In this issue of Blood, Han et al report a new therapeutic vulnerability for T-cell acute lymphoblastic leukemia (T-ALL) by targeting the essential histone modifier chromatin-associated WD40 repeat domain protein 5 (WDR5).1 The authors demonstrate synergistic efficacy of combining a direct WDR5 inhibitor with inhibition of the cell cycle regulator caseine kinase 2 (CK2), which indirectly reduces WDR5 expression.
The prognosis of patients with T-ALL at relapse is devastating in all age groups.2 Therefore, novel therapies are urgently needed. Although immunotherapeutic strategies have made some progress in the past few years, nonimmunological targeted therapy options in T-ALL are scarce. Although the mutational landscape of T-ALL has been comprehensively characterized,3 the majority of T-ALL subtypes lack well-defined genomic vulnerabilities or mutated genes that could serve as drug targets. This has led to alternative treatment directions, such as targeting the epigenome, which has demonstrated compelling therapeutic benefit in various hematological malignancies. For example, hypomethylating agents have been approved by the Food and Drug Administration for acute myeloid leukemia (AML) and myelodysplastic syndrome.4 Similarly, lysine methyltransferase 2A (KMT2A) rearranged hematological malignancies, including B-cell precursor ALL and AML, have been treated with epigenetic interventions, such as small-molecule inhibitors targeting the disruptor of telomeric silencing 1-like (DOT1L) and the menin inhibitor revumenib, recently approved for relapsed or refractory acute leukemia with KMT2A translocation.5 However, for other acute leukemia subtypes, such as high-risk relapsed or refractory T-ALL, specific epigenetic therapies are not available, underscoring the need for continued exploration in this area.
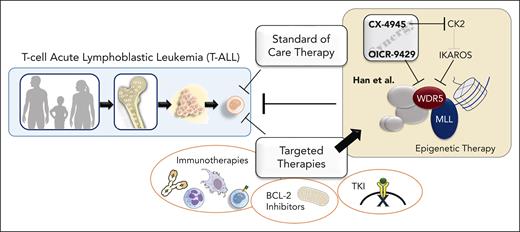
In this article, Han et al identified a promising novel epigenomic targeting strategy in T-ALL. They present an approach based on direct inhibition of WDR5 and indirect inhibition of WDR5 via CK2 and its downstream target IKAROS. WDR5 is an essential subunit of the mixed lineage leukemia (MLL) histone methyltransferase complexes and is associated with increased histone H3 lysine 4 (K4) methylation,6 which results ultimately in increased expression of oncogenes like MYC or genes from the HOX cluster in acute leukemias.7
This comprehensive study includes correlative analyses in a T-ALL patient cohort, in vitro and in vivo loss-of-function studies in cell lines, and drug treatments in patient-derived xenograft (PDX) mouse models. The authors identified the correlation of high WDR5 expression with increased cell cycle activity and worse prognosis in patients with T-ALL. To test this correlation, they showed that inhibition of WDR5 induced cell cycle arrest in vitro and delayed leukemia onset in mouse models in vivo. Interestingly, WDR5 can be targeted by medications, for instance, by the small molecule inhibitor OICR-9429 that competitively blocks the WDR5 interaction with MLL1. The authors also observed that the CK2 inhibitor CX-4945 was able to block WDR5 function by reducing its expression, probably through activation of the WDR5 antagonizing transcription factor IKAROS, thus providing an alternative mechanism to impede WDR5 activity. As a downstream mediator of WDR5, the authors identified adenosine triphosphatase family AAA domain containing 2 (ATAD2). An ATAD2 knockdown showed a comparable phenotype to that of WDR5 inhibition. Finally, in vivo experiments demonstrated that dual targeting of WDR5 by a combination of WDR5 and CK2 inhibition was synergistic in animal models of T-ALL (see figure).
Schematic overview of the potential therapeutic benefits of a novel epigenetic treatment strategy for T-ALL, as described by Han et al. Next to the current standard of care (chemotherapy), several targeted therapies have emerged. These include immunotherapies, the BCL-2 inhibitor venetoclax, and tyrosine kinase inhibitors (TKI). The promising new approach involves direct inhibition of the histone modifier WDR5 using OICR-9429. Adding indirect WDR5 inhibition by targeting the casein kinase 2 (CK2)-IKAROS axis through the cell cycle repressor CX-4945 resulted in remarkable synergistic activity.
Schematic overview of the potential therapeutic benefits of a novel epigenetic treatment strategy for T-ALL, as described by Han et al. Next to the current standard of care (chemotherapy), several targeted therapies have emerged. These include immunotherapies, the BCL-2 inhibitor venetoclax, and tyrosine kinase inhibitors (TKI). The promising new approach involves direct inhibition of the histone modifier WDR5 using OICR-9429. Adding indirect WDR5 inhibition by targeting the casein kinase 2 (CK2)-IKAROS axis through the cell cycle repressor CX-4945 resulted in remarkable synergistic activity.
This study found impressive synergistic activity of the 2 compounds, both in high-risk T-ALL PDX models and in a model of early T-cell precursor ALL, a disease carrying a particularly poor prognosis. As OICR-9429 has not reached clinical trials yet, the observation of CX-4945 (silmitasertib) inhibiting WDR5 at the transcriptional level increases the potential of this study. Silmitasertib has shown promising results in solid tumors, including locally advanced carcinoma.8 Also, a comprehensive clinical study of silmitasertib in combination with chemotherapy has been initiated for relapsed or refractory solid tumors (NCT06541262). Strikingly, the synergistic activity was observed in PDX models of overt T-ALL with ≥5% blasts in the peripheral blood. In that setting, only highly efficient therapies are able to provide significant survival prolongations in PDX mice. Furthermore, the drug combination caused irreversible growth arrest in the G1 phase of the cell cycle, which was followed not by regrowth, but by apoptosis. Sending cells into growth arrest and successfully directing them into cell death from there is a therapeutic strategy of great potential to target chemotherapy-resistant cancer cells.9
Obviously, a number of open questions remain: one is the identification of T-ALL subgroups that would benefit the most from this novel intervention. Also, it may be interesting to investigate biomarkers in patient leukemia cells to help predict therapeutic response to these therapies. From a biological standpoint, further investigations to determine if the WDR5-knockdown phenotype is associated with cellular senescence may be attractive. This approach may then be suitable for targeting dormant cancer cells, thereby leading to long-term remissions. Finally, from a clinical perspective, it will be of great importance to assess the safety and tolerability of this regimen.
In conclusion, the work by Han and colleagues has made first steps in an important direction. It is possible that novel approaches targeting the epigenome will play a role in improving the prognosis of T-ALL at relapse, if they are tolerable in this often heavily pretreated patient population.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal