Background: Chronic lymphocytic leukemia (CLL) is the most common chronic leukemia. Patients in active surveillance with untreated high-risk CLL (defined as presence of del17p or TP53, del11q or ATM mutation or unmutated IGVH) have an unmet need as they tend to progress rapidly and develop resistance to standard therapies. There is evidence that deferring treatment in high-risk patients may be detrimental due to a more aggressive course of disease and that patients with clonal evolution into high-risk features may fare even worse prognosis (Shanafelt et al, JCO 2006). The CLL12 trial (ibrutinib for asymptomatic high risk CLL patients, Langerbeins et al ICML2019) showed event (EFS) and progression (PFS) free survival benefit when compared to placebo. In addition, T-cell dysfunction and immunosuppressive environment have been shown in CLL. The rationale of the trial is that the combination of PD1:PDL1 Blockade (PDB) and ibrutinib will reverse T cell dysfunction commonly seen in CLL patients, potentiating more robust anti-infective and anti-tumor immune responses.
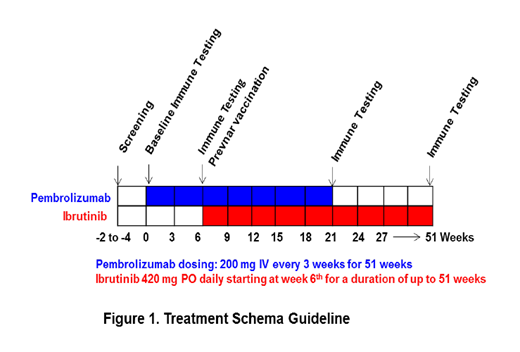
Methods: This is a phase 2 clinical trial. Enrollment goal: 25. Treatment will consist of the combination of pembrolizumab (Pem) 200 mg IV every 3 weeks and ibrutinib 420 mg daily orally (to be started after 2nd infusion of PDB) for up to 51 weeks, as depicted in Figure 1. Eligibility criteria includes: Age > 18, confirmed diagnosis of CLL, presence of at least 1 high risk factor for CLL (Del17p, Del11q and or unmutated IGVH), and not meeting iwCLL criteria to start treatment. Primary endpoints are incidence of complete remission (CR) and time to CLL response. Secondary endpoints: overall response rate (ORR), restoration of immune response (decreased markers of T-cell exhaustion and improvement in quantitative immunoglobulins), safety, PFS. Exploratory biomarkers include evaluation of T-cell and B-cell function and cytokine profile at different time points through therapy.
Chavez:Karyopharm: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Bayer: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Kite: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees. Nodzon:Pfizer: Consultancy; Pharmacyclics: Consultancy; Genentech: Consultancy, Other: Speaker Fees; Abbvie: Other: Speaker Fees. Pinilla Ibarz:Novartis: Consultancy; Bristol-Myers Squibb: Consultancy; Takeda: Consultancy, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Janssen: Consultancy, Speakers Bureau; Teva: Consultancy; TG Therapeutics: Consultancy; Bayer: Speakers Bureau; Sanofi: Speakers Bureau.
The combination of PD1:PDL1 Blockade (PDB) and ibrutinib will reverse T cell dysfunction commonly seen in CLL patients, potentiating more robust anti-infective and anti-tumor immune responses
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal