In this issue of Blood, Izquierdo et al demonstrate that lymphoma cells with TP53 mutations reduce macrophage phagocytic capacity after chemoimmunotherapy (CIT), possibly as a result of programmed death ligand-1 (PD-L1)–expressing extracellular vesicles (EVs) (see figure), suggesting a role for checkpoint inhibition in the treatment of B-cell malignancies with TP53 loss.1
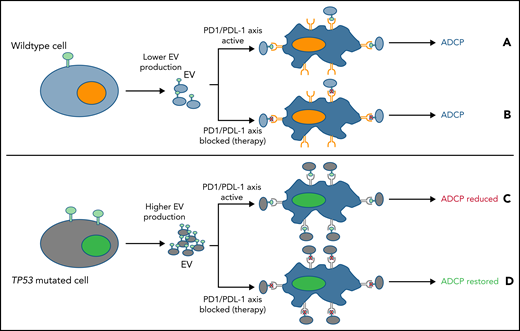
EV expressing PDL-1 may reduce macrophage phagocytosis. (A) Lower EV numbers and PD-L1 expression in tumors with a functional TP53 pathway may modestly, but not completely, reduce macrophage function. (B) However PD-L1/PD-1 axis inhibition (red cross), fully maximized macrophage phagocytic activity. (C) In contrast, cells with a TP53 mutation produced more EVs expressing PD-L1, which blocked macrophage phagocytosis to a greater extent. (D) However, when the PD-L1/PD-1 axis was inhibited (red X), ADCP was fully restored in lymphoma models with TP53 mutations, whereas WT cells remained unaffected.
EV expressing PDL-1 may reduce macrophage phagocytosis. (A) Lower EV numbers and PD-L1 expression in tumors with a functional TP53 pathway may modestly, but not completely, reduce macrophage function. (B) However PD-L1/PD-1 axis inhibition (red cross), fully maximized macrophage phagocytic activity. (C) In contrast, cells with a TP53 mutation produced more EVs expressing PD-L1, which blocked macrophage phagocytosis to a greater extent. (D) However, when the PD-L1/PD-1 axis was inhibited (red X), ADCP was fully restored in lymphoma models with TP53 mutations, whereas WT cells remained unaffected.
Directed therapies targeting Bruton tyrosine kinase such as ibrutinib and acalabrutinib or B-cell lymphoma 2 (Bcl-2) inhibitors such as venetoclax are commonly used to treat patients with B-cell non-Hodgkin lymphoma (NHL), including mantle cell lymphoma (MCL) and chronic lymphocytic leukemia (CLL). However, CIT still has an important place in treating patients with more aggressive lymphomas such as diffuse large B-cell lymphoma (DLBCL) and MCL. Monoclonal antibodies that target CD20 such as rituximab (R) have been approved in combination with fludarabine and cyclophosphamide (R-FC), and in combination with cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or combined with bendamustine (BR) to treat various lymphomas.2,3 Monoclonal antibodies such as rituximab aid tumor killing via antibody-dependent cellular cytotoxicity and antibody-dependent cellular phagocytosis (ADCP), which are mediated by natural killer cells and macrophages, respectively, and are key to the mechanism of action of CIT. Many patients relapse after CIT; therefore, understanding what mechanisms drive resistance and which pathways can be targeted to restore killing of resistant tumor cells is of paramount importance.
The frequency of TP53 mutations in B-cell NHL is heterogeneous, and 20% to 25% of patients with aggressive lymphoma and 6% to 10% of patients with CLL harbor these mutations.4,5 However, TP53 mutations are enriched in patients with transformed disease such as Richter’s syndrome (50% to 60%).4 The loss of TP53 is commonly known as a mediator of chemoresistance because of the role of TP53 in apoptosis. However, the impact of monoclonal antibody therapy on tumor cells that harbor TP53 mutations is less well defined.
Data from the study by Izquierdo et al suggest that tumor cells with mutations within the DNA damage response pathway, and in particular TP53, alter the tumor microenvironment by impairing macrophage-dependent phagocytosis via cellular crosstalk with EVs. The authors investigated this in a number of aggressive and indolent human and murine B-cell lymphoma models with consistent results. Although CIT treatment promoted tumor cell phagocytosis by macrophages compared with the untreated controls, the presence of tumor cells with TP53 mutations after CIT demonstrated impaired phagocytosis. To confirm that this was not a phenomenon specific to the cell line, primary tumor cells from patients with CLL or multiple myeloma (MM) with or without TP53 mutations were cultured with macrophages, and phagocytosis was quantitated. Tumor cells with a TP53 mutation similarly demonstrated impaired macrophage phagocytosis compared with the wild-type (WT) control. Interestingly, ADCP impairment was suggested to be independent of the TP53 clone size or of a specific mutation. Whether this observation in CLL and MM, which are diseases of the blood and bone marrow, respectively, can be replicated with primary tissue from aggressive lymphoma, in which tumor cells reside in the lymph nodes, has yet to be proven.
EVs are lipid bound vesicles that are secreted by cells in the extracellular space.6 EVs can be split into 3 main subtypes—microvesicles, exosomes, and apoptosis bodies—that are characterized by differences in their biogenesis, function, size, and content. EVs have been shown to contain proteins, lipids, and nucleic acids that have been suggested to alter cell function by reprogramming target cells using messenger RNA (mRNA), microRNA (miRNA), and long noncoding RNA (lncRNA).7,8 Indeed, in the Izquierdo et al study, mice engineered to prevent EV production demonstrated a longer overall survival, suggesting that EVs may be involved in reducing phagocytosis by macrophages.
Izquierdo et al demonstrated that the size and miRNA abundance of the EVs produced by WT and TP53-mutated cells showed no significant differences. However, TP53-deficient cells released significantly higher numbers of EVs compared with the WT control, which may in some part explain why the TP53 clone size did not directly correlate with phagocytic impairment; only a few cells were capable of producing a large number of EVs. Interestingly, the TP53 mutation seemed to abrogate only FC receptor–mediated phagocytosis by macrophages because bead phagocytosis was unaffected.
Phagocytosis is known to be regulated by a number of “eat-me” and “don’t-eat-me” signals on the surface of the tumor cell. Izquierdo et al evaluated the expression of CD200 and CD47 on the surface of the tumor cells and the EVs because they are the most recognized of the don’t-eat-me receptors that directly interact with CD200R and SIRPα on the macrophage, respectively. There was no or low levels of CD47 and CD200 expressed on the EVs, respectively. However, the lack of CD47 and CD200 does not conclusively rule out the role of don’t-eat-me signals on macrophage phagocytosis, because several other markers such as the recently identified CD249 were not evaluated. In contrast to CD47 and CD200, Izquierdo et al noted that the checkpoint regulator PD-L1 was shown to be expressed on EVs from both WT and TP53-mutated cells. The team went on to demonstrate that PD-L1 blocked macrophage phagocytosis and that by blocking PD-L1 with atezolizumab or by preventing PD-L1 expression on the surface of the EVs from TP53-mutated cells, they were able to restore macrophage phagocytic capacity, which suggests a role for PD-L1/programmed cell death protein-1 (PD-1) in patients with TP53-mutated B-cell NHL. Izquierdo et al subsequently evaluated anti-PD-1 therapy and demonstrated superior results in mice treated with anti-PD-1, cyclophosphamide, and alemtuzumab compared with mice treated with cyclophosphamide and alemtuzumab combination therapy. The authors suggest that this may be a promising strategy because a recent phase 2 trial combining pembrolizumab with R-CHOP was reported to be safe and to yield complete response rates of 77%.10 However, the clinical trial by Smith et al10 did not clearly evaluate whether patients had a TP53 mutation. Therefore, the specific role of TP53 mutations in macrophage phagocytosis and the clinical effects of pembrolizumab still remain to be elucidated and clinical response could well be independent of TP53 mutation.
Conflict-of-interest disclosure: A.J.S. is an employee of Janssen R&D.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal