Abstract
The success of differentiation therapy is limited to acute promyelocytic leukemia (APL), and approaches to overcome the differentiation block in non-M3 AML have been unsuccessful. Nuclear hormone receptors (NHR) belong to ligand-inducible transcription factors that govern many cellular functions like differentiation, metabolism, and development. Retinoic Acid Receptor Alpha (RXRA) is a class of NHR that, when activated by all-trans retinoic acid (ATRA), successfully alleviates differentiation block in APL.
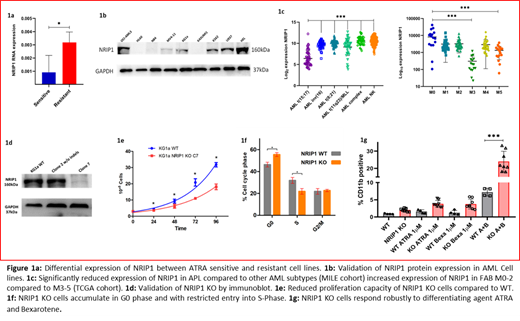
To identify the NHRs/cofactors that could mediate or prevent differentiation in AML, we examined the differentially expressed NHRs and cofactors between ATRA sensitive (ATs) (NB4 and HL60) vs. ATRA resistant (ATr) AML cell lines (KG1a, Hel, K562, MV4-11, and OCI-AML3). Nuclear Receptor Interacting Protein 1 (NRIP1), a corepressor known to prevent transactivation of ligand-activated NHRs preferentially, was one of the top upregulated targets in the ATr cell lines (3.5 fold increase in RNA expression, figure 1a ). Immunoblot analysis also showed a significant increase in NRIP1 protein expression in the ATr than ATs cell lines (Figure 1b). Further, probing for NRIP1 expression in the publicly available TCGA and MILE AML study cohorts showed decreased NRIP1 expression in the APL cohort compared to other AML subtypes. Methylation profile from CCLE database of the NRIP1 promoter in AML cell lines showed ATs cell lines to be highly methylated compared to the ATr cell lines, suggesting the involvement of NRIP1 in mediating differentiation block in non-M3 AML (Figure 1c). To further dissect the role of NRIP1 in mediating this differentiation block, we carried out experiments in the AML cell line KG1a (having primitive blast features, high expression of NRIP1, and unresponsive to ATRA). Using CRISPR-cas9, we developed an NRIP1 knock-out (KO) cell line (Figure 1d). NRIP1 KO cell line showed a significant reduction in proliferation rate (Doubling time 26.2 vs. 36.5Hrs p<0.05). Further, cell cycle analysis revealed that NRIP1 KO leads to increased accumulation of cells in the G0 phase than in the S-phase (Figure 1e & f). We next assessed the sensitivity of the NRIP1 WT/KO cells to retinoic acids ATRA and bexarotene. Cells were treated with 1µM ATRA / bexarotene or in combination for 72 hours and evaluated for differentiation using CD11b marker by flow cytometry. NRIP1 KO alone leads to a marginal increase in basal CD11b expression compared to the WT cells (Mean CD11b expression 2.03% Vs 0.91%). ATRA treatment further increased the CD11b expression to 3.8% in KO cells compared to 1.6% in the WT cells. A similar increase in CD11b expression was observed in bexarotene-treated cells (3.7% Vs 1.24%). Combination of ATRA with bexarotene showed a 3-fold increase in CD11b expression in the KO cells compared to the WT (23.9% Vs 7.2%, Figure 1g). NRIP1 KO diminishes its repressive action on ligand-activated RARA (ATRA activated) and RXRA (Bexarotene-activated), thereby allowing synergistic differentiation induction by retinoic acids in AML cells.
This study suggests a potential mechanism of differentiation inhibition mediated by corepressor NRIP1 in AML cells unresponsive to retinoic acids. Further in-depth analyses of molecular pathways governed by NRIP1 during ligand activation of NHRs are warranted to design differentiation therapies for AML.
Mathews: Christian Medical College: Patents & Royalties: US 2020/0345770 A1 - Pub.Date Nov.5, 2020; AML: Other: Co-Inventor.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal