Abstract
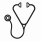
Introduction
Patients with E2A-PBX1 fusion are expected to have an aggressive disease course. Because of the rarity of this genotype (nearly 5% of pediatric and 3% of adult B-cell acute lymphoblastic leukemia (B-ALL) cases), a consensus on the clinical and prognostic characteristics of adult E2A-PBX1-positive B-ALL patients, especially in adult patients, has not yet been reached.
Patients and Methods
We retrospectively summarized our clinical findings from 137 B-ALL patients diagnosed with E2A-PBX at our centers from 2009 to 2019, including 56 adolescents/adults (≥15 years old) and 81 children (<15 years old). Genomic investigated analysis was performed on sufficient bone marrow at diagnosis, relapse and remission as well as matched hair follicle cells using somatic copy number variation detection (n=25), whole-exome sequencing (n=29), the next-generation sequencing pane (tumor only, n=14) and RNA sequencing (n=22).
Results
The proportions of E2A-PBX1-positive B-ALL in our centers were 5.3% (81/1526) in children, 4.6% (43/925) in AYA and 2.1% (15/713) in older adults. The complete remission rate among all E2A-PBX1-positive B-ALL patients in this study was 94.9% (129/136) after one course of induction chemotherapy. The 5-year overall survival (OS) and disease-free survival (DFS) rates of the whole cohort were 68.6% and 61.0%, respectively.
Allo-HSCT at CR1 in adolescents/adults could dramatically improve the 5-year prognoses (OS: 80.8% vs. 25.7%, P<0.001; DFS: 73.3% vs. 15.5%, P<0.001; cumulative incidence of relapse (CIR): 20.0% vs. 80.5%, P<0.001) (Figure 1A). Haploidentical-HSCT decreased the CIR compared with HLA-matched-HSCT in adolescents/adults (12.5% vs 58.3%, P=0.017) (Figure 1B).
A total of 12 patients received CD19-targeted CAR-T cell therapy for disease progression (Figure 1C), and 91.7% (11/12) of patients achieved remission. Two patients died of relapse, and 3 patients died of complications (One died of grade 4 CRS, one died of cerebral hemorrhage after transfusion, and the other one died of infection after 14 months). Three patients received CAR-T bridging to allo-HSCT, and all of them remained in remission within the follow-up period.
Univariate and multivariate analysis showed that t(1;19)(q23;p13) only (OS: P=0.020, HR=0.387, 95% CI: 0.174-0.862; DFS: P=0.004, HR= 0.375, 95% CI: 0.193-0.729; CIR: P=0.009, HR= 0.400, 95% CI: 0.200-0.799), Age (DFS: P=0.037, HR=1.009, 95% CI: 1.001-1.018; CIR: P=0.005, HR=1.025, 95% CI: 1.008-1.044) and the level of minimal residual disease (MRD) after induction chemotherapy (OS: P=0.020, HR=2.971, 95% CI: 1.185-7.452; DFS: P=0.002, HR= 3.218, 95% CI: 1.510-6.861; CIR: P=0.006, HR= 3.190, 95% CI: 1.406-7.246) were independent risk factors in E2A-PBX1-positive B-ALL (Figure 1D).
In the diagnosis samples, mutations in PBX1, PAX5, CTCF and SETD2, amplification of AKT3, and deletion of CDKN2A/B were common in the total cohort, while transcriptome differences were found in the cell cycle, NGF signaling pathway and transcriptional regulation by TP53 between adolescents/adults and children (Figure 2A,B). More DNA repair gene mutations were detected in the relapse samples (7.9% vs. 57.1%, P<0.001). The median number of subclones in E2A-PBX1-positive B-ALL at diagnosis was 2 (range 1-4). Patients with multiple subclones at diagnosis tended to have unfavorable 3-year prognoses (DFS: P=0.010; CIR: P=0.021). Leukemia clones with DNA repair gene mutations showed aggressive and treatment-refractory phenotypes in this subtype of ALL (Figure 2C).
Conclusions
Our study indicated that age, the level of MRD and DNA repair gene mutations were associated with E2A-PBX1-positive B-ALL outcomes. Allo-HSCT, especially haploidentical-HSCT, could improve the prognosis of adolescent/adult patients.
No relevant conflicts of interest to declare.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal