Abstract
One hundred consecutive patients with severe aplastic anemia (SAA) received horse antilymphocyte globulin (ALG), cyclosporin A (CyA), 6-methylprednisolone (6Mpred), and granulocyte colony-stimulating factor (G-CSF) as first-line therapy. The median age was 16 years (range, 1-72 years) and median neutrophil count was 0.2 × 109/L (range, 0-0.5 × 109/L). Trilineage hematologic recovery (at a median interval of 96 days from treatment) was seen in 77 patients (48 complete, 29 partial) after 1 (n = 50) or more courses of ALG (n = 27). Of the 23 nonresponders, 11 patients died at a median interval of 83 days (range, 16-1132 days), 6 were considered treatment failures and underwent transplantation, and 6 were pancytopenic. Cytogenetic abnormalities were seen in 11% of patients, clonal hematologic disease in 8%, and relapse of marrow aplasia in 9%. The actuarial survival at 5 years was 87% (median follow-up 1424 days): 76% versus 98% for patients with neutrophil counts less than versus greater than 0.2 × 109/L (P = .001) and 88% versus 87% for patients aged less than versus more than 16 years (P = .8). The actuarial probability of discontinuing CyA was 38%. Patients who did not achieve a white blood cell (WBC) count of 5 × 109/L during G-CSF treatment have a low probability of responding (37%) and a high mortality rate (42%). This update confirms a high probability for SAA patients of becoming transfusion independent and of surviving after treatment with ALG, CyA, 6Mpred, and G-CSF, with a significant effect of neutrophil counts on outcome. Problems still remain, such as absent or incomplete responses, clonal evolution, relapse of the original disease, and cyclosporine dependence. Early transplantation, also from alternative donors, may be warranted in patients with poor WBC response to G-CSF.
Combined immunosuppressive regimens have been shown to be superior to single-drug therapy in the treatment of aplastic anemia. Response rate, but not survival, was improved in patients receiving antilymphocyte globulin (ALG), 6-methylprednisolone (6Mpred), and androgens compared with ALG and 6Mpred alone1; in patients receiving ALG, 6Mpred, and cyclosporin A (CyA) compared with ALG and 6Mpred alone2; and recently in patients with nonsevere disease receiving ALG and CyA compared with CyA alone.3 The combination of ALG, 6Mpred, CyA, and granulocyte colony-stimulating factor (G-CSF) has produced encouraging results in a single-arm pilot study.4 The aim of the present report is to update that study with 100 consecutive patients treated with the 4-drug combination as first-line therapy.
Patients and methods
Treatment
The treatment regimen consisted of horse ALG (IMTIX, Lyon, France) 15 mg/kgd on days 1, 2, 3, 4, and 5; CyA 5 mg/kgd from day 1 for at least 6 months; 6Mpred 2 mg/kgd for 5 days, then halving the dose every 5 days until discontinuation on day +30; and G-CSF (either filgrastim or lenograstim) 5 μg/kgd subcutaneously from day 0 to day +90. Some patients required slow tapering of G-CSF because of declining counts; the median duration of G-CSF treatment was 3 months (range, 1-48 months). Most patients also continued with CyA for many months until recovery, and then started very slow tapering. Supportive care included oral ciprofloxacin during the ALG treatment, oral fluconazole, and transfusion support as required. Patients not achieving hematologic reconstitution could be retreated with a second course of rabbit ALG (Thymoglobulin; IMTIX). This occurred in 27 patients at a median time of 144 days after the first ALG course. Six patients were referred for allogeneic bone marrow transplantation from an identical donor (n = 4) or an unrelated donor (n = 2) and were censored as surviving and nonresponding at the time of transplantation.
Patients
One hundred patients with severe aplastic anemia (SAA) were studied. Clinical characteristics are outlined in Table1. Median age was 16 years (range, 1-72 years), and there were 57 males and 43 females. The median interval from diagnosis to treatment was 22 days (range, 1-460 days). At the time of treatment, the median neutrophil count was 0.2 × 109/L (range, 0-0.5 × 109/L) and the median platelet count was 5 × 109/L (range, 0-40 × 109/L). Thirty-seven patients were infected and 43 had hemorrhages.
Clinical data at the time of treatment
| Characteristic . | . | Range . |
|---|---|---|
| No. of patients | 100 | NA |
| Gender: male/female | 57/43 | NA |
| Age (y) | 16 | 1-72 |
| Neutrophil count (×109/L) | 0.2 | 0-0.5 |
| Platelet count (×109/L) | 5 | 0-40 |
| Infection (yes/no) | 37/58 | NA |
| Hemorrhage (yes/no) | 43/53 | NA |
| Interval from diagnosis to treatment (d) | 22 | 0-460 |
| Response | ||
| No response | 23 | NA |
| Partial response | 29 | NA |
| Complete response | 48 | NA |
| No. of patients with clonal evolution | 8 | NA |
| No. of patients with relapse | 9 | NA |
| Interval from treatment to relapse (d) | 335 | 125-1032 |
| Deaths | 12 | NA |
| Cause of death | ||
| Sepsis | 8 | NA |
| Hemorrhage | 4 | NA |
| Follow-up of surviving patients (d) | 1424 | 81-2889 |
| Follow-up of deceased patients (d) | 84 | 16-1132 |
| Characteristic . | . | Range . |
|---|---|---|
| No. of patients | 100 | NA |
| Gender: male/female | 57/43 | NA |
| Age (y) | 16 | 1-72 |
| Neutrophil count (×109/L) | 0.2 | 0-0.5 |
| Platelet count (×109/L) | 5 | 0-40 |
| Infection (yes/no) | 37/58 | NA |
| Hemorrhage (yes/no) | 43/53 | NA |
| Interval from diagnosis to treatment (d) | 22 | 0-460 |
| Response | ||
| No response | 23 | NA |
| Partial response | 29 | NA |
| Complete response | 48 | NA |
| No. of patients with clonal evolution | 8 | NA |
| No. of patients with relapse | 9 | NA |
| Interval from treatment to relapse (d) | 335 | 125-1032 |
| Deaths | 12 | NA |
| Cause of death | ||
| Sepsis | 8 | NA |
| Hemorrhage | 4 | NA |
| Follow-up of surviving patients (d) | 1424 | 81-2889 |
| Follow-up of deceased patients (d) | 84 | 16-1132 |
NA indicates not applicable.
Participating centers
Centers contributing patients were: Genova Ospedale San Martino (M. T. Van Lint), Torino Clinica Pediatrica (U. Ramneghi), Genova Istituto Gaslini (P. G. Mori), Nuoro Ospedale San Francesco (A. Gabbas), Pavia Policlinico San Matteo (F. Locatelli), Vicenza Ospedale San Bortolo (F. Rodeghiero), Ancona Ospedale Torrette (P. Leoni), Bolzano Ospedale San Maurizio (P. Coser), Brescia Clinica Pediatrica (F. Porta), Monza Clinica Pediatrica (C. Uderzo), Roma Università La Sapienza (W. Arcese), Cagliari Ospedale Businco (G. Broccia), Bruxelles Clinic Pediatrique (A. Ferster), S. G. Rotondo Ospedale Casa Sollievo della Sofferenza (M. Carotenuto), Bergamo Divisione Ematologia (T. Barbui), Roma Università La Sapienza (A. Testi), Bologna Ospedale Sant'Orsola (G. Rosito), Cuneo Divisione Ematologia (A. Gallamini), Firenze Ospedale Pediatrico (A. Lippi), Pescara Ospedale Civile (P. Di Bartolomeo), Reggio Calabria Ospedale Bianchi-Melacrino-Morelli (P. Iacopino), Verona Policlinico Borgo Roma (G. Todeschini), Bari Clinica Pediatrica (D. De Mattia), Besancon Hopital Jean Minjoz (J. Y. Chan), Bristol Hospital Sick Children (D. Pamphilon), Copenaghen Pediatric Clinic II (C. Heilmann), Genova Medicina Interna (M. Gobbi), Milano Ospedale San Raffaele (A. Rugarli), Napoli Ospedale Cardarelli (R. Cimino), Napoli Clinica Pediatrica (S. Perrotta), Nijemegen Pediatric Oncology (M. Boaekkering), Padova Clinica Oncoematologica (P. Rossetti), Monza Ospedale San Gerardo (E. Pogliani), Pisa Divisione Ematologia (G. Caracciolo), Sidney Royal Perth Hospital (P. K. Cannell), Udine Cattedra Ematologia (M. Russo), Lund Dept Pediatric (A. Bekassy).
Responses
Complete responses were defined as transfusion independence associated with hemoglobin (Hb) greater than 11 g/dL, neutrophils greater than 1.5 × 109/L, and platelets greater than 100 × 109/L. We defined partial responses as transfusion independence associated with Hb greater than 8 g/dL, neutrophils greater than 0.5 × 109/L, and platelets greater than 30 × 109/L. Transfusion dependence was taken as evidence of no response.
Relapse
A patient was considered in relapse if he or she had transfusions with red blood cells or platelets after having been independent from transfusions for at least 3 months. Some patients showed declining peripheral blood counts that could be controlled by increasing the dose of CyA and did not require transfusions; these episodes were not recorded as relapses.
Cytogenetic analysis and glycosyl-phosphatidylinositol–anchored proteins (GPI-AP)
Fifty-four patients were followed with cytogenetic analyses from bone marrow aspirates. A small number of patients were also followed for GPI-AP expression on peripheral blood cells, such as CD14 on monocytes and CD16 on neutrophils. The limit for a significant GPI-AP–deficient population was calculated as follows: mean of negative cells in controls ± (2 × standard deviation).5
Statistical analysis
Actuarial survival was calculated according to Kaplan and Meier.6
Results
Response
Trilineage hematologic recovery was seen in 77 patients after 1 (n = 50) or more courses of ALG (n = 27). The median time to transfusion independence was 96 days (range, 7-917 days), and the actuarial probability of becoming transfusion independent at 48 months was 95%. Forty-eight patients (48%) were complete responders and 29 (29%) were partial responders; 1 partial responder died after day +100. There were more male than female responders (85% versus 66%;P = .02). Response was not influenced by neutrophil PMN) counts (73% versus 80%, P = .3, for PMN less than versus greater than 0.2 × 109/L) or by age (77% both in patients aged less than and more than 16 years).
The median maximum white blood cell count (WBC) during G-CSF treatment was 14 × 109/L. The proportion of nonresponders was 63% for patients with maximum WBC counts of less than 5 × 109/L (n = 19), 13% for maximum WBC counts of 5 to 14 × 109/L (n = 24), and 4% for WBC counts exceeding 14 × 109/L (P = .002). The numbers of deaths in the same 3 groups were 8 of 19 (42%), 2 of 24 (8%), and 0 of 44 (0%) (P < .00001). In 13 patients the maximum WBC count was not available, and 2 of these died. Twenty-three patients (23%) showed no recovery. Of these, 11 died (8 by day 100), 6 had transplantation at a median interval of 241 days (range, 81-828 days) and were censored as surviving at the time of transplantation, and 6 were pancytopenic at a median interval of 393 days (range, 329-1929 days).
Quality of response
Peripheral blood counts in partial responders were as follows: Hb 12 g/dL (range, 8-17.6 g/dL), PMN 2.2 × 109/L (range, 0.5-10 × 109/L), and platelets 65 × 109/L (range, 30-169 × 109/L). Peripheral blood counts in complete responders were as follows: Hb 13 g/dL (range, 11-16.2 g/dL), PMN 2.9 × 109/L (range, 2-8.5 × 109/L), and platelets 160 × 109/L (range, 100-387 × 109/L).
Causes of death
Twelve patients died at a median interval of 83 days (range, 16-1132 days). Of these, 3 patients died after developing clonal disease (myelodysplasia = 2, leukemia = 1), 1 in each of the age groups of less than 16, 17 to 45, and more than 45 years. Five patients died of septicemia, 2 of hemorrhage, and 2 of fungal infections.
Cytogenetic analysis and GPI-AP
Cytogenetic analyses were available in 54 patients. Fifty-three had normal cytogenetic analyses at diagnosis; of these, 45 (85%) remained normal, 6 (11%) developed a cytogenetic abnormality, and 2 (4%) had no cytogenetic study available at follow-up. The following abnormalities were recorded: −y (n = 2), −7 (n = 2), 5q− (n = 1), and random deletion (n = 1). The probability of developing a cytogenetic clone was 40% (2/5) in nonresponders, 6% (1/19) in partial responders, and 10% (3/30) in complete responders. One 7-year-old child had a 5q− abnormality at diagnosis, and is now 4 years post-treatment with a normal karyotype. A hematologic clonal disease was diagnosed in 8 patients: 4 myelodysplasias (2 had a −7 abnormality), 2 acute leukemias (1 with random deletions), 1 hairy cell leukemia (possible misdiagnosis), and 1 clinically evident paroxysmal nocturnal hemoglobinuria. The distribution of clonal disease was 3 of 13 in nonresponders (13%), 3 of 29 in partial responders (10%), and 2 of 48 in complete responders (4%).
GPI-AP were tested in 14 patients, and 8 had decreased expression of at least 1 marker either at diagnosis or during follow-up.
Relapse
Nine patients relapsed at a median interval of 335 days from treatment (range, 125-1032 days), with an actuarial probability at 3 years of 12%. Most patients were still taking CyA, and the actuarial probability at 5 years of discontinuing CyA was 38% (Figure1). This suggests that most patients still require immunosuppressive treatment to maintain their peripheral blood counts, especially because some of the relapses were seen in the first patients who discontinued CyA on day +180.
Actuarial probability of discontinuing cyclosporin A (CyA).
Probability was 38% at 5 years, suggesting CyA dependence in many patients.
Actuarial probability of discontinuing cyclosporin A (CyA).
Probability was 38% at 5 years, suggesting CyA dependence in many patients.
Survival
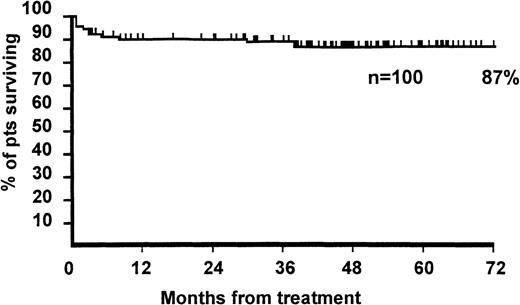
The actuarial survival at 5 years was 87% (Figure2), and the median follow-up for surviving patients was 1424 days (range, 81-2889 days). Survival was 76% versus 98% for patients with neutrophil counts less than versus greater than 0.2 × 109/L (P = .001) and 88% versus 87% for patients aged less than versus greater than 16 years (P = .8). The survival of 22 patients aged 36 years or older was 72%, which was significantly different from the age group of 1 to 35 years (92%) (P = .01). Survival was 92% versus 81% for males versus females (P = .09).
Actuarial survival of 100 patients.
Patients with severe aplastic anemia were treated with antilymphocyte globulin (ALG), cyclosporin A (CyA), 6-methylprednisolone (6Mpred), and granulocyte colony-stimulating factor (G-CSF).
Actuarial survival of 100 patients.
Patients with severe aplastic anemia were treated with antilymphocyte globulin (ALG), cyclosporin A (CyA), 6-methylprednisolone (6Mpred), and granulocyte colony-stimulating factor (G-CSF).
Discussion
This update confirms the high probability of becoming transfusion independent and of surviving for SAA patients after combined treatment with ALG, CyA, 6Mpred, and G-CSF. We have also confirmed that patients who do not achieve a WBC of 5 × 109/L during G-CSF treatment have a low probability of responding (37%) and a high mortality rate (42%). Another significant predictor of survival is the neutrophil count at the time of treatment.1 There is no effect of age up to 35 years, but the 22 patients over 35 years seemed to have a poorer prognosis. Children with very severe aplasia (< 0.2 × 109/L) had an actuarial survival of 82%, which is very encouraging when compared with a previous pediatric study.7
A recent report8 has suggested a high risk (20%) of cytogenetic abnormalities after prolonged administration of G-CSF and CyA in children, especially deletion of chromosome 7, raising the question of the safety of G-CSF administration. In the present study, we had 29 children with normal cytogenetics at diagnosis; 2 children (7%) developed monosomy 7 and myelodysplasia. This is not different from our previous experience9 or from a separate pediatric study.10 The latter study has also shown that careful analysis of patients at treatment may disclose undiagnosed abnormalities.10 Overall, 11% of evaluable patients developed cytogenetic abnormalities and 8% developed clonal disease. The risk was somewhat greater in patients not responding as compared with responders, but there seemed to be no significant difference between partial (10%) and complete responders (4%).
The quality of life in surviving patients was good, with few side effects, although problems remain, such as CyA dependence, clonal evolution, relapse of the aplasia, and persistent cytopenia. Some of these problems are transient; relapse can be treated; reduction in the expression of GPI-AP is not necessarily associated with clinical problems; and cytogenetic abnormalities can disappear. However, a small number of patients will progress to develop hematologic myelodysplasia or leukemia and may die of these disorders. Whether new growth factors or high-dose cyclophosphamide11 will improve hematologic recovery and reduce these complications remains to be determined. Given these drawbacks and the results reported for HLA-identical sibling transplants,12 we suggest first-line bone marrow transplantation in patients up to the age of 40 when an identical sibling is available. On the other hand, survival after alternative donor transplants13-15 is still inferior to results shown in the present study in any age group, making combined immunosuppressive therapy more attractive for patients lacking an identical sibling.
One important question is whether G-CSF is required to achieve the excellent survival shown with the 4-drug combination, and the only way to answer this question is with a prospective randomized trial. One such trial has been conducted and has shown earlier neutrophil recovery and fewer infections, but has failed to show a higher rate of trilineage response or survival in the G-CSF–treated group.16 The number of patients in that trial was small and mortality was higher than reported in this study (19%), despite a shorter follow-up. A second trial is being activated by the European Group for Blood and Marrow Transplantation Working Party on Severe Aplastic Anemia. Although there is no formal proof of a significant advantage for patients receiving G-CSF in association with ALG and CyA, this remains an excellent combination for first-line therapy in SAA patients. In addition, the significant prognostic effect of a WBC increase during G-CSF treatment may allow early identification of patients who are eligible for alternative donor transplants.
Supported by an Associazione Italiana Ricerca Contro il Cancro (A.I.R.C.) Milano grant to A.B. and by Associazione Ricerca Trapianto Midollo Osseo (A.R.I.T.M.O.) Genova.
Reprints:A. Bacigalupo, Divisione Ematologia 2 (PAD 5/II), Ospedale San Martino, Largo Rosanna Benzi 10, 16132 Genova, Italy; e-mail: apbacigalupo@smartino.ge.it.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal