Introduction
Truncating ASXL1 mutations in chronic myelomonocytic leukemia (CMML) are associated with inferior overall survival (OS) and leukemia free survival (LFS). Conversely, loss-of-function mutations in TET2 are associated with better outcomes, with the ASXL1wt/ TET2mut genotype being associated with the best OS and LFS-rates (Coltro G, et al. Leukemia. 2020). Contemporary CMML prognostic scoring systems - such as the Mayo Molecular Model (MMM; Patnaik MM, et al. Leukemia. 2014) and the CPSS-Molecular Model (CMM; Elena C, et al. Blood. 2016) - do not consider concomitant ASXL1/ TET2 mutational status or mutational VAF. We hypothesized that the ASXL1/ TET2 genotype would better risk stratify patients in comparison to ASXL1 mutations alone, and that this effect would be proportional to the underlying mutational VAF.
Methods
After IRB approval, we compiled a large (n = 888), molecularly annotated database of CMML patients seen at two US medical centers (Mayo Clinic and MD Anderson) via retrospective chart review. All statistical analyses considered the clinical and laboratory parameters obtained at the time of presentation to the respective institution. Categorical variables were compared by Fisher exact or Pearson χ 2 tests and continuous variables by Mann-Whitney U tests. Univariate and multivariate analyses were performed using Cox proportional hazards regression models. Survival was assessed via the Kaplan-Meier method. P-values < 0.05 were considered significant. All statistical calculations were performed using the BlueSky Statistics (v10.3.1) interface for R.
Results
The median age of the cohort was 71 years (range 20 - 94), 67% were male, 46% had proliferative CMML (pCMML), and 19% had CMML-2. The distribution of low, intermediate-1, intermediate-2, and high-risk patients was 15%, 11%, 32%, and 42% by the MMM and 9%, 23%, 40%, and 28% by the CMM, respectively. The most frequently mutated genes were ASXL1 (45%), TET2 (43%), SRSF2 (41%), and RUNX1 (17%), while 40% harbored at least one RAS pathway mutation ( NRAS 16%, CBL 15%, KRAS 8%, PTPN11 4%, FLT3 2%). The median OS and LFS of the entire cohort were 31.8 months (95% CI 28.4 - 33.5 months) and 28.4 months (95% CI 24.6 - 32.1 months), respectively.
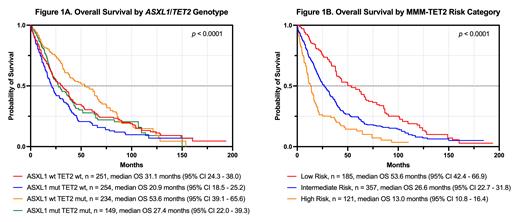
Patients were stratified into ASXL1wt/ TET2wt (28%), ASXL1mut/ TET2wt (29%), ASXL1wt/ TET2mut (26%), and ASXL1mut/ TET2mut (17%) genotypes. Those with isolated TET2 mutations had the longest median OS (53.6 months; p < 0.02 for all comparisons) whereas those with isolated ASXL1 mutations had the shortest median OS (20.9 months; p < 0.04 for all comparisons); mutations in neither or both genes performed similarly (p = 0.9224; Figure 1A). The same trend was seen for LFS. We further hypothesized that the ASXL1 or TET2 mutation VAF would be more predictive of outcomes than a binary metric. The respective median VAFs were 37% (quartiles 28% - 45%) and 45% (quartiles 40% - 49%). When treated as a continuous variable, however, there was no correlation between either VAF and OS or LFS by both Pearson linear regression and Cox proportional hazard modeling.
Unlike the overall cohort, the ASXL1/ TET2 genotypes did not further stratify patients with pCMML, CMML-2, or those considered high risk by the CMM in Kaplan-Meier analyses, suggesting that the high-risk features inherent in these subgroups outweigh the benefits of the TET2 mutation. In contrast, patients considered high-risk by the MMM were further stratified by the ASXL1/ TET2 genotypes. Incorporating TET2 mutations into the MMM as a protective factor (worth -1.5 points) improved detection of low- (≤ 1 point) and high-risk (≥ 4 points) patients, while intermediate-1 and intermediate-2 coalesced into a single intermediate risk category ( Figure 1B). A similar phenomenon was observed when TET2 was incorporated into the CMM (worth -1 point, yielding low- (0 points), intermediate- (1-2 points), and high-risk (≥ 3 points) groups with median OS of 83 months (95% CI 66.2 - 104.2), 34.3 months (95% CI 31.6 - 38.7), and 17.7 months (95% CI 15.7 - 20.2), respectively.
Conclusions
Truncating and hypomorphic TET2 mutations are associated with better outcomes in CMML, with the ASXL1mut/ TET2wt genotype being associated with improved risk stratification in comparison to ASXL1 mutational status alone. Counterintuitively, in our data set, this stratification was not impacted by mutational VAF, supporting binary mutational assessments in prognostic models.
Disclosures
Chien:AbbVie: Consultancy; Rigel Pharmaceuticals: Consultancy. Alkhateeb:Mayo Clinic: Current Employment. Garcia-Manero:Genentech: Research Funding; Bristol Myers Squibb: Other: Medical writing support, Research Funding; AbbVie: Research Funding. Montalban-Bravo:Rigel: Research Funding; Takeda: Research Funding. Patnaik:CTI BioPharma: Membership on an entity's Board of Directors or advisory committees; Kura: Research Funding; Epigenetix: Research Funding; StemLine: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal