Introduction:
Management of elderly patients with Diffuse Large B-Cell Lymphoma (DLBCL) is challenging. A simplified Comprehensive Geriatric Assessment (sCGA) based on ADL (Activity of Daily Living), IADL (Instrumental ADL) and CIRS-G (Comorbidity Index Rating Scale for Geriatrics) scales has demonstrated to be better than clinical judgement to stratify patients' outcome but has never been included in initial assessment. To further assess the impact of sCGA on patients' outcome, we conducted a prospective observational study on a large series of elderly patients with DLBCL.
Methods:
Patients were enrolled if 65 year old or older, with an untreated de novo DLBCL. sCGA was available at a web based platform that classified patients as FIT, UNFIT, and FRAIL, as shown in Table 1. Treatment choice was left at physician discretion. According to anthracycline dose, therapy was classified as curative (≥70% of full anthracycline dose), intermediate (<70%) or palliative (no anthracycline). Primary study endpoint was Overall Survival (OS).
Results:
From December 2013 to December 2017, 1353 patients have been registered by 37 centres and 1207 were eligible. Median age was 76 years (65-94), 68% had stage III-IV, and 55% had an International Prognostic Index(IPI) ≥3; 500 (42%), 304 (25%), and 403 (33%) were classified as FIT, UNFIT and FRAIL, respectively. Data on treatment were available in 1164 patients: rituximab was used in 96% of patients; treatment was curative in 89%, 53%, and 36% of FIT, UNFIT, and FRAIL patients, respectively; intermediate in 10%, 39%, and 31%, palliative in 0%, 8%, and 33% of patients.
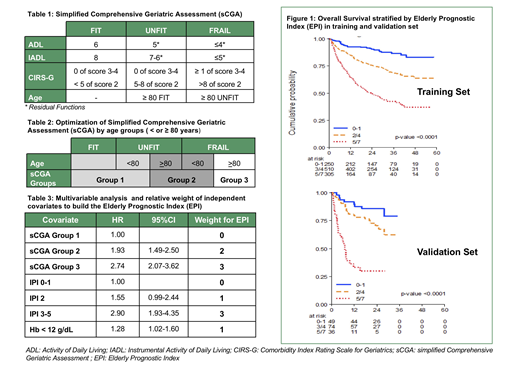
The OS was available in 1158 out 1164 cases. With a median follow up of 30 months (1-59) 3y-OS was 64% (95% CI 61% to 67%). According to sCGA the OS was significantly different among the 3 geriatric groups. Correlation with OS was improved when sCGA was integrated with age < or ≥ 80 years to define 3 groups of patients (Table 2): FIT and UNFIT younger than 80 years (sCGA Group 1; 55%, 3 yr OS 75%), UNFIT ≥ 80 years and FRAIL younger than 80 years (sCGA Group 2: 28%, 3yr OS 58%), FRAIL ≥ 80 years (sCGA Group 3: 17%; 3yr OS 43%).
Univariable and multivariable analysis for OS was conducted using the 3 sCGA groups and other clinical and laboratory features. The 3 sCGA groups were shown as independent prognostic factors with IPI and with anemia (Hb < 12 g/dl). We used results of multivariable analysis to build a categorical prognostic index assigning different weights to prognostic features based on their Hazard Ratio (HR) (Table 3).
The Elderly Prognostic Index (EPI) was defined as the score obtained from the sum of the weights and allowed to define 3 risk groups: Low Risk (LR: score 0-1; 23% of patients); Intermediate Risk (IR; score 2-4; 48%); High Risk (HiR; score 5-7; 29%).
The 3 EPI risk groups had a different 3 year OS of 87%(95%CI 81-91), 69%(95%CI 63-73), and 42% (95%CI 36-49); HR for IR vs LR 2.57 (1.72, 3.84); HiR vs LR 6.21(4.17 -9.25), HiR vs IR 2.42 (1.91-3.05) (Figure1).
Regarding treatment modality, curative, intermediate and palliative therapies were adopted in 89%, 10%, and 1% of the LR group; 70%, 24%, 7% of the IR group, and 37%, 35%, 28% of the HiR group.
The model was internally validated by means of 1000 procedures confirming good performance (slope shrinkage 0.935 and c-Harrell 0.675 in validation sample compared with 0.682 in training sample).
The EPI was also tested in an external validation data set that was identified from the pivotal study of sCGA in DLBCL (N=172 patients, Tucci A. et al, Leuk Lymph, 2015) (Figure 1).
Conclusion:
Using data from this large prospective observational study on elderly DLBCL patients we were able to build a new prognostic index that allows to identify 3 risk groups with significant differences in terms of 3 years OS. The EPI is the first index that integrates geriatric assessment with clinical features and contributes to improving management and clinical research in elderly patients with DLBCL.
Spina:Servier: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee; Sandoz: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Mundipharma: Membership on an entity's Board of Directors or advisory committees, Other; Roche: Other: lecture fee; Teva: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee; GILEAD: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee; Celgene: Other: lecture fee; BMS: Other: lecture fee; Sanofi Genzyme: Other: lecture fee; CTI: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee; Menarini: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee, Research Funding; Takeda: Other: lecture fee; Janssen-Cilag: Membership on an entity's Board of Directors or advisory committees, Other: lecture fee; Pfizer: Membership on an entity's Board of Directors or advisory committees. Merli:Janssen: Honoraria; Takeda: Honoraria, Other: Travel Expenses; Gilead: Honoraria; Mundipharma: Honoraria; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel expenses, Research Funding; Sandoz: Membership on an entity's Board of Directors or advisory committees; Teva: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees, Other: Travel Expenses. Cavallo:Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees. Ladetto:Roche: Honoraria; AbbVie: Honoraria; J&J: Honoraria; Celgene: Honoraria; ADC Therapeutics: Honoraria; Acerta: Honoraria, Speakers Bureau; Pfizer: Honoraria, Speakers Bureau. Chiappella:Celgene: Other: advisory board, Speakers Bureau; Janssen: Other: advisory board, Speakers Bureau; Servier: Other: advisory board, Speakers Bureau; Roche: Speakers Bureau; Teva: Speakers Bureau. Nassi:Takeda: Consultancy; Janssen: Consultancy; Merck: Consultancy. Ferrero:Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Speakers Bureau; Servier: Speakers Bureau; EUSA Pharma: Membership on an entity's Board of Directors or advisory committees. Luminari:ROCHE: Membership on an entity's Board of Directors or advisory committees; CELGENE: Membership on an entity's Board of Directors or advisory committees, Other: Travel Grant; GILEAD: Other: Lecturer; TAKEDA: Other: Travel Grant.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal