneum was analyzed using 2-color flow cytometry with PE-labeled anti-Id and FITC-labeled anti-CD22. In mice treated with [huCD64 × Id] BsAb, tumor cells were detected using FITC-labeled anti-CD22 and PE-labeled anti-CD19 (see “Materials and methods”). The graph shows the percentage of tumor cells in the peritoneal cavity present on each day of therapy after treatment with PBS (●), [huCD64 × Id] BsAb (○), [huCD64 × MHC II] BsAb (■), or [huCD64 × CD19] BsAb (▵). Symbols on plots show BsAb specificity. The inset shows scatter profiles of tumor (gated) on day 5, from mice receiving control PBS (top) or anti-MHC II BsAb (bottom). Similar results were obtained in at least 2 similar experiments.
In vivo tracking of BCL1 tumor in peritoneal cavity.
HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and throughout the course of therapy. Mice were then inoculated with 107BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse total). One animal per group was killed each day of therapy, and the percentage tumor cells in the peritoneum was analyzed using 2-color flow cytometry with PE-labeled anti-Id and FITC-labeled anti-CD22. In mice treated with [huCD64 × Id] BsAb, tumor cells were detected using FITC-labeled anti-CD22 and PE-labeled anti-CD19 (see “Materials and methods”). The graph shows the percentage of tumor cells in the peritoneal cavity present on each day of therapy after treatment with PBS (●), [huCD64 × Id] BsAb (○), [huCD64 × MHC II] BsAb (■), or [huCD64 × CD19] BsAb (▵). Symbols on plots show BsAb specificity. The inset shows scatter profiles of tumor (gated) on day 5, from mice receiving control PBS (top) or anti-MHC II BsAb (bottom). Similar results were obtained in at least 2 similar experiments.
Despite this dramatic reduction in the number of tumor cells recovered during the early course of tumor development, with one exception, all groups of mice had developed a large splenic tumor load by day 15 (Figure 8). The exception was the group treated with [huCD64 × Id] BsAb. By day 15 these animals contained relatively few splenic tumor cells; furthermore, in parallel experiments, it was found that they survived significantly longer than controls despite having received an inoculum of 107 tumor cells (not shown).
In vivo tracking of BCL1 tumor in the spleen.
HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and continuing throughout the course of therapy (10 days in total). Mice were then inoculated with 107 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse in total). One animal per group was killed on days 5, 10, and 15, and the percentages tumor cells in spleen were determined using flow cytometry with PE-labeled anti-Id versus FITC-labeled anti-CD22. Shown are dot plots from a representative mouse at days 10 and 15, treated with BsAb of the specificities indicated. Control BsAb was the nonbinding [huCD64 × A31 Id].
In vivo tracking of BCL1 tumor in the spleen.
HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and continuing throughout the course of therapy (10 days in total). Mice were then inoculated with 107 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse in total). One animal per group was killed on days 5, 10, and 15, and the percentages tumor cells in spleen were determined using flow cytometry with PE-labeled anti-Id versus FITC-labeled anti-CD22. Shown are dot plots from a representative mouse at days 10 and 15, treated with BsAb of the specificities indicated. Control BsAb was the nonbinding [huCD64 × A31 Id].
Calcium signaling after hyper-cross-linking of BsAb
Several reports indicate that cross-linking the B-cell antigen receptor (BCR) with anti-Ig on various types of B cell generates antiproliferative signals leading to cell cycle arrest (CCA) or apoptosis and that these require the release of intracellular calcium.6 34 To determine whether [huCD64 × Id] BsAb was capable of causing similar responses in mouse tumor cells, we next measured intracellular calcium fluxes after short-term treatment in culture. The variant cell line, πBCL1, was labeled with Indo-1-Am and then exposed to BsAb at a final concentration of 2 μg/mL before measuring calcium mobilization by flow cytometry. Figure 9shows calcium fluxes in cells cultured with the various BsAb derivatives either alone or when cross-linked by polyclonal anti-Ig serum. As expected, without cross-linking none of these univalent derivatives was capable of generating a calcium flux. However, whereas in the presence of a polyclonal anti-Ig serum the [huCD64 × Id] BsAb–treated cells showed a sharp mobilization of intracellular calcium, the [huCD64 × MHC II] and [huCD64 × CD19] derivatives were still without effect.
Measurement of the levels of intracellular free calcium in lymphoma cells treated with BsAb.
πBCL1 were loaded with Indo-1-am fluorescent dye, warmed to 37°C for 5 minutes, and then incubated with either BsAb alone (2 μg/mL) or BsAb (2 μg/mL) plus appropriate polyclonal anti-IgG antibody (20 μg/mL), and analyzed immediately using a FACS Vantage. Shown are plots of FL5/ FL4 versus time. An increase in the ratio of FL5/FL4 indicates an increase in the level of intracellular free calcium. BsAb specificity is as indicated. Control BsAb was the non-binding [huCD64 × A31 Id]. Similar results were obtained in at least 3 separate experiments.
Measurement of the levels of intracellular free calcium in lymphoma cells treated with BsAb.
πBCL1 were loaded with Indo-1-am fluorescent dye, warmed to 37°C for 5 minutes, and then incubated with either BsAb alone (2 μg/mL) or BsAb (2 μg/mL) plus appropriate polyclonal anti-IgG antibody (20 μg/mL), and analyzed immediately using a FACS Vantage. Shown are plots of FL5/ FL4 versus time. An increase in the ratio of FL5/FL4 indicates an increase in the level of intracellular free calcium. BsAb specificity is as indicated. Control BsAb was the non-binding [huCD64 × A31 Id]. Similar results were obtained in at least 3 separate experiments.
Lymphoma treatment with [CD64 × Id] BsAb leads to T-cell immunity
A surprising feature of the current work with [huCD64 × Id] BsAb was the lack of Id-negative variants emerging in long-term survivor mice. Such escape variants have been a common feature and a disadvantage of anti-Id mAb therapy since it was first investigated in the early 1980s.35 36 To examine whether this is related to the establishment of long-term immunity, we next rechallenged mice that had survived 100 days or more after tumor and [huCD64 × Id] with 104 fresh lymphoma cells (intraperitoneally). To date, 7 mice have been rechallenged (2 with BCL1 and 5 with A31), and, interestingly, unlike naive mice that developed lymphoma as normal, all the “BsAb-cured” mice were completely resistant to this rechallenge and remained disease free for another 100 days.
To determine the importance of T cells in the early phases of the BsAb therapy, we next tested [huCD64 × Id] therapy in mice depleted of CD4 or CD8 T cells, or both. The results clearly show (Figure 10) that though 70% of T-cell–intact mice were protected from BCL1 by this BsAb, depletion of the CD4 or CD8, or both, removed almost all its therapeutic efficacy. It is important to note that in this system, BCL1 lymphoma develops with the same kinetics in T-cell–depleted mice as in intact mice.8 Thus we can conclude that [huCD64 × Id] therapy is dependent on both CD4 and CD8 cells for its long-term therapeutic activity. This is an important observation because it points to a likely explanation for the potency of [huCD64 × Id] compared to the other BsAb and the anti-Id IgG, which are unable to initiate effective T-cell responses.
Therapeutic efficacy of [huCD64
× Id] BsAb against BCL1 is dependent on CD4 and CD8 T cells. This figure represents a composite of 2 identical independent experiments. For each experiment, groups of 5 huCD64+, age-matched mice received 2 μg/d murine G-CSF subcutaneously 4 days before tumor inoculation and throughout the course of therapy (10 days in total). Mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). To deplete T cells, mice received either anti-CD8 (YTS169; 0.5 mg), anti-CD4 (GK1.5; 1 mg) or a mixture of both YTS169 (0.5 mg) and GK1.5 (1 mg) intraperitoneally 1 day before tumor and again on days 2 and 5 after the tumor inoculation. (The full treatment schedule is shown in the inset.) Treatments were as indicated: PBS (●), anti-CD4 + [huCD64 × Id] (▵), anti-CD8 + [huCD64 × Id] (▴), a mixture of anti-CD4 and anti-CD8 + [huCD64 × Id] (♦), control rat IgG + [huCD64 × Id] (○). Survival was recorded daily. Only cohorts treated without T-cell depletion and [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P ≤ .01).
Therapeutic efficacy of [huCD64
× Id] BsAb against BCL1 is dependent on CD4 and CD8 T cells. This figure represents a composite of 2 identical independent experiments. For each experiment, groups of 5 huCD64+, age-matched mice received 2 μg/d murine G-CSF subcutaneously 4 days before tumor inoculation and throughout the course of therapy (10 days in total). Mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). To deplete T cells, mice received either anti-CD8 (YTS169; 0.5 mg), anti-CD4 (GK1.5; 1 mg) or a mixture of both YTS169 (0.5 mg) and GK1.5 (1 mg) intraperitoneally 1 day before tumor and again on days 2 and 5 after the tumor inoculation. (The full treatment schedule is shown in the inset.) Treatments were as indicated: PBS (●), anti-CD4 + [huCD64 × Id] (▵), anti-CD8 + [huCD64 × Id] (▴), a mixture of anti-CD4 and anti-CD8 + [huCD64 × Id] (♦), control rat IgG + [huCD64 × Id] (○). Survival was recorded daily. Only cohorts treated without T-cell depletion and [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P ≤ .01).
Discussion
In this work we have shown for the first time that huCD64-directed F(ab')2 BsAb can efficiently eradicate lymphoma cells and generate T-cell immunity in tumor-bearing mice and that such reagents are more potent than IgG mAb. Furthermore, with one reagent, [huCD64 × Id] BsAb, long-term survival was reproducibly achieved in 2 B-cell lymphomas (BCL1 and A31)—a goal that has not been reached with anti-Id IgG mAb.7 G-CSF increased the expression of huCD64 on PMN cells in a manner similar to that seen with human PMN28 and was an absolute requirement for therapeutic activity by BsAb. G-CSF was not inherently tumoricidal, as has been seen in some other tumor models.37 We have not yet shown whether this cytokine activation is needed simply to increase the expression of huCD64 on myeloid cells or if it is also important to increase the cytotoxic potency of this or an unidentified population of effector cells. Interestingly, in attempting to understand the mechanisms by which BsAb operate, we observed little correlation between their cytotoxic activity in vitro and the levels of tumor protection they provide. Thus, in vitro [huCD64 × MHC II] BsAb was the most effective for recruiting cytotoxic PMN in RCC assays and resulted in more than 90% specific 51Cr-release, yet in vivo this derivative resulted in only a modest survival advantage. This was surprising because in 5-day tumor-tracking experiments, all 3 BsAb constructs were able to clear tumor cells effectively in vivo. However, only the [huCD64 × Id] provided long-term protection and prevented development of lymphoma. BsAb directed at CD19, which is expressed less strongly than MHC II, did not perform as well in vitro or in vivo. This is consistent with the idea that BsAb, like IgG mAb, are more effective when recruiting against targets that are highly expressed on the tumor cells.7 The failure of [huCD64 × MHC II] BsAb to provide long-term protection to tumor-bearing mice cannot be fully explained, but it suggests that treatment over a short period (5 days) may not be the most appropriate regimen. Interestingly, its lack of activity does not appear to result from inadequate BsAb to coat the tumor cells because increasing the dose to 1 mg did not overcome this limitation (not shown), and further work is underway to optimize the treatment regimen.
In view of the clinical success achieved with IgG anti-Id mAb in patients with non-Hodgkin lymphoma38 and the potency of [CD3 × Id] BsAb in controlling B-cell lymphoma in mouse models,39 the therapeutic activity of the [huCD64 × Id] BsAb derivatives reported here is perhaps not surprising. Accumulating evidence suggests that much of the success of IgG anti-Id mAb relates to their ability to generate antiproliferation signals.5,6 Extensive literature shows that engaging the BCR with anti-Id, anti-IgM, or antigen itself can trigger a cascade of biochemical changes that control the growth of normal and neoplastic B cells.4,40,41 Given its relative in vitro and in vivo potency and the unique ability of [huCD64 × Id] BsAb to mobilize intracellular calcium, we propose that this reagent probably operates by a number of mechanisms, including RCC, common to all 3 BsAb; direct growth control through transmembrane signaling of the BCR; and the induction of T-cell immunity (below). The relative contribution of these processes remains to be determined. However, a BsAb, because it is univalent for its target molecules on the effectors and tumor cells, can only generate a transmembrane signal(s) when it is cross-linked by multivalent cell surfaces. Thus, to achieve the signaling component of their therapeutic activity, BsAb directed at the Id require effector cells, not only for their cytotoxic capacity but also as multivalent surfaces to provide hyper-cross-linking. Interestingly, the fact that F(ab')2 fragments from anti-Id mAb are inactive in vivo (Figure 3) indicates that the degree to which neighboring BCR molecules are cross-linked at the cell surface is critical for triggering such growth control and suggests that both BsAb and IgG anti-Id derivatives require effector-cell cross-linking to achieve therapeutic activity.42
The current data show that in these models at least, BsAb ([huCD64 × Id]), in combination with G-CSF, is more therapeutically active than anti-Id IgG. The explanation for such potency seems to lie in the ability of this BsAb to generate a potent T-cell response. Mice that had achieved long-term survival after BsAb treatment were resistant to tumor rechallenge 100 days later, indicating that the immunity was robust and long lasting. Furthermore, depletion of either the CD4 or the CD8 cells before and during BsAb therapy removed almost all the efficacy of the BsAb. This is a very exciting development that has not been observed with other anti-Id–based therapies. Why the [huCD64 × Id] BsAb has this unique activity is unclear and is the subject of intensive ongoing investigations. This finding has brought forth a wealth of new questions relating to the mechanisms leading to immunity and the identity of the tumor antigens being recognized. Although CD64 has been shown to be highly active in targeting antigens for immunization,43 in the current work it does not automatically lead to tumor immunity because the 2 other CD64-specific BsAb did not show such activity. Therefore, we can conclude that immunization is dependent on the anti-Id specificity (Figure 9). One possibility is that [huCD64 × Id] BsAb signals through the BCR and induces a form of cell death in the lymphoma that is conducive to a T-cell response. For example, because anti-Id based reagents are likely to induce extensive apoptosis of lymphoma cells, these are probably engulfed and then cross-primed by antigen-presenting cells, such as activated dendritic cells, leading to the subsequent antitumor CTL response.44 45 In contrast, perhaps the other BsAb are less apt to induce apoptosis of target cells in vivo but promote direct phagocytosis by CD64 with or without myeloid cells. Finally, we should not overlook the possibility that cross-linking the BCR with BsAb, by triggering in a manner somewhat akin to multivalent antigen, probably results in a surge of B-cell lymphoma-derived cytokines and other immunologic factors, all of which will create a general inflammatory milieu conducive to a T-cell response. The fact that [huCD64 × Id] cleared most of the lymphoma cells from the peritoneal cavity in 24 hours (Figure 7) suggests that if the lymphoma does release inflammatory mediators, the opportunity for this to occur is very short.
In addition to explaining why [huCD64 × Id] is more potent than the other BsAb, we must also consider its activity relative to that of the anti-Id IgG. The anti-Id IgG consistently gives a modest protection of lymphoma-bearing mice, but it was unable to provide long-term T-cell, protection. There are a number of reasons why the anti-Id IgG would not be as active as the BsAb in this respect. First, both anti-Id IgG reagents (A31 and BCL1) were of the rat IgG2a isotype, which is known not to be particularly effective at binding to mouse FcR. Second, because the BsAb binds to an epitope on the FcγR that is outside the IgG binding site, it is not blocked in vivo by endogenous IgG.12 Third, it is probable that, unlike bivalent IgG, the univalent BsAb will remain at the cell surface without inducing modulation of the BCR until cross-linked by effector cells.46 Finally, the BsAb, unlike IgG reagents, will only engage the selected trigger FcR and not inhibitory FcR (eg, FcγRIIB).9 10
For the future, huCD64-directed BsAb in conjunction with the appropriate activating cytokine appears a highly flexible and effective treatment option for lymphoma and probably other neoplastic diseases. The current work underscores the importance of target selection and suggests that molecules that trigger appropriate transmembrane signals in the target may be more effective than those simply able to recruit cytotoxic effectors. Whatever mechanisms BsAb use in vivo, including recruitment of cytotoxic effectors, cross-linking of signaling molecules, and potentiation of host antitumor responses, it appears they are considerably more active than unmodified IgG mAb. Other candidate molecules that might provide interesting targets on NHL are CD79α, CD79β,47 and CD20.2 CD79α/β are the immune-receptor tyrosine-based activation motif (ITAM)-containing signaling molecules that associate with surface immunoglobulin and make up the BCR complex. CD79-directed antibodies are attractive for immunotherapy because they are B-cell specific and should provide the same signaling as anti-Id mAb. Unlike Ig however, CD79 is not secreted into extracellular fluid, does not undergo genetic mutation, and, most important, is not patient specific. Similarly, growing evidence now points to an important signaling component in the activity of CD20.42Unfortunately, anti-CD20 mAb reagents do not exist for the mouse, so such questions cannot be tested in these syngeneic lymphoma models. Given that anti-CD20 and anti-Id mAb have proved the most promising therapeutic antibodies used in non-Hodgkin lymphoma to date,2,5 7 and that in the current work BsAb was more potent than equivalent IgG mAb, [huCD64 × CD79] and [huCD64 × CD20] BsAb appear interesting constructs for future work.
Acknowledgments
We thank all the members of the Tenovus Cancer Laboratory, who provided expert technical support. We thank Dr Mark Cragg, Dr Ruth French, Dr Heidi van Ojik, and Dr Aymen Al-Shamkhani for valuable input and discussion. We also thank J. Andresen (Amgen) for generously providing recombinant murine G-CSF.
Supported by the European Union, Tenovus of Cardiff, the Cancer Research Campaign, and the Leukaemia Research Fund. T.V. is funded by Deutsche Forschungsgemeinschaft (Va 124/1-3).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Martin J. Glennie, Tenovus Laboratory, Cancer Sciences Division, School of Medicine, General Hospital, Tremona Rd, Southampton, SO16 6YD United Kingdom; e-mail: mjg@soton.ac.uk.

![Fig. 3. Immunotherapy of A31- or BCL1-bearing mice with BsAb directed against huCD64 on the effector cells and Id on the tumors. / Groups of 5 huCD64+ age-matched mice received 2 μg/d murine G-CSF subcutaneously, 4 days before tumor inoculation, and throughout the course of therapy (10 days in total). Mice were inoculated with either 105 (BCL1) or 2 × 105 (A31) tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). (This treatment schedule is shown in the inset.) Treatment was as indicated: PBS (●), anti-huCD64 F(ab')2 (⋄), anti-Id F(ab')2 (■), a mixture of anti-Id and anti-huCD64 F(ab')2 (♦), [huCD64 × Id] (○). Symbol labeling on plots shows antibody specificity. Anti-Id Ab preparations were selected for reaction with A31 or BCL1 Id Ig, as appropriate. Survival was recorded daily. Only cohorts treated with [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P < .02).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347003.jpeg?Expires=1769110667&Signature=WfY~s9FS5f5~vset1z77oAS8LYOfZNrle8z6eFeEDWS~IQ5jmj3OzNYfnTtQJjMZ00ZMYBLMSQsp0qf2bcvACFsAxrplRi1JiFcyL~hT~vDZPLMiVYVA0XyJpsEd7hab~i0siRpV3mfI3G1FYeRfTLhE9HALW~kVhtRM6OL-8sJk9KM2MlfQ8M-j5Tu1NuLDdnkOInQCVyQYbCNgy0hl2199g96LXXeakmJjsTB83UyBhZTzcNixmzMuFaL4pZZgYrpxbf6dtoQM9WHhczd6PCxtq9S2AoMKEA~hRgD3urvi-Laqgup0mI0THXLzE8t--uNgDCr70O91CQRmpdPhWg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 4. Immunotherapy of lymphoma-bearing mice with BsAb directed to Id, MHC II, or CD19 on the tumors. / Groups of 5 huCD64+ age-matched mice received a treatment schedule as shown in Figure 3. Treatment was as indicated: PBS (●); [huCD64 × CD19] (▵); [huCD64 × MHC II] (■); [huCD64 × Id] (○). Survival was recorded daily. In the A31 model all treatment modalities resulted in a statistically significant increase in survival over PBS-treated controls (P < .02), though only [huCD64 × Id] BsAb–treated cohorts showed long-term survival. Similar results were obtained in at least 3 similar experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347004.jpeg?Expires=1769110667&Signature=oSuVKSSDSMUBZD9P9eOj7lrRMUZrkAbJO2dWPqmUkVN~5jB49OyP2ry~GGE2g0HFpsHSXk3DMXVILSRm3vPsbZQ-2Zi6psuFIxiN51lfGkURCAkQaN6a7WMx5bjTL15otRPQLYCnUl~X4u~e8h5fa5CBka4kFAXJVPRfEC66YlM8Sw-aROHt8~zH0F2WJPfUiTPztKkqHxbvnDV0a3vEg9ypgN7n9mWkRa8qwrF9hqQePaBdiL0JFg36yMh6tJa0-jq~fwVB5zjDUmwJDK1RLAfNka-xtNQwGcycJHq8pFCF~rn-eICvKx2c1swZLKrlWC-aqQXHO3~Ai9Jto2ExZw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 5. Only huCD64 Tg mice treated with G-CSF are protected by [huCD64 × Id] BsAb. / Groups of 5 age-matched mice received a treatment schedule as shown in Figure 3 using BCL1 lymphoma cells. However, mice were either huCD64 Tg (c, d) or nonTg littermates (a, b) that were (b, d) or were not (a, c) given 10 days of recombinant G-CSF. Antibody-treatment was as indicated: PBS (●), anti-Id IgG (▵), [huCD64 × Id] (○). Survival was recorded daily. Similar results were obtained with the A31 model, but in this case the IgG anti-Id gave approximately 50 days protection in all groups (not shown).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347005.jpeg?Expires=1769110667&Signature=tqOCwSeYL2pOD5LDEeQDVVtN8AxwWuri8QP90gFRRY5pVtCeamxUEhlSJPyNLyDkWkmXeCc323IdYeWGgEc7TqvZB2Y43X-BwlpV5W2ULmVu4QuLdzkof9kDyy39sWyPotYwjvezMcx690KFfXoRlz2k75k5nignioRGyrZ41O0ETt1K~Rw8ZQtuRr9yvxqwx2gvWXgQZ2Ib4EJHY8o3Ja~FA3abHHTuLeuXt1qTcWnfVr0sNf9ND0H-2XauFNw36eCySpJqBQwNX9YE9mqkdgWOLc0Ab~zCZaHqZxeGmviENmRNQ9ikB1plCxDJ3ffX6uDxlPhxcZ5ZjX0CsDm95g__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 6. Comparison of the immunotherapy achieved against BCL1 with anti-Id IgG and BsAb. / Groups of 5 huCD64+ age-matched mice received the treatment schedule shown in Figure 3. However, mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of (a) 0.1, (b) 1, (c) 10, or (d) 100 μg mAb or BsAb intraperitoneally on days 1 to 5 (either 1, 10, 100, or 1000 μg in total/mouse). Treatment was as indicated: PBS (●), anti-Id IgG (▵), or [huCD64 × Id] BsAb (○). Symbols on plots show antibody specificity. Survival was recorded daily, and similar results were obtained in at least 3 similar experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347006.jpeg?Expires=1769110667&Signature=BqehHY7BKcMv5W7FTb~iubRhTj8G1ONMt6dT4CR0DGzN21jFQMHLvMJNACDXUoQqcerNzFlsqwRft70UtjMDdXh1c0klI3GgIbT~EFcv2EyNSCG8Noy7B2MZ51DlTqVWqQV0zCrQAYSTb-~HHM6-kgNWvZwBFeWJVG4CbQFCA9x3Q9cS1MR45FpVGKZdmzLVT0HD4c50RQnrzMilq2PmdvNpbAp49rQ50CCJUDXrzjSPzLBfPo9aJ-XkwWGe6BwNnzcs-u7nOqFUMIb~AIe0h2~tq8dhUe8ZZd5u-N9h9~B1c4-rYLayNbtlHwXjA7XnMtREFvGvYz-hETyqvYwKuQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 7. In vivo tracking of BCL1 tumor in peritoneal cavity. / HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and throughout the course of therapy. Mice were then inoculated with 107BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse total). One animal per group was killed each day of therapy, and the percentage tumor cells in the peritoneum was analyzed using 2-color flow cytometry with PE-labeled anti-Id and FITC-labeled anti-CD22. In mice treated with [huCD64 × Id] BsAb, tumor cells were detected using FITC-labeled anti-CD22 and PE-labeled anti-CD19 (see “Materials and methods”). The graph shows the percentage of tumor cells in the peritoneal cavity present on each day of therapy after treatment with PBS (●), [huCD64 × Id] BsAb (○), [huCD64 × MHC II] BsAb (■), or [huCD64 × CD19] BsAb (▵). Symbols on plots show BsAb specificity. The inset shows scatter profiles of tumor (gated) on day 5, from mice receiving control PBS (top) or anti-MHC II BsAb (bottom). Similar results were obtained in at least 2 similar experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347007.jpeg?Expires=1769110667&Signature=qNGq~SLXMEY2pFlSv9YaUXRj3KuTKzDg3teEQfoM~6MlqOs-PtNjB785RHeHVxovX969ldGT69aKzbHAdbNUOhh0J0ek7Q9SvYJhcCscX9Jya41KUTOeTEd9CSWQa~Yw81AbVwzeJF1roA0L75ViaeHkVhGSoxg5K8yIPWLVJokmkEfVaopNhTjeoMdL8AjxWUUWb2c2fsL0VMJVt1bBTlOZo~htWZWy2X~pXkseXTSKwEmzO33sUuU~Emhz~36UO08Dg0jUf5dH3P10hiAMqVdmV5ppqPFD2YBEFGiul-PDV9QGFnfjatAt6fNcpfstFX3tNhbWOmIzGuHvLx2X0Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 8. In vivo tracking of BCL1 tumor in the spleen. / HuCD64+ mice received 2 μg murine G-CSF/d subcutaneously 4 days before tumor and continuing throughout the course of therapy (10 days in total). Mice were then inoculated with 107 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice daily injections of 50 μg BsAb intraperitoneally on days 1 to 5 (500 μg/mouse in total). One animal per group was killed on days 5, 10, and 15, and the percentages tumor cells in spleen were determined using flow cytometry with PE-labeled anti-Id versus FITC-labeled anti-CD22. Shown are dot plots from a representative mouse at days 10 and 15, treated with BsAb of the specificities indicated. Control BsAb was the nonbinding [huCD64 × A31 Id].](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347008.jpeg?Expires=1769110667&Signature=YtPZSpPwYHCjHMjfv0JDohz86ay69sQFnRnyQ1qPA3OS25ug2M19X-76j-uZLckcK0MEteFtjv7DTwOqnUq7~7J2STNRYtfQ1S4knYdqt2JmAhIvSmIew4lvpMokHax-DTnYIWtWEn9pDQ95HgMhiV9WP4zz9R663JbYuC3c10gFvKm5K2Dv9gRY2uaKe7fEl5izx-UxIZX87vMUB93i3wkEERqfBRf9T4hSOdZV17o9LhQnaO0Hbdlzcz3TkuG61MdXPmaj5VtYF9HAm5vKc~aNj-~FHH-9O9JH1D0K9er~WuEWf5SUxJbzSMKVj5eP9HL9x5D~Fn1XnDxAIQIKWA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 9. Measurement of the levels of intracellular free calcium in lymphoma cells treated with BsAb. / πBCL1 were loaded with Indo-1-am fluorescent dye, warmed to 37°C for 5 minutes, and then incubated with either BsAb alone (2 μg/mL) or BsAb (2 μg/mL) plus appropriate polyclonal anti-IgG antibody (20 μg/mL), and analyzed immediately using a FACS Vantage. Shown are plots of FL5/ FL4 versus time. An increase in the ratio of FL5/FL4 indicates an increase in the level of intracellular free calcium. BsAb specificity is as indicated. Control BsAb was the non-binding [huCD64 × A31 Id]. Similar results were obtained in at least 3 separate experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347009.jpeg?Expires=1769110667&Signature=12JBuWxLEmi3de8JMqTx2fiQOJdHQ259r7oaB2zvnUs9Q6rkPOjoiXHypd8TbMf-Yjmn7QufXgP2T49Vp0SZBlw9Y-MG5~~WEzQB~42p9aXVAHGZ0nUQri91WSwFuVxGdYn9qH~~UxFoYZm-PzJYKeIRd6aXl8z-PMj1hN3ErUHyGblVTYJ1W6qZKXA0Drj8WYUmTgHfuf2ML7sESo3NlJlodCuGHrpWXlKurUmpwm6x3Y5g2rjDhxwUhSNKBljBRI2Az-i7q5LCPSIptb7SnA6JThV6lxlhXcaxt~yZyAyBHCUScZJcCceNwZijPHy72qAyxOg~d7S6wNakslQy0w__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 10. Therapeutic efficacy of [huCD64. / × Id] BsAb against BCL1 is dependent on CD4 and CD8 T cells. This figure represents a composite of 2 identical independent experiments. For each experiment, groups of 5 huCD64+, age-matched mice received 2 μg/d murine G-CSF subcutaneously 4 days before tumor inoculation and throughout the course of therapy (10 days in total). Mice were inoculated with 105 BCL1 tumor cells intraperitoneally on day 0 and were treated with twice-daily doses of 5 μg BsAb intraperitoneally on days 1 to 5 (50 μg/mouse in total). To deplete T cells, mice received either anti-CD8 (YTS169; 0.5 mg), anti-CD4 (GK1.5; 1 mg) or a mixture of both YTS169 (0.5 mg) and GK1.5 (1 mg) intraperitoneally 1 day before tumor and again on days 2 and 5 after the tumor inoculation. (The full treatment schedule is shown in the inset.) Treatments were as indicated: PBS (●), anti-CD4 + [huCD64 × Id] (▵), anti-CD8 + [huCD64 × Id] (▴), a mixture of anti-CD4 and anti-CD8 + [huCD64 × Id] (♦), control rat IgG + [huCD64 × Id] (○). Survival was recorded daily. Only cohorts treated without T-cell depletion and [huCD64 × Id] BsAb showed a significant increase in survival over all control groups (P ≤ .01).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/96/10/10.1182_blood.v96.10.3544/5/m_h82200347010.jpeg?Expires=1769110667&Signature=zXmHEvTgDSjGt6EcE33BGP2QTwRqG-GQHq87yXWR~XiUinIZbPGBio0Nq-c2RvjYvr~jzcjaTa3kiOBIHkKX-~BofPoj0XcIceS9hS2X5bL8wexkeTnQeD6wZzaII5zwL2S-FDqjXb2aMsQ08Q9a8DErGWTs-G~jxGgQQ~1xwaAh1PO5EmVds~T6P56fzag5sWFXwztB6cvF8PRy8tz5onC-LfrXnAtV5WN05tlU~31QHoGIKURmjI53lu-T7e37wwplCsXFKtiPkUjnk2PTN7X38P7CUAv96xwAbPvxw4f2EhBzA1uaHjyiD8zEk8WQiUIqThbEsdI3mUrKLAT1XQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal