Abstract
Mutations of the ribosomal protein S19 (RPS19) gene were recently identified in 10 patients with Diamond Blackfan anemia (DBA). To determine the prevalence of mutations in this gene in DBA and to begin to define the molecular basis for the observed variable clinical phenotype of this disorder, the genomic sequence of the 6 exons and the 5′ untranslated region of the RPS19 gene was directly assessed in DBA index cases from 172 new families. Mutations affecting the coding sequence of RPS19 or splice sites were found in 34 cases (19.7%), whereas mutations in noncoding regions were found in 8 patients (4.6%). Mutations included nonsense, missense, splice sites, and frameshift mutations. A hot spot for missense mutations was identified between codons 52 and 62 of the RPS19 gene in a new sequence consensus motif W-[YFW]-[YF]-x-R-[AT]-A-[SA]-x-[AL]-R-[HRK]-[ILV]-Y. No correlation between the nature of mutations and the different patterns of clinical expression, including age at presentation, presence of malformations, and therapeutic outcome, could be documented. Moreover, RPS19 mutations were also found in some first-degree relatives presenting only with isolated high erythrocyte adenosine deaminase activity and/or macrocytosis. The lack of a consistent relationship between the nature of the mutations and the clinical phenotype implies that yet unidentified factors modulate the phenotypic expression of the primary genetic defect in families with RPS19 mutations.
DIAMOND BLACKFAN anemia (DBA), or congenital pure red blood cell aplasia, is a rare disorder affecting 4 to 7 children per million live births.1-4 It typically presents in infancy as a regenerative macrocytic anemia and is variably associated with a wide range of physical abnormalities, predominantly craniofacial, but also including thumb, cardiac, and urogenital malformations.2,4-6 Although the anemia is steroid-responsive in 70% of affected children, this response may not be sustained and up to 40% of individuals become eventually dependent on a long-term red blood cell transfusion program.4 In steroid-responders, a macrocytosis and a mild anemia generally persist despite response to therapy,2,7 most often in association with elevated erythrocyte adenosine deaminase (eADA) activity.8,9 DBA is most commonly sporadic, with a clearly positive family history in only 10% to 25% of patients,2,4,5 usually showing an autosomal dominant pattern of inheritance. However, increased eADA activity may also be found in some apparently unaffected individuals from DBA families,8,9 whereas in other cases, a family history may be inferred from a previously unexplained episode of moderate macrocytic anemia during childhood or pregnancy.10 11
It has been uncertain whether DBA, which is clearly heterogeneous with respect to clinical phenotype, represents a single or overlapping group of disorders. Its pathophysiology is still difficult to define, despite extensive in vitro characterization of bone marrow or peripheral erythroid progenitors. Results from in vitro progenitor culture studies are generally consistent with an intrinsic erythroid cell defect predominantly affecting differentiation.7,12-15 Preliminary studies have ruled out a number of potential candidate genes, including those encoding the erythropoietin receptor,16 stem cell factor or its receptor,17-19 and interleukin-9.20 Recently, a balanced translocation, t(X;19), was identified in a DBA patient,21 allowing a first locus for DBA to be assigned to chromosome 19q13.2,22although genetic heterogeneity was shown by the failure of DBA to cosegregate with this locus in some families.23,24 The cloning of the chromosome 19q13 translocation breakpoint showed the translocation to interrupt the gene encoding the ribosomal protein S19 (RPS19). Subsequent analysis identified mutations in the coding sequence of 1 allelle of RPS19 gene in 10 of 40 unrelated DBA patients.25 In the present work, we systematically studied the RPS19 gene in a cohort of 172 DBA families (190 patients) who fulfilled diagnostic criteria for DBA agreed by the DBA Working Group of the European Society for Paediatric Haematology and Immunology (ESPHI).26 We found heterozygosity for mutations affecting the RPS19 gene in 42 of 172 index patients (24.4%), including many mutations that had not been previously described. We were unable to find a mutation in RPS19 gene in the other 76% of families, confirming the genetic heterogeneity of DBA. Mutations in RPS19 gene identified included nonsense, missense, splice site, and frameshift mutations, as well as complete loss of a presumed normal allele. These different mutations were found scattered along the entire span of the RPS19 gene. Interestingly, mutations in RPS19 gene were also found in some apparently unaffected individuals from DBA families, presenting only with an isolated elevation of eADA.9 We also collected extensive clinical and biological features of 216 DBA patients (172 from the present report and the 44 previously reported3,22,23 25). RPS19 gene was mutated in 56 of these 216 patients (26%). The presence or absence of different types of physical anomalies or the various types of long-term therapeutic responses could not be associated with a specific pattern of mutations. The lack of a consistent relationship between the nature of the mutations and the clinical phenotype implies that yet unidentified factors modulate the phenotypic expression of the primary genetic defect in families with RPS19 mutations.
MATERIALS AND METHODS
Patients
To define the frequency and type of mutations in RPS19 gene in association with DBA, 172 patients enrolled by pediatricians or hematologists participating in the ESPHI DBA Working Group were studied. All applicable human experimentation committee approvals were obtained from the participating institutions. The diagnosis of DBA was established on the basis of the finding of aregenerative anemia under the age 2 years, with absence or severe reduction in the number of erythroblasts in bone marrow, according to diagnostic criteria agreed to by the ESPHI DBA Working Group.26 Patients more than 2 years of age at onset of anemia were included only if there was a positive family history of classical DBA or if typical physical anomalies were present.
We also collected clinical and biological characteristics from the 172 families described in the present report as well as the 44 families included in earlier reports.3,22,23 25Phenotypic characterization of index cases included clinical assessment for malformations, age at presentation, response to an initial course of steroids, current treatment, and, if transfusion-independent, current hemoglobin level, mean corpuscular volume (MCV), and eADA activity. Hemoglobin, MCV, and eADA were measured in all available first-degree relatives, and families were questioned about any history of anemia.
Measurement of eADA Activity
eADA activity was determined in hemolysates by a radioisotopic method9,27 or spectrophotometrically.28 To allow comparison of results obtained in different laboratories, results were expressed in terms of standard deviations from the mean normal value obtained for the relevant laboratory. Results below mean + 2 standard deviations (SD) were considered normal.
RPS19 Sequence Analysis
The genomic DNA sequence for the 6 exons and the 450-bp sequence upstream of the first exon (5′UTR) were determined for each DNA sample. Four polymerase chain reaction (PCR) fragments spanning the 5′UTR and the 6 exons were amplified from 200 ng of genomic DNA in 50 μL reactions using Taq polymerase. The PCR primers25 and additional internal primers were used for fluorescent automated DNA sequencing, performed using Applied Biosystems 373 or 377 DNA sequencer and ABI Big Dye Terminator sequencing kits (Perkin Elmer, Foster City, CA).29 All sequence variations identified were verified on the complementary strand using an independent PCR product. When a particular mutation was detected in a patient, its presence was determined in DNA of all other available family members. To confirm that the observed changes in nucleotide sequence found in DBA patients represented mutations rather than polymorphisms, DNA derived from 50 healthy blood bank donors representing 100 unrelated chromosomes was also sequenced for the 5′UTR and the first 5 exons.
Comparative Analysis of RPS19 Sequence
The amino acid sequence of human RPS19 was analyzed for various sequence motifs using ScanProsite Protein against PROSITE (ExPASy; HCU, Geneva, Switzerland). Analysis of secondary and tertiary structures of RPS19 was determined using Hierarchical Neural Network and GOR IV (IBCP, Lyon, France).30,31 Alignment of RPS19 amino acid sequence originating from 20 different species was performed using the software package MultAlin (Corpet F; INRA, Toulouse, France; accession no. of sequences in Appendix A1). High consensus value was set at 90%, and the low consensus value was set at 50%. From the set of 20 protein sequences used, a search for blocks was performed with MATCH-BOX_server 1.2 (Molecular Biology, University of Namur, Namur, Belgium).32 Similarly, the search for motifs from the set of sequences was performed using MEME version 2.2 from Baylor College of Medicine (Houston, TX).33
Statistical Analysis
Descriptive statistics are presented as the percentages and means ± 1 SD. The significance of observed differences was tested using the χ2 statistic and, when more appropriate for small samples, a two-tailed Fisher's exact test. Kruskal-Wallis H nonparametric test was used for comparison of age at diagnosis and prevalence of malformations, because variance within groups appeared to be different. Statistical analysis was performed using Epi-Info 6.04b (Center for Disease Control, Atlanta, GA).
RESULTS
RPS19 Gene Mutations in 172 DBA Index Cases
Using direct sequencing of genomic DNA, we screened for RPS19 mutations in the index cases of 172 families. In 42 of the 172 families studied, 1 of the RPS19 alleles was found to harbor a mutation, defined as a sequence variation not seen in 100 normal chromosomes. Thirty-one of the mutations were in the coding region of the gene, whereas 3 others were at splice sites, 3 were in the first noncoding exon, and 7 were in intronic regions (2 patients each exhibiting 2 mutations). Nonsense mutations generating a premature stop codon were detected in 10 cases (Fig 1). These mutations were restricted to exons 2 to 5, up to codon 94. Missense mutations resulting in an amino acid substitution were noted in 12 cases (Fig 2). Insertions of 1 to 3 nucleotides were found in 2 cases, resulting in a shift of the reading frame in 1 case, with the other presenting with a 3 nucleotides insertion resulting in the addition of a glutamine at codon 19 (Fig 3). Deletions of 1 or 2 nucleotides were found in 6 cases, affecting the reading frame, whereas a larger deletion (31 bp) was found in 1 case (Fig 3). Mutations affecting splice sites were observed in 3 cases.
Predicted amino acid sequence resulting from nonsense mutations. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values.
Predicted amino acid sequence resulting from nonsense mutations. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values.
Predicted amino acid sequence resulting from missense mutation. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate the position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values, whereas bold letters indicate the missense mutations.
Predicted amino acid sequence resulting from missense mutation. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate the position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values, whereas bold letters indicate the missense mutations.
Predicted amino acid sequence resulting from small deletions and insertions. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate the position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values, whereas bold letters indicate the new amino acid sequence resulting from the frameshift.
Predicted amino acid sequence resulting from small deletions and insertions. Consensus sequence was determined according to results of alignment of 20 different species of RPS19 performed with MultAlin (Corpet. F., 1988). ! stands for any of Isoleucine or Valine, % for any of Phenylalanine or Tyrosine, and # for any of Aparagine or Aspartic acid or glutamine or glycine. Upper case letters indicate high consensus values (>90%), and lower case letters indicate low consensus values (between 50% and 90%). Numbers indicate the position along human amino acid sequence. Bold letters in the human wild-type RPS19 sequence indicate the high consensus values, whereas bold letters indicate the new amino acid sequence resulting from the frameshift.
Mutations in the first noncoding exon or in introns were detected in 10 cases. The same 4-bp deletion in exon 1 was found in 3 independent DBA families, but in none of the control chromosomes. In 1 family, this deletion was the only abnormality found, whereas in 2 other sporadic cases, the 4-bp deletion coexisted with other abnormalities of the RPS19 gene, an acceptor splice site defect, and a 31-bp deletion in exon 5, respectively. The other recurrent abnormality found in 4 other independent families was a single nucleotide substitution in intron 1 (G→T at position −460 from the ATG), an abnormality that was not identified in any of the control chromosomes. No promoter site or transcription factor binding site could be identified at either position. In 2 sporadic cases, a single nucleotide substitution was found, respectively, in intron 4 and intron 1. A single base difference in intron 5 was seen in the index case of a dominant family (F5), but not in other affected family members. These mutations in the first exon or in introns may still represent normal polymorphic differences, with their absence in the controls being explained by disparate genetic backgrounds. Sequence analysis of a wider range of controls should resolve this question.
Polymorphisms Affecting the RPS19 Gene
The following variations from published sequence were found in the RPS19 gene of 50 normal individuals: exon 1, in position −550 from the ATG, C in 54% or T in 46% of normal chromosomes; intron 2, starting at position 77 downstream of the end of exon 2, GGT CCC TGG CAG GCG AGG in 54% and GGT CCT GGC AGG GGA GG in 46%; exon 3, 164C→T in 1 chromosome; intron 4, 14 nucleotides downstream of the end of exon 4, A in 50% or G in 50%.
Association Between Mutations and Conserved Sequence Motifs
Distribution of all 42 different mutations along the RPS19 gene reported in the present study as well as the 10 mutations identified in the earlier report25 is summarized in Fig 4. The majority (11/16) of the missense mutations was observed between codons 52 and 62, although this region of the predicted protein did not correspond to any previously identified functional motif. However, the search for amino acid motifs from the alignment of different RPS19 protein sequences showed 3 significant sequence motifs. Motif 1 started at codon 52 in human RPS19, with the multilevel consensus sequence W52-[YFW]-[YF]-[VTILK]-R-[AT]-A-[SA]-[IVLT]-[AL]-R-[HRK]-[ILV]-Y65. Most of the missense mutations (11/16) affected this motif, which was only found in the different RPS19e proteins using various databases. Interestingly, mutations affecting the highly conserved Arg56 and Arg62 residues were seen in 3 and 6 cases, respectively. Motif 2 started at codon 31 of human RPS19 with the following sequence: P31-[EGDQ]-[WY]-[VAIS]-D-[IFTLV]-[VIT]-K-[TLM]-[GAS]-[VAIKMRST]-[HSDFGN]-[KNR]-E-[LMR]-[APRS]-P47. No mutation was identified in this motif. Motif 3 started at codon 120 of human RPS19 with the following sequence: G120-R-[RVKLI]-[ILV]-[TS]-[PKEQS]-[QEKNS]-G-[RQ]-[RKS]-[DF]-L-D-[RK]-[IV]-A135. Two of the missense mutations identified were found within motif 3.
Location of point mutations in RPS19 gene. The position of single nucleotide substitutions or small deletions or insertions in RPS19 gene found in DBA patients is indicated along the structure of the gene. Nucleotide and amino acid numbers are shown at start of exons and at the end of exons. Nucleotide numbers start from the ATG of the gene. Data include results from the present study, as well as mutations described in an earlier report.25 Larger deletions or translocations are not shown in this figure.
Location of point mutations in RPS19 gene. The position of single nucleotide substitutions or small deletions or insertions in RPS19 gene found in DBA patients is indicated along the structure of the gene. Nucleotide and amino acid numbers are shown at start of exons and at the end of exons. Nucleotide numbers start from the ATG of the gene. Data include results from the present study, as well as mutations described in an earlier report.25 Larger deletions or translocations are not shown in this figure.
Phenotype of Patients Studied
A total of 216 families were available for phenotype-genotype correlations analysis, 76 families from France, 56 from Italy, 37 from the United Kingdom, 29 from Germany, and 18 from Sweden. The cohort of index cases (sex ratio: M/F: 1/1.04) exhibited the expected range of DBA phenotypes (Table1). Their median age at presentation was 2.0 months (range, 0 to 240 months) and 41.5% had associated physical anomalies. Sixty-two percent had initially responded to steroids. At the time of the genetic study, the mean age was 151 ± 134 months, 45.1% of the patients were transfusion-dependent, 31.4% were steroid-dependent, and 19% were transfusion-independent with no other treatment, whereas 5 patients had received bone marrow transplantation (BMT; 2.5%) and 4 (2%) were dead.
Clinical Characteristics and Outcome, According to the Type of Mutation of RPS19 Gene in DBA Patients
| Patient (sex) . | Age at Diagnosis of DBA (mo) . | Malformative Status . | Response at First Steroid Course . | Status at Last Follow- Up . | Genomic DNA Mutation . | Expected Protein Alteration . | Inheritance . | Mutation in Other Family Members* . |
|---|---|---|---|---|---|---|---|---|
| Nonsense mutations | ||||||||
| F23 (F) | 1 | Kidney hypoplasia/aplasia, low hair line, short stature | NR | Dead | 34 C → T | Gln12Stop | s | 0 |
| F49 (F) | 2 | Triphalangeal thumb, low implantation of thumbs | NR | TD | 144 C → A | Tyr48Stop | s | 0 |
| F44 (M) | 2 | Hydrocephaly, unique median cerebral ventricule, atrioventricular septal defect, modeling defect of long bones | NR | TD | 99 G → C | Trp33Stop | s | 0 |
| F16 (M) | 2 | None | CR | SD | 34 C → T | Gln12Stop | s | 0 |
| F69 (F) | 22 | Short stature | CR | SD | 166 C → G | Arg56Stop | s | 0 |
| G7 (F) | 2 | Short stature | CR | SD | 280 C → T | Arg94Stop | se | e: 2/3 |
| I6 (M) | 3 | None | CR | SD | 155 G → A | Trp52Stop | s | 0 |
| I7 (M) | 0 | None | NR | TD | 31 C → T | Gln11Stop | s | 0 |
| I8 (F) | 2 | None | NR | TD | 280 C → T | Arg94Stop | d | d: 1/1 |
| I9 (F) | 0 | Congenital bilateral glaucoma | CR | TD | 280 C → T | Arg94Stop | d | d: 2/2 |
| G27 (M) | NA | NA | NA | NA | 280 C → T | Arg94Stop | s | 0 |
| Missense mutations | ||||||||
| F42 (F) | 1 | Short stature | NR | TD | 380 G → A | Gly127Gln | s | 0 |
| F18 (F) | 2 | Triphalangeal thumb, anogenital fistula, interventricular septal defect | NR | TD | 184 C → T | Arg62Trp | s | 0 |
| F32 (F) | 1 | None | PR | TD | 167 G → A | Arg56Gln | s | 0 |
| F28 (M) | 1 | None | PR | TD | 167 G → A | Arg56Gln | s | 0 |
| F7 (M) | 3 | None | NR | TD | 167 G → A | Arg56Gln | se | e: 0/3 |
| G20 (F) | 2 | None | CR | SD | 43 G → T | Val15Phe | s | 0 |
| F70 (F) | 2 | None | NR | TD | 358 G → C | Gly120Ser | s | 0 |
| F12 (M) | 0 | None | CR | CRem | 184 C → T | Arg62Trp | d | d: 2/2 |
| F9 (M) | 0 | None | CR | NA | 1 A → G | Met1Val | d | d: 2/2 |
| G24 (M) | 2 | None | NR | BMT | 184 C → T | Arg62Trp | se | e: 1/1 |
| I10 (M) | 2 | Short stature | NR | Dead | 184 C → T | Arg62Trp | s | 0 |
| I11 (F) | 4 | Duplication of thumb | CR | CRem | 302 G → A | Arg101His | s | h: 1/1 |
| I53 (M) | 0 | Micrognathia, proximal implanted thumbs, short stature | NR | TD | 184 C → T | Arg62Trp | s | 0 |
| S1 | NA | NA | NA | NA | 154 T → C | Trp52Arg | s | 0 |
| E28 (F) | 2 | None | NA | CRem | 182 C → A | Ala61Ser | se | e: 0/1 h: 1/1 |
| S2 (F) | <12 | None | NR | TD | 184 C → T | Arg62Trp | d | d: 1/1 |
| Insertions and deletions | ||||||||
| F31 (M) | 2 | None | NR | TD | ins A between nt 14 and 15 | Frameshift at codon 5 stop at codon 50 | s | 0 |
| F4 (F) | 1 | Hypertelorism, epicanthus, low ear set, antimongoloid eye line | NR | CRem | del 295 bp 356 (+281-+576) | Deletion of exon 5 | de | d: 1/1 e: 1/1 |
| F19 (M) | 2 | Epicanthus, low hairline, short stature | CR | SD | del 1 bp nt 222 | Frameshift at codon 74 stop at codon 75 | s | 0 |
| F26 (M) | 2 | Dysplasia of mitral valve, short stature | NR | TD | ins A between nt 104 and 105 | Frameshift at codon 35 stop at codon 50 | s | 0 |
| F25 (F) | 1 | Short stature | CR | SD | del 31bp (274-314) | Deletion of 31 nt of exon 4, frameshift at codon 92 stop at codon 100 | s | 0 |
| F64 (F) | 11 | Microcephaly, short stature | CR | CRem | del 2 bp (390-391) | Frameshift at codon 130 stop at codon 151 | s | 0 |
| G22 (F) | 3 | None | NA | NA | del 2 bp (293-294) | Frameshift at codon 98 stop at codon 152 | s | 0 |
| I1 (M) | 2 | Short stature | NR | CRem | deletion of a complete allele | s | 0 | |
| I2 (M) | 1 | None | NR | TD | del A 341 | Frameshift at codon 115 stop at codon 123 | s | 0 |
| I4 (M) | 1 | None | CR | CRem | Ins G between nt 238 and 239 | Frameshift at codon 80 stop at codon 153 | s | 0 |
| I5 (F) | 0 | Bilateral congenital glaucoma, short stature, left eyelid ptosis, atrial septal defect | NR | TD | Ins AGA between nt 53 and 54 | Insertion E at codon 19 no frameshift | s | 0 |
| E10 (M) | 3 | None | CR | SD | del 2 bp (383-384) | Frameshift at codon 128 stop at codon 153 | se | e: 2/2 |
| E13 (M) | 3 | High arch palate, facial asymetry | CR | SD | del 2 bp (250-251) | Frameshift at codon 84 stop at codon 153 | s | 0 |
| S10 (F) | 1 | Left kidney aplasia/hypoplasia, short stature | PR | TD | t(X;19) | s | ||
| S11 (M) | 2 | Macrocephaly, mental retardation, short broad bones, extra ribs, malformations of the spine, short stature | PR | TD | deletion of a complete allele | s | 0 | |
| I24 (F) | 2 | Mental retardation, short stature | CR | TD | 46, XX, t(8;19)(q35;q13) | s | 0 | |
| Splice sites defects | ||||||||
| F21 (M) | 2 | Short stature | NR | TD | 173 (−2) agCTT → tgCTT | Acceptor splice site IVSA | s | 0 |
| F66 (F) | 2 | None | CR | SD | del 4 bp 71 (+3-+6) Aagtgagtttgg → Agtttggg | Donor splice site defect IISD | de | d: 2/2 e: 1/ 1 te: 1/1 |
| F77 (M) | 2 | None | NR | BMT | 356 (+2) T → A ATGGgt → ATGGga | Donor splice site defect IVSD | s | 0 |
| I3 (M) | 0 | None | NR | TD | del G 173(−1) agCTT → aCTT | Acceptor splice site defect IVSA | s | 0 |
| E3 (M) | 13 | None | NR | CRem | 1 (−1) agATG → atATG | Acceptor splice site defect ISA | de | d: 1/1 e: 0/1 |
| Exon 1 (non-coding) | ||||||||
| F21 (M) | 2 | Short stature | NR | TD | del 4 bp (−629-−625) | s | 0 | |
| F25 (F) | 1 | Short stature | CR | SD | del 4 bp (−629-−625) | s | 0 | |
| F1 (M) | 0 | None | NR | TD | del 4 bp (−629-−625) | se | e: 2/3 h: 1/15 | |
| Mutations in intronic regions | ||||||||
| F11 (M) | 3 | None | NR | TD | intron 1 −460 G → T | se | e: 0/2 h: 1/2 | |
| F5 (M) | 2 | None | NR | TD | intron 5 6 pp downstream of exon 5 G → C | d | d: 0/3 | |
| F53 (F) | 0 | None | NR | TD | intron 1 −460 G → T | s | 0 | |
| F74 (F) | 2 | None | CR | SD | intron 4 18 bpdownstream of exon 4 G → C | s | 0 | |
| F15 (M) | NA | None | NR | TD | intron 1 −460 G → T | s | 0 | |
| G28 (M) | NA | None | CR | SD | intron 1 −460 G → T | s | 0 | |
| F58 (M) | 4 | None | CR | SD | −836 G → C | d | NA | |
| Patient (sex) . | Age at Diagnosis of DBA (mo) . | Malformative Status . | Response at First Steroid Course . | Status at Last Follow- Up . | Genomic DNA Mutation . | Expected Protein Alteration . | Inheritance . | Mutation in Other Family Members* . |
|---|---|---|---|---|---|---|---|---|
| Nonsense mutations | ||||||||
| F23 (F) | 1 | Kidney hypoplasia/aplasia, low hair line, short stature | NR | Dead | 34 C → T | Gln12Stop | s | 0 |
| F49 (F) | 2 | Triphalangeal thumb, low implantation of thumbs | NR | TD | 144 C → A | Tyr48Stop | s | 0 |
| F44 (M) | 2 | Hydrocephaly, unique median cerebral ventricule, atrioventricular septal defect, modeling defect of long bones | NR | TD | 99 G → C | Trp33Stop | s | 0 |
| F16 (M) | 2 | None | CR | SD | 34 C → T | Gln12Stop | s | 0 |
| F69 (F) | 22 | Short stature | CR | SD | 166 C → G | Arg56Stop | s | 0 |
| G7 (F) | 2 | Short stature | CR | SD | 280 C → T | Arg94Stop | se | e: 2/3 |
| I6 (M) | 3 | None | CR | SD | 155 G → A | Trp52Stop | s | 0 |
| I7 (M) | 0 | None | NR | TD | 31 C → T | Gln11Stop | s | 0 |
| I8 (F) | 2 | None | NR | TD | 280 C → T | Arg94Stop | d | d: 1/1 |
| I9 (F) | 0 | Congenital bilateral glaucoma | CR | TD | 280 C → T | Arg94Stop | d | d: 2/2 |
| G27 (M) | NA | NA | NA | NA | 280 C → T | Arg94Stop | s | 0 |
| Missense mutations | ||||||||
| F42 (F) | 1 | Short stature | NR | TD | 380 G → A | Gly127Gln | s | 0 |
| F18 (F) | 2 | Triphalangeal thumb, anogenital fistula, interventricular septal defect | NR | TD | 184 C → T | Arg62Trp | s | 0 |
| F32 (F) | 1 | None | PR | TD | 167 G → A | Arg56Gln | s | 0 |
| F28 (M) | 1 | None | PR | TD | 167 G → A | Arg56Gln | s | 0 |
| F7 (M) | 3 | None | NR | TD | 167 G → A | Arg56Gln | se | e: 0/3 |
| G20 (F) | 2 | None | CR | SD | 43 G → T | Val15Phe | s | 0 |
| F70 (F) | 2 | None | NR | TD | 358 G → C | Gly120Ser | s | 0 |
| F12 (M) | 0 | None | CR | CRem | 184 C → T | Arg62Trp | d | d: 2/2 |
| F9 (M) | 0 | None | CR | NA | 1 A → G | Met1Val | d | d: 2/2 |
| G24 (M) | 2 | None | NR | BMT | 184 C → T | Arg62Trp | se | e: 1/1 |
| I10 (M) | 2 | Short stature | NR | Dead | 184 C → T | Arg62Trp | s | 0 |
| I11 (F) | 4 | Duplication of thumb | CR | CRem | 302 G → A | Arg101His | s | h: 1/1 |
| I53 (M) | 0 | Micrognathia, proximal implanted thumbs, short stature | NR | TD | 184 C → T | Arg62Trp | s | 0 |
| S1 | NA | NA | NA | NA | 154 T → C | Trp52Arg | s | 0 |
| E28 (F) | 2 | None | NA | CRem | 182 C → A | Ala61Ser | se | e: 0/1 h: 1/1 |
| S2 (F) | <12 | None | NR | TD | 184 C → T | Arg62Trp | d | d: 1/1 |
| Insertions and deletions | ||||||||
| F31 (M) | 2 | None | NR | TD | ins A between nt 14 and 15 | Frameshift at codon 5 stop at codon 50 | s | 0 |
| F4 (F) | 1 | Hypertelorism, epicanthus, low ear set, antimongoloid eye line | NR | CRem | del 295 bp 356 (+281-+576) | Deletion of exon 5 | de | d: 1/1 e: 1/1 |
| F19 (M) | 2 | Epicanthus, low hairline, short stature | CR | SD | del 1 bp nt 222 | Frameshift at codon 74 stop at codon 75 | s | 0 |
| F26 (M) | 2 | Dysplasia of mitral valve, short stature | NR | TD | ins A between nt 104 and 105 | Frameshift at codon 35 stop at codon 50 | s | 0 |
| F25 (F) | 1 | Short stature | CR | SD | del 31bp (274-314) | Deletion of 31 nt of exon 4, frameshift at codon 92 stop at codon 100 | s | 0 |
| F64 (F) | 11 | Microcephaly, short stature | CR | CRem | del 2 bp (390-391) | Frameshift at codon 130 stop at codon 151 | s | 0 |
| G22 (F) | 3 | None | NA | NA | del 2 bp (293-294) | Frameshift at codon 98 stop at codon 152 | s | 0 |
| I1 (M) | 2 | Short stature | NR | CRem | deletion of a complete allele | s | 0 | |
| I2 (M) | 1 | None | NR | TD | del A 341 | Frameshift at codon 115 stop at codon 123 | s | 0 |
| I4 (M) | 1 | None | CR | CRem | Ins G between nt 238 and 239 | Frameshift at codon 80 stop at codon 153 | s | 0 |
| I5 (F) | 0 | Bilateral congenital glaucoma, short stature, left eyelid ptosis, atrial septal defect | NR | TD | Ins AGA between nt 53 and 54 | Insertion E at codon 19 no frameshift | s | 0 |
| E10 (M) | 3 | None | CR | SD | del 2 bp (383-384) | Frameshift at codon 128 stop at codon 153 | se | e: 2/2 |
| E13 (M) | 3 | High arch palate, facial asymetry | CR | SD | del 2 bp (250-251) | Frameshift at codon 84 stop at codon 153 | s | 0 |
| S10 (F) | 1 | Left kidney aplasia/hypoplasia, short stature | PR | TD | t(X;19) | s | ||
| S11 (M) | 2 | Macrocephaly, mental retardation, short broad bones, extra ribs, malformations of the spine, short stature | PR | TD | deletion of a complete allele | s | 0 | |
| I24 (F) | 2 | Mental retardation, short stature | CR | TD | 46, XX, t(8;19)(q35;q13) | s | 0 | |
| Splice sites defects | ||||||||
| F21 (M) | 2 | Short stature | NR | TD | 173 (−2) agCTT → tgCTT | Acceptor splice site IVSA | s | 0 |
| F66 (F) | 2 | None | CR | SD | del 4 bp 71 (+3-+6) Aagtgagtttgg → Agtttggg | Donor splice site defect IISD | de | d: 2/2 e: 1/ 1 te: 1/1 |
| F77 (M) | 2 | None | NR | BMT | 356 (+2) T → A ATGGgt → ATGGga | Donor splice site defect IVSD | s | 0 |
| I3 (M) | 0 | None | NR | TD | del G 173(−1) agCTT → aCTT | Acceptor splice site defect IVSA | s | 0 |
| E3 (M) | 13 | None | NR | CRem | 1 (−1) agATG → atATG | Acceptor splice site defect ISA | de | d: 1/1 e: 0/1 |
| Exon 1 (non-coding) | ||||||||
| F21 (M) | 2 | Short stature | NR | TD | del 4 bp (−629-−625) | s | 0 | |
| F25 (F) | 1 | Short stature | CR | SD | del 4 bp (−629-−625) | s | 0 | |
| F1 (M) | 0 | None | NR | TD | del 4 bp (−629-−625) | se | e: 2/3 h: 1/15 | |
| Mutations in intronic regions | ||||||||
| F11 (M) | 3 | None | NR | TD | intron 1 −460 G → T | se | e: 0/2 h: 1/2 | |
| F5 (M) | 2 | None | NR | TD | intron 5 6 pp downstream of exon 5 G → C | d | d: 0/3 | |
| F53 (F) | 0 | None | NR | TD | intron 1 −460 G → T | s | 0 | |
| F74 (F) | 2 | None | CR | SD | intron 4 18 bpdownstream of exon 4 G → C | s | 0 | |
| F15 (M) | NA | None | NR | TD | intron 1 −460 G → T | s | 0 | |
| G28 (M) | NA | None | CR | SD | intron 1 −460 G → T | s | 0 | |
| F58 (M) | 4 | None | CR | SD | −836 G → C | d | NA | |
Nucleotide number is based on the cDNA sequence data of RPS19 gene (Kondoh et al37), starting from the ATG. Mutations occurring upstream of ATG are numbered according to the genomic sequence, starting from the ATG (Draptchinskaia et al25). Amino acids are numbered from the first Methionine.
Abbreviations: TD, transfusion dependent; SD, steroid dependent; CRem, complete remission (free of treatment); BMT, treated with bone marrow transplantation; NA, not available; NR, no response; PR, partial response; CR, complete response; h, healthy; s, sporadic; d, dominant; e, occurrence of isolated high eADA phenotype in the family; te, transient erythroblastopenia; nt, nucleotide number; del, deletion; ins, insertion.
For each group of individuals is indicated the number sharing the same mutation than the DBA patient.
In 33 patients, a family history of at least 2 classical DBA in different generations was consistent with an autosomal dominant pattern of inheritance and they were classified as dominant, although some of these families also included individuals with more equivocal hematological abnormalities. One hundred fifty-four index cases were classified as sporadic, with neither history of unexplained anemia in first-degree relatives nor abnormal hematological results such as increased MCV or eADA activity. Twenty-one DBA patients, despite the absence of family history of DBA, had a first-degree relative with a history of either unexplained anemia and/or elevated MCV and/or eADA activity greater than mean +2 SD. This group included a pair of monozygotic twins whose otherwise healthy father had an elevated MCV, but with normal eADA activity. In the remaining cases (n = 8), family data were unavailable or incomplete.
RPS19 Mutations in First-Degree Relatives
Sporadic cases.
No mutation was found in any of the healthy and hematologically normal family members of the 38 sporadic cases in whom a mutation had been detected (of a total of 154 sporadic index cases studied).
Dominant cases.
Cosegregation between the observed RPS19 mutation and the DBA phenotype was consistently found in the 8 dominant families in which mutations affecting coding regions or splice sites had been identified in the index case. A patient from 1 additional dominant family (F5) showed a mutation in the noncoding region of RPS19 gene, and this mutation could be documented in only 1 of 3 affected individuals in this family. A mutation occurring in intron 1 in another dominant case (F58) still remains to be explored in other family members, when DNA becomes available. Of the 23 dominant families in which no mutations had been detected, 3 had previously been shown not to be linked with 19q13 and 3 had shown equivocal linkage, whereas cosegregation analysis in the remaining families would have been consistent with 19q13 linkage (data not shown). However, it should be noted that many of the latter group of families often comprises no more than 4 individuals. It remains unclear whether the DBA phenotype thus segregates with markers on 19q13.2 by chance or more complex rearrangements of the chromosome 19q13.2 region are occurring in these DBA patients.
Familial high eADA individuals.
RPS19 mutations were found in 11 of 25 index cases with 1 or more first-degree relatives showing isolated high eADA activity (familial high eADA) or other hematological abnormalities. In 5 families, the mutation detected in the proband was found in all family members with high eADA or macrocytosis. In 4 of these 5 families (F4, E10, F66, and G24), either a mutation in the coding sequence or a mutation affecting a splice site cosegregated with either classical DBA or other hematological abnormalities (Fig 5A and B). All of these individuals with an RPS19 mutation had an increase of eADA activity greater than +3 SD of the normal values. In the fifth family (I11), the same missense mutation was detected in a pair of monozygotic twins with classical DBA and their father, who had persistent macrocytosis but a normal eADA. Conversely, in 6 other families in which a mutation in RPS19 gene was found, there was only partial cosegregation of the mutation with isolated hematological or biochemical abnormalities. A missense mutation in 1 transfusion-dependent DBA patient (F7) was not found in any of the 3 brothers who presented with a mild increase in eADA. In family F1, the 4-bp deletion in exon 1 was present in the DBA affected proband as well in 2 individuals presenting with the isolated high eADA phenotype (Fig6A). However, this deletion was also found in another family member despite both normal red blood cell indices and normal eADA activity. A splice site defect found in the 2 DBA cases of the dominant family E3 was not found in the brother of the proband, whose eADA activity was between +2 and +3 SD above the normal mean (Fig 6B).
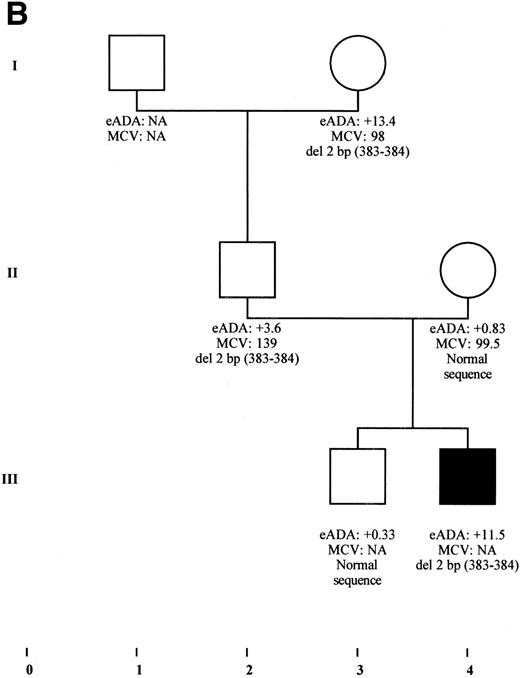
Cosegregation of mutation in RPS19 gene in 2 families sharing both DBA phenotype and isolated high eADA phenotype. (A) Cosegregation of a donor splice site defect of exon 2 in individuals from family F66 with various clinical presentation: DBA phenotype (individuals III-1, III-2, III-3), silent phenotype restricted to an isolated elevation of eADA activity (individual II-2), and transient erythroblastopenia in individual III-3, with a persistent high eADA activity. The mutation was not found in any of the other family members explored. eADA values are expressed as SD from the mean. Fetal hemoglobin is expressed as a percentage. NA, not available. (B) Cosegregation of a 2-bp deletion in exon 5 in family E10. DBA proband III-4 is transfusion-independent under steroid therapy, whereas individuals II-2 and I-3 never received any treatment, although they were anemic during early childhood. eADA values are expressed as SD from the mean. NA, not available.
Cosegregation of mutation in RPS19 gene in 2 families sharing both DBA phenotype and isolated high eADA phenotype. (A) Cosegregation of a donor splice site defect of exon 2 in individuals from family F66 with various clinical presentation: DBA phenotype (individuals III-1, III-2, III-3), silent phenotype restricted to an isolated elevation of eADA activity (individual II-2), and transient erythroblastopenia in individual III-3, with a persistent high eADA activity. The mutation was not found in any of the other family members explored. eADA values are expressed as SD from the mean. Fetal hemoglobin is expressed as a percentage. NA, not available. (B) Cosegregation of a 2-bp deletion in exon 5 in family E10. DBA proband III-4 is transfusion-independent under steroid therapy, whereas individuals II-2 and I-3 never received any treatment, although they were anemic during early childhood. eADA values are expressed as SD from the mean. NA, not available.
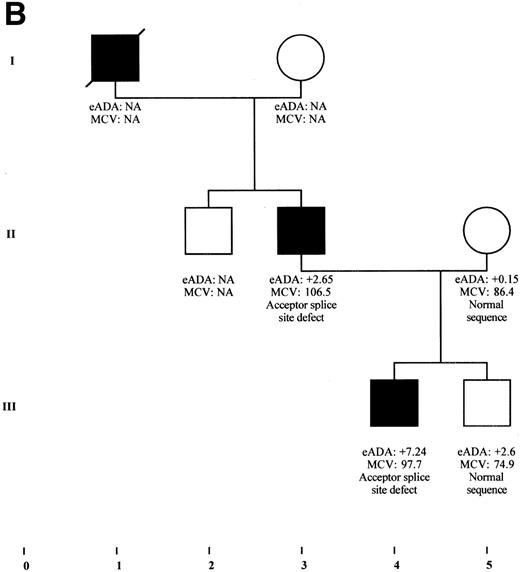
(A) Transmission of a deletion in exon 1 of RPS19 gene in a family with DBA phenotype and isolated high eADA phenotype. The 4-bp deletion found in the first, noncoding exon of RPS19 gene in the DBA proband IV-3 from family F1 was also found in the mother (III-3) and the grandmother (II-5) who presented only with a high eADA phenotype. Other factors are probably involved in this family, because individual III-4 carries the same deletion, but with a strictly normal phenotype, whereas the deletion could not be found in individual II-11, despite an isolated high eADA phenotype. eADA values are expressed as SD from the mean. NA, not available. (B) Partial segregation of a mutation in a DBA family. The acceptor splice site defect of exon 4 was found in the DBA proband III-4, as well as in his father (II-3) presenting with macrocytic anemia. Erythrocyte ADA value measured in the healthy brother III-5 is between +2 and +3 SD, although he does not carry this mutation. eADA values are expressed as SD from the mean. NA, not available.
(A) Transmission of a deletion in exon 1 of RPS19 gene in a family with DBA phenotype and isolated high eADA phenotype. The 4-bp deletion found in the first, noncoding exon of RPS19 gene in the DBA proband IV-3 from family F1 was also found in the mother (III-3) and the grandmother (II-5) who presented only with a high eADA phenotype. Other factors are probably involved in this family, because individual III-4 carries the same deletion, but with a strictly normal phenotype, whereas the deletion could not be found in individual II-11, despite an isolated high eADA phenotype. eADA values are expressed as SD from the mean. NA, not available. (B) Partial segregation of a mutation in a DBA family. The acceptor splice site defect of exon 4 was found in the DBA proband III-4, as well as in his father (II-3) presenting with macrocytic anemia. Erythrocyte ADA value measured in the healthy brother III-5 is between +2 and +3 SD, although he does not carry this mutation. eADA values are expressed as SD from the mean. NA, not available.
Phenotype-Genotype Correlations
No constant clinical features or therapeutic outcome could be found within patients from different families displaying identical mutations in RPS19 gene. Similarly, the clinical expression or therapeutic response was different in various family members in dominant families, although displaying the same mutations in RPS19 gene. This was also illustrated by the discrepancy between the pair of monozygotic twins from family I11 with respect to physical anomalies (Fig 7). Despite their apparent genotypic identity, only 1 showed duplication of the thumb. The twins were identical for the following markers: D19S197, D19S408 at the DBA locus on 19q13.2; STR at the PAH locus on chromosome 12q; (GGAA)n, repeat microsatellite located −548 bp upstream from the ATG start codon of the R-EPO locus on 19s13.2; and D5S658 and RPS on 5q. The twins were thus considered to be monozygotic. When grouping individuals with mutations in the coding sequence or affecting splice site of RPS19 gene according to the location of the mutation, we were unable to correlate the clinical phenotype with the length of conservation of the native protein or with the conservation of any of the 3 motifs described above. Specifically, mutations involving motif 1 were not associated with a specific phenotype or outcome.
Cosegregation of a missense mutation of RPS19 gene in a family with various clinical phenotypes in monozygotic twins. A single nucleotide substitution (302 G→A) resulting in an amino acid change of a consensus residue (Arg101His) was found in monozygotic twins (II-2 and II-3) from family I11, with only 1 of them presenting with a duplication of the thumb. The same mutation was also found in the healthy father (I-1), who only exhibited a macrocytosis. eADA values are expressed as SD from the mean. NA, not available.
Cosegregation of a missense mutation of RPS19 gene in a family with various clinical phenotypes in monozygotic twins. A single nucleotide substitution (302 G→A) resulting in an amino acid change of a consensus residue (Arg101His) was found in monozygotic twins (II-2 and II-3) from family I11, with only 1 of them presenting with a duplication of the thumb. The same mutation was also found in the healthy father (I-1), who only exhibited a macrocytosis. eADA values are expressed as SD from the mean. NA, not available.
We then looked at the clinical phenotype of patients with sporadic DBA to determine whether individuals with RPS19 mutations could be distinguished from those without. Probands with dominant or familial high eADA families were not included in this part of the analysis because of the intrafamilial variation. Among the 154 sporadic cases, mutations either in the coding region or in other regions of the gene were not associated with a specific age at presentation, clinical phenotype, or outcome of the disease. Surprisingly, RPS19 gene mutations were found to be less prevalent among initially steroid responders (19.7%) as compared with initially steroid-resistant patients (40%; P = .006). However, mutation frequency was found to be comparable in transfusion-dependent individuals (33.3%), in long term steroid-dependent patients (23%), or in patients independent of any treatment on long-term follow-up (P = .20). These findings remained unchanged when only mutations affecting coding regions of RPS19 gene were considered for analysis (data not shown).
DISCUSSION
A major advance in the genetics of DBA was recently achieved with the description of a series of mutations affecting the RPS19 gene in 10 unrelated DBA patients.25 In the present study involving a large international collaborative effort, we report the detailed characterization of mutations in RPS19 gene in 194 patients from 172 new families with DBA and attempt to establish the potential relationship between molecular defects and phenotype. Our results significantly extend the initial findings based on a small group of patients and show that alterations in RPS19 gene can be documented in up to 26% of DBA patients.
We have demonstrated that a wide range of clinical presentations may be associated with mutations affecting the RPS19 gene, even for an identical mutation within the same family. Thus, a mutation affecting the RPS19 gene in a single family may be either clinically silent or associated with an overt DBA phenotype, including transfusion dependence and/or multiple physical anomalies. The clinical expression of a given mutation must therefore be modified by other factors. These findings have immediate implications for genetic counseling, including antenatal diagnosis and presymptomatic diagnosis. A mutation in RPS19 gene in a fetus or a sibling of a DBA patient will not help to predict between an overt DBA phenotype and a silent expression without any physical or hematological abnormality. On the other hand, the assessment of a family of a DBA patient considered as eligible for a BMT should include the full clinical and biological characterization of the family, including eADA assessment, and rule out donors sharing with the DBA proband a mutation in RPS19 gene.
These results also show that mutations affecting the RPS19 gene could not be detected in 74% of patients with DBA. In addition, in some families with an RPS19 mutation, the mutation was not detected in every family member with increased eADA activity. It is unlikely that mutations were missed in all of these cases, although direct sequencing of PCR products will not detect complete allelic loss. In a previous study, only 2 of 50 patients with sporadic DBA were found to have deletions affecting the 19q13.2 locus.23 Moreover, there was no cosegregation of DBA with 19q13.2 markers in a proportion of dominant families.23 24 It is noteworthy that the clinical phenotype of patients with and without RPS19 mutations is effectively indistinguishable, including the occurrence of mild hematological abnormalities or isolated raised eADA activity in family members. In 5 families, we found that the silent phenotype described as elevated eADA activity (4 families) or macrocytosis (1 case) cosegregated with RPS19 gene mutations identified in the proband. Conversely, in 6 other families, the RPS19 gene mutations identified in the proband was not found in all family members exhibiting the silent phenotype. The likely explanations for this discrepancy include the threshold used to define the high eADA phenotype, or other factors modifying the expression of ADA or the expression of RPS19 gene.
Although the identified mutations were scattered throughout the RPS19 gene, we did find 1 hot spot of missense mutations between codons 52 and 62. The region W52-[YFW]-[YF]-[VTILK]-R-[AT]-A-[SA]-[IVLT]-[AL]-R-[HRK]-[ILV]-Y65is highly conserved among all species, ranging from Archeobacteria,C elegans, Drosophila Melanogaster, to mamalian, for which RPS19 sequence is currently available. This observation does suggest that this region of RPS19 is likely to have a critical role in its function, which is yet to be defined.
Our finding that mutations affect only a single allele is consistent with the observed pattern of dominant transmission in most of familial DBA. This further implies that haploinsufficiency of the RPS19 may underlie the pathogenesis of DBA. Interestingly, we were unable to identify any mutations in RPS19 gene in 7 DBA patients born to consanguine patients. This finding suggests that another yet to be identified gene may be responsible for the potentially recessive forms of DBA.
In our study, we did not find any direct correlation between phenotype (age at presentation, clinical phenotype, and long-term outcome) and genotype (presence or absence of RPS19 gene mutation or the nature of the mutation identified). Interestingly, we did notice that RPS19 gene mutations were more prevalent in initially steroid resistant patients (40% v 19.7% in initially steroid responders). However, this finding cannot be used as a prognostic factor, because the difference was not sustained on long-term follow-up.
DBA thus represents the first known human disease in which a defect in a ribosomal protein is involved in the pathophysiology of the disease. Two potential mechanisms might apply: (1) function of ribosomal proteins in protein synthesis and (2) extraribosomal function for the RPS19 protein. On the one hand, haploinsufficiency, with variable reduction in the rate of protein synthesis, could contribute to modulate the expression of the disease. Such a mechanism is strongly suggested by the Drosophila models, for which the various minute phenotypes have been assigned to mutations in several ribosomal proteins and attributed to haploinsufficiency during embryonic development. Modeling of the influence of probabilistic factors in the expression of genes recently suggested that increased delays in gene initiation or interruption of gene expression might account for part of the variable phenotypic expression of haploinsufficiency syndromes.34 However, at our current state of knowledge, it is difficult to explain such a specific effect on both embryogenesis and postnatal erythropoiesis. Alternatively, some ribosomal proteins have been shown to have extraribosomal functions, such as rat and Drosophila melanogaster ribosomal protein S3 that appears to be also a DNA-repair endonuclease.35 No such activity has been reported so far for RPS19, although human RPS19 dimers have been shown to be chemotactic factors for monocytes.36 However, it is unlikely that monocytes play a direct role in the pathogenesis of DBA. It is now important to define the role of the RPS19 product in normal and abnormal erythropoiesis and to identify interacting factors, which may both elucidate the pathophysiology of the erythroid failure in DBA and suggest alternative candidate genes, which may be mutated in DBA patients without RPS19 mutations.
ACKNOWLEDGMENT
The authors acknowledge the DBA working groups of the European Society for Paediatric Haematology and Immunology (ESPHI) and of the Société d'Hématologie et d'Immunologie Pédiatrique (SHIP) and the Associazione Italiana di Ematologia e Oncologia Pediatrica (AIEOP). We are grateful to Dr Philippe Gascard (LBNL) and Dr Sylvain Choquet for their helpful suggestions. We gratefully acknowledge Dr Jamie Cope (University of California, Berkeley, CA) for his help in alignment of protein sequences and Michael Patterson for his help in the sequencing work.
Sequences used were retrieved from NCBI Entrez Protein query: from mammalian species: human HUMS19RP (337733), rat RS19RAT (133857), pig RS_19PIG (2500494); from Mya arenariaRS19_MYAAR (2500495); from Drosophila melanogaster RS19_DROME (730639); from fungi: Schizosaccharomyces pombe SPBC649 (3136024), Emericella nidulans RS19_EMENI (133844),Saccharomyces cerevisiae S19A (133864), Saccharomyces cerevisiae S19eB (2119095), Saccharomyces pastorianus S16A (4369); from Archaea bacteria: Pyrococcus horikoshii (3257748),Methanobacterium thermoautotrophicum R19E_METTH (3122650),Methanococcus jannaschii R19E_METJA (1591407), Archeoglobus fulgidus rps19E (2648466); from Entamoeba histolyticaRS19_ENTHI (3122799); from nematodes: Ascaris suum RS19_ASCSU (730457), Ascaris suum eliminated protein n°1 R19G_ASCSU (133843), Ascaris lumbricoides (84483), Caenorhabditis elegans (3924845); from plants: rice RS19_ORYSA (730456).
Participating clinical investigators from Italy: A. Lippi (Firenze), G. Forni (Genova), F. Locatelli (Pavia), P.G. Mori (Genova), M. Mair (Brunico), S. Varotto (Padova), M. D'Avanzo (Napoli), B. Nobili (Napoli), G. Andria (Napoli), F. Massolo (Modena), G. Russo (Catania), G.P. Bagnara (Bologna), G. Izzi (Parma), G. Castaman (Vicenza), M.R. Govoni (Ferrara), R. Galanello (Cagliari).
Participating clinical investigators from France: A. Babin Boilletot (Strasbourg), A. Barruchel (Paris), D. Berets (Clamart), C. Berger (Saint Etienne), F. Bernaudin (Créteil), Y. Bertrand (Lyon), S. Blanche (Paris), J.F. Boccara (Paris), P. Bordigoni (Nancy), C. Chenel (Papeete), B. Chevallier (Boulogne Billancourt), B. Coiffier (Lyon), G. Daltroff (Belfort), M. Debré (Paris), A. Deville (Nice), H. Dombret (Paris), J.P. Dommergues (Le Kremlin-Bicêtre), J. Donadieu (Paris), V. Dorvaux (Metz), F. Dreyfus (Paris), R. Girot (Paris), R. Goddon (Montluçon), J.M. Guillard (Bordeaux), M. Guillot (Lisieux), P. Labrune (Clamart), C. Lajarrige (Laon), J.P. Lamagnere (Tours), A. Lambiliotte (Lille), T. Leblanc (Paris), O. Lejars (Tours), Cl. Lejeune (Colombes), G. Leverger (Paris), L. de Lumley (Limoges), G. Margueritte (Montpellier), F. Méchinaud (Nantes), J.L. Mesnil (Flers), M.J. Milleret Proyart (Sens), M. Monconduit (Rouen), M. Munzer (Reims), C. Narcy (Saint Germain en Laye), R. Ouelbany (Margency), B. Pautard (Amiens), H. Perrimond (Marseille), N. Philippe (Lyon), M.P. Pignol (Mont de Marsan), H. Piguet (Rouen), A. Plou (Nantes), A. Robert (Toulouse), Cl. Roy (Paris), D. Quillerou (Troyes), G. Souillet (Lyon), H. Testard (Saint Denis de la Réunion), C. Saint-Aimé (Fort de France), G. Schaison (Paris), D. Stamm (Lyon), J.L. Stéphan (Saint Priez en Jarrez), Ph. Tron (Rouen), and C. Vervel (Compiègne).
Participating clinical investigators from Germany: K. Bode (Bonn), J. Budde (Freiburg), H. Cario (Ulm), W.M. Debatin (Heidelberg), W. Dörffel (Berlin), W. Eberl (Braunschweig), E. Harms (Münster), C. Hasan (Bonn), L. Kanz (Tubingen), E. Kohne (Ulm), S. Müller-Weihrich (München), J. Ritter (Münster), T. Wiesel (Datteln), N. Yudina (Voronezh).
Participating clinical investigators from United Kingdom: B. Gibson, E. Simpson (Glasgow), P. Darbyshire, D. Milligan (Birmingham), R. Stevens, A. Will (Manchester), S. Davies (Yeovil), A. Parker (Edinburgh), A. Goringe (Cardiff), M. Barraclough (Hull), S. Kinsey, C. Shiach (Leeds), P. Skacel (London), J. Reiser (Stevenage), J. Martin (Liverpool), J. Wimperis (Norwich), K. Dodd (Derby), D. Walker (Nottingham),MReid (Newcastle), L. Lamont (Chichester), S. Haider (Bury), D. King (Aberdeen), R. Wilkie (Dundee).
Participating clinical investigators from Sweden: G. Elinder (Stockholm), J.-I. Henter (Stockholm), M. Donner (Lund).
Contributors from other countries: G. Cornu (Bruxelles, Belgium), J. Otten (Bruxelles, Belgium), J. Humbert (Genève, Switzerland), T. Révész (Utrechts, The Netherlands), P. Philippet (Montegnee, Belgium), Nancy Olivieri (Toronto, Ontario, Canada), Elizabeth Smibert (Melbourne, Australia), Silvia Brandalise (Campinas, Brasil).
Participating laboratories: J.P. Cartron (Paris), L. Croisille (Le Kremlin-Bicêtre), L. Coulombel (Villejuif), P. Gane (Paris), J.L. Pérignon (Paris), P.H. Roméo (Créteil).
N. Draptchinskaia, I. Dianzani, and S. Ball contributed equally to this work
Supported by grants from the Children's Cancer Foundation of Sweden, the DBA Foundation Inc, the Swedish Medical Research Council, T. and R. Söderbergs Fund, The Beijer Foundation, the Borgström Foundation, R. McDonalds fund, Lundbergs Foundation and Uppsala University, Association Française contre les Myopathies (AFM), Généthon, and Direction de la Recherche Clinique (AP-HP Paris; CRC 950183), Telethon Italia (Grant No. E619), the Max Reinhardt Charitable Trust, LDRD funds from Lawrence Berkeley National Laboratory, and National Institutes of Health Grants No. DK32094 and DK26263.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.
REFERENCES
Author notes
Address reprint requests to Thiébaut-Noël Willig, MD, LBNL-LSD, Mail stop 74-217, 1 Cyclotron Rd, Berkeley, CA 94720; e-mail:tnwillig@lbl.gov.








This feature is available to Subscribers Only
Sign In or Create an Account Close Modal