Background: The overall participation of cancer patients in interventional clinical trials in the United States remains very low, with ~5% of patients being enrolled in clinical trials nationwide. The outcomes of patients with MM have improved significantly over the past decade, but available data suggest that the participation rates for patients with MM is comparable to other cancers. The drivers of participation and the potential impact of clinical trial participation have not been systematically studied in MM.
Patients and Methods: We identified 228 patients who were enrolled into clinical trials for initial therapy of newly diagnosed MM between 2004 and 2018, and 4 controls for each of these patients. Controls were patients with NDMM, who were diagnosed closest in time to the index patients and did not participate in an interventional treatment trial. Various baseline characteristics as well as overall survival were compared between the two groups.
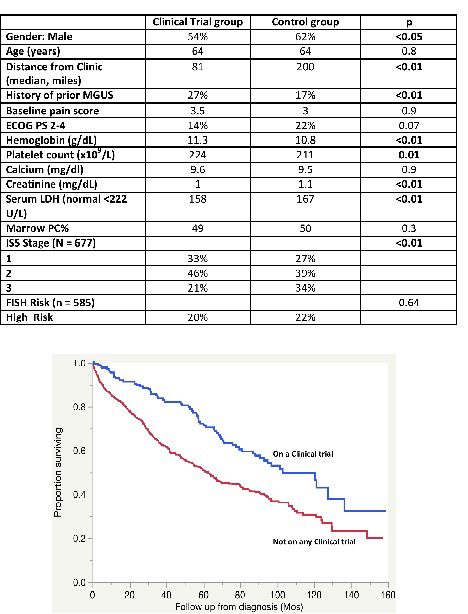
Results: The baseline characteristics of the two groups are as shown in the Table. Patients who entered clinical trials were more likely to be female, resided closer to the clinic, and were more likely to have a prior history of MGUS. They were more likely to have higher ISS Stage, and a higher serum LDH, but there was no difference in the FISH risk status. Other indices of disease burden such as lower hemoglobin and platelets, higher serum creatinine were all seen more often in the control group, but may have been influenced by the trial entry criteria. Looking at the outcomes, the overall survival was longer among those enrolled into clinical trials compared to those who did not [median 103 (95% CI; 86, 136) vs. 63 (95% CI; 53, 69) months, p<0.001 (Figure).
Conclusions: The current study provides important clues regarding demographic determinants of trial participation and disease biology related features that reflect likelihood of trial participation. Overall survival was significantly longer among the trial participants, which likely represent a mix of reasons including baseline status of patients, intensity of monitoring and efficacy of novel treatment approaches.
Kapoor:Janssen: Research Funding; Celgene: Honoraria; Cellectar: Consultancy; Sanofi: Consultancy, Research Funding; Amgen: Research Funding; Glaxo Smith Kline: Research Funding; Takeda: Honoraria, Research Funding. Dispenzieri:Alnylam: Research Funding; Janssen: Consultancy; Pfizer: Research Funding; Takeda: Research Funding; Celgene: Research Funding; Akcea: Consultancy; Intellia: Consultancy. Gertz:Ionis: Honoraria; Alnylam: Honoraria; Prothena: Honoraria; Celgene: Honoraria; Janssen: Honoraria; Spectrum: Honoraria, Research Funding. Lacy:Celgene: Research Funding. Dingli:Karyopharm: Research Funding; Rigel: Consultancy; alexion: Consultancy; Janssen: Consultancy; Millenium: Consultancy. Leung:Omeros: Research Funding; Aduro: Membership on an entity's Board of Directors or advisory committees; Takeda: Research Funding; Prothena: Membership on an entity's Board of Directors or advisory committees. Russell:Imanis: Equity Ownership. Kumar:Takeda: Research Funding; Janssen: Consultancy, Research Funding; Celgene: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal