In this issue of Blood, Enns et al1 provide compelling evidence that hepatocyte neogenin (NEO1) is essential for systemic iron homeostasis. NEO1 function in bone morphogenetic protein (BMP) signaling and the regulation of hepcidin expression requires its association with hemojuvelin (HJV), the protein mutated in juvenile hemochromatosis.
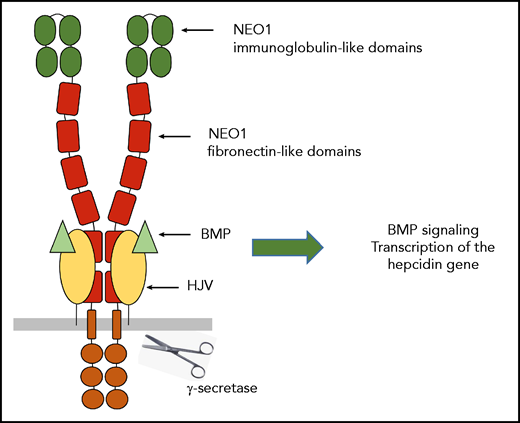
Neogenin is a transmembrane receptor belonging to the immunoglobulin superfamily (see figure). It has crucial functions in diverse cellular processes ranging from cell motility and adhesion to survival and differentiation. To mediate these functions, neogenin binds different ligands such as repulsive guidance molecules (RGM) and Netrin-1.2
HJV (or RGMC) is 1 of the 3 members of the RGM family, and thus is a ligand for NEO1. It is also coreceptor for BMP ligands. In mice, hemojuvelin is indispensable for hepcidin expression and iron homeostasis.3 Mutations in the HJV gene in humans reduce hepcidin expression in the liver and cause juvenile hemochromatosis.4 Interestingly, the most common juvenile hemochromatosis–causing mutation in HJV, G320V, disrupts its interaction with NEO1,5 suggesting that the function of HJV relies on its interaction with NEO1. However, the role of NEO1 in the maintenance of iron homeostasis has not always been recognized,6 in part because hepatoma cell lines in which most studies have been carried out are not appropriate models to study NEO1 induction of hepcidin expression.
NEO1 is composed of 4 N-terminal immunoglobulin-like domains, followed by 6 fibronectin type III–like domains, a single transmembrane helix, and an intracellular domain. Interactions between neogenin and HJV are mediated by fibronectin domains 5 and 6 of neogenin. Two HJV molecules act as molecular staples to bring together the juxtamembrane regions of 2 NEO1 receptors.2 The binding of HJV to NEO1 on the plasma membrane is essential for systemic iron homeostasis and triggers the γ-secretase–like protease cleavage of NEO1 to delete its cytoplasmic domain. This cleavage prevents the degradation of truncated NEO1, leading to its accumulation on plasma membrane. The consequence is an activation of BMP signaling and the transcription of the hepcidin gene by a mechanism that still needs to be clarified. Because HJV is also a coreceptor for BMPs, the NEO1/HJV complex could act as a shuttle for BMP ligands.
NEO1 is composed of 4 N-terminal immunoglobulin-like domains, followed by 6 fibronectin type III–like domains, a single transmembrane helix, and an intracellular domain. Interactions between neogenin and HJV are mediated by fibronectin domains 5 and 6 of neogenin. Two HJV molecules act as molecular staples to bring together the juxtamembrane regions of 2 NEO1 receptors.2 The binding of HJV to NEO1 on the plasma membrane is essential for systemic iron homeostasis and triggers the γ-secretase–like protease cleavage of NEO1 to delete its cytoplasmic domain. This cleavage prevents the degradation of truncated NEO1, leading to its accumulation on plasma membrane. The consequence is an activation of BMP signaling and the transcription of the hepcidin gene by a mechanism that still needs to be clarified. Because HJV is also a coreceptor for BMPs, the NEO1/HJV complex could act as a shuttle for BMP ligands.
The targeted deletion of the Neo1 gene in mice is by far the best means to determine the role of this gene in iron homeostasis. Mice with global Neo1 ablation have been produced in the past, and they have reduced hepcidin expression and severe iron overload. However, they also present with severe defects of neural, bone, and skeletal muscle development, and they die before postnatal day 30.7 Whether the iron accumulation resulted from increased absorption, tissue iron redistribution, or lack of iron utilization by erythropoiesis was not clear. That is why Enns et al generated a hepatocyte-specific conditional Neo1 knockout mouse model that circumvented the developmental defects of global Neo1 ablation. Similar to Hjv knockout mice, these mice exhibit reduced Bmp signaling, decreased hepcidin expression, high serum iron, and age-dependent iron accumulation in the liver. Increased hemoglobin levels ruled out the possibility that the iron overload in these mice was caused by reduced iron utilization by erythropoiesis. This is the first unambiguous demonstration that in vivo hepatocyte Neo1 is required for iron homeostasis.
Then, in careful replacement studies, the authors demonstrated that Neo1-mediated hepcidin expression depends on its interaction with Hjv. They had previously shown that, in Hjv knockout mice, the low Bmp signaling and low hepcidin expression were fully corrected by expression of transduced wild-type Hjv but not by expression of Hjv mutants with disrupted interaction with Neo1.8 In this study, they showed that transducing wild-type Neo1, but not the Neo1 mutant that abolishes interaction with Hjv, into the liver of hepatocyte-specific Neo1 knockout mice was able to correct the low Bmp signaling, low hepcidin expression, and high serum iron status. These observations convincingly demonstrate that appropriate hepcidin expression in the mouse requires an interaction between Neo1 and Hjv.
In these conditions, how does NEO1 induce hepcidin expression? Surprisingly, the authors’ additional studies in hepatoma cells do not support the idea that NEO1 regulates iron homeostasis by inhibiting HJV shedding as previously proposed.7 Their data favor the idea that HJV shedding constitutes the major pathway of cellular HJV turnover rather than the control of hepcidin expression. Therefore, the authors sought insights into a different mechanism that might explain the induction of hepcidin expression by hepatocyte NEO1.
Earlier studies indicate that HJV and NEO1 traffic separately to the plasma membrane and that they interact after reaching the plasma membrane. Consistent with this observation, the Hjv level in liver membrane extracts was not altered by disrupting its interaction with Neo1. However, the levels of membrane-associated Neo1 were increased by Hjv interaction, suggesting that the interaction with Hjv may stabilize Neo1 at the cell membrane and prevent rapid internalization and degradation. It appears that the binding of Hjv to Neo1 on the plasma membrane triggers the γ-secretase–like protease cleavage of Neo1 to delete its cytoplasmic domain. Interestingly, in Hjv knockout mice, the marked decrease in protease-cleaved Neo1 was largely restored by expressing wild-type Hjv but not Hjv that fails to interact with Neo1. It may be the protease cleavage of Neo1 that prevents its degradation and leads to its accumulation on plasma membrane, and the authors proposed that the truncated form of Neo1 may act as a scaffold to facilitate Bmp signaling and the transcription of the hepcidin gene.
In summary, Enns et al provide persuasive data supporting the importance of hepatocyte neogenin in the maintenance of iron homeostasis. When the different genetic backgrounds of the available mouse models are taken into account, it clearly appears that ablation of hepatocyte neogenin leads to repression of hepcidin expression and accumulation of liver nonheme iron that are as severe as those observed in mice lacking Hjv or Bmp6 and much more severe than those observed in mice lacking Hfe or Tfr2, the genes involved in adult-onset hemochromatosis.9 This work adds neogenin to the list of molecules that have a major regulatory role on hepcidin expression. Nevertheless, given the other crucial functions of neogenin, it is unlikely that mutations in the NEO1 gene are compatible with normal development and possibly found in yet uncharacterized juvenile patients with hemochromatosis. Whereas there is no more doubt that NEO1/HJV interaction activates BMP signaling and upregulates hepcidin expression, the question now is how. Could the NEO1/HJV complex act as a shuttle for BMP ligands? Based on the observations made by Healey et al,10 it is tempting to hypothesize that the NEO1/HJV complex might sequester BMP ligands at the hepatocyte membrane and transport them via the endosomal pathway to compartments enriched in BMP receptors where, after dissociation from HJV, they would bind to their receptors, activate signaling, and induce hepcidin expression. This is an opportunity for future research.
Conflict-of-interest disclosure: The author declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal