Acute myeloid leukemia(AML) originates from the abnormal clonal proliferation of myeloblast which often combined with clinical symptoms. Cytogenetic and molecular abnormalities are frequent in AML patience. To date, the driver genes for leukemia remain largely undiscovered. Monoclonal immunoglobulinemia is a group of diseases caused by excessive proliferation of plasma cells or immunoglobulin-producing lymphoid plasma cells and B lymphocytes. It can develop into malignant plasma cell disease. Herein, we report a AML patient was concomitant with monoclonal immunoglobulinemia, the patient was also accompanied by BCOR mutation and TLS-ERG fusion gene.
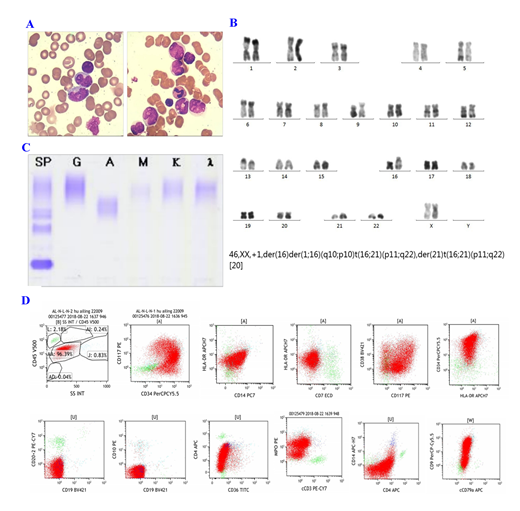
A 55-year-old married female was admitted into our hospital due to repeated edema for 3 weeks. On admission, peripheral blood counts: PLT142×10^9/L, HB77g/L↓, WBC35.2×10^9/L.Bone marrow examination showed the mononuclear cell system proliferated actively, and the primitive infantile monocytes accounted for 86%. Cell morphology suggested M5b(Figure1A ). Fusion gene screening in bone marrow revealed that TLS-ERG expression. Immunophenotype of bone marrow cell:Abnormal myeloid primitive cells accounted for 96.39% of the nuclear cells,expressCD33, CD13, CD123, CD34, CD9, MPO(Figure 1D). Karyotype analysis of bone marrow cells showed in Figure 1B. Thus, AML was diagnosed. Next-generation DNA sequencing technology showed that BCOR (51.7%),PLCG1(49.9%),DIS3(48.4%),BRAF(51.6%), JAK2(45.1%) ,JAK3(49.0%) were mutated. Meanwhile, we found that Peripheral blood immunofixation electrophoresis showed that Gamma region is seen with a monoclonal light chain lambda component((Figure 1C.).Then, the patient underwent one cycle of IA(Idabisine hydrochloride 10mg d1-4, cytarabine 0.075g q12h d1-7). Twenty-five after chemotherapy onset, bone marrow examination showed that primitive and immature monocytes accounted for 3%. Chromosome become normal. Minimal residual disease(MRD):0.01%. The disease reached complete remission(CR). Peripheral blood immunofixation electrophoresis turned negative. Fusion gene detection showed that TLS-ERG turned negative. BCOR mutation was not detected by Next-generation DNA sequencing. Mutations of PLCG1,DIS3,BRAF,JAK2,JAK3 still exist.
Monoclonal immunoglobulinemia and AML are both clonal diseases, but originated from different clones. This case has both malignant clones of granulocyte stem cell and malignant clones of B line, so it is worthy of discussion. By comparing CR before and after we found that while the patient's M protein turned negative, the TLS-ERG fusion gene and BCOR gene mutation also disappeared. The TLS-ERG fusion gene is formed by the rearrangement of TLS and ERG genes on chromosomes 16 and 21. The current study holds that the expression of this fusion gene indicates rapid disease progression and poor prognosis. BCOR mutations can be found in AML and often coincide with DNMT3 gene mutations, suggesting it may affect the occurrence of leukemia through epigenetics. BCOR is a newly discovered corepressor of BCL-6, which can play a supporting role when BCOR combines with DNA; when BCOR is overexpressed, it can enhance the inhibition of BCL-6. BCL-6 is highly expressed in tumor cells,it encodes transcriptional repressors which are required for the formation of germinal center and may affect apoptosis. We thinked that the monoclonal immunoglobulinemia of this patient may caused by the BCOR abnormal expression which increased the inhibitory effect of BCL-6 and affect the apoptosis of B cells, and B cells continue to secrete immunoglobulin. BCOR mutations are associated with poor prognosis. The patient with TLS-ERG fusion gene which is a poor prognosis gene.However, the BCOR gene mutation site is a non-hot spot mutation which has few clinical studies. Whether the BCOR gene mutation results in the combination of the two diseases requires further study.
Acknowledgment:The research was supported by fundings of the public technology research projects of Yiwu,China (2016-S-05), the key medical discipline of Yiwu,China(Hematology,2018-2020),and the academician workstation of the Fourth Affiliated Hospital of Zhejiang University School of Medicine.
Correspondence to: Dr Jian Huang, Department of Hematology, The Fourth Affiliated Hospital of Zhejiang University School of Medicine. N1 Shangcheng Road. Yiwu, Zhejiang, Peoples R China.
Email: househuang@zju.edu.cn
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal