Introduction:
Asparaginase (ASP) has demonstrated a survival benefit in pediatric patients (pts) with acute lymphoblastic leukemia (ALL) and is now part of standard-of-care frontline treatment. As a result, asparaginase preparations have been incorporated into the treatment of adult ALL to improve outcomes. Pegaspargase (PEG-ASP), a modified version of asparaginase with prolonged asparagine depletion, appears to be safe in adults up to age 40 (Stock, et al., Blood, 2019), but is associated with a unique spectrum of toxicities, the risks of which appear to increase with age. Therefore, the safety of PEG-ASP remains a significant concern in older adults w/ ALL.
Methods:
We conducted a single center retrospective chart review of pts age ≥40 years who received PEG-ASP as part of frontline induction/consolidation or reinduction, between March 2008 and June 2018 at Memorial Sloan Kettering Cancer Center. The primary objective was to evaluate the tolerability and toxicity of PEG-ASP based on the incidence and severity of ASP-related toxicities (hypersensitivity reactions, hypertriglyceridemia, hyperbilirubinemia, transaminitis, pancreatitis, hypofibrinogenemia, etc) according to the Common Terminology Criteria for Adverse Events, version 4.03. Laboratory values recorded were either the peak or the nadir, the more appropriate for toxicity assessment, within a 4-week period following PEG-ASP administration. Secondary objectives were to determine the total number of doses of PEG-ASP administered in comparison to the number of doses intended, and to characterize the rationale for PEG-ASP discontinuation when applicable. Fisher's exact test was used to compare the incidence of PEG-ASP toxicities with respect to pt and treatment characteristics (regimen, age, BMI, gender, Philadelphia chromosome positive (Ph+) vs. Ph-, presence of extramedullary disease, PEG-ASP dose). P values were not adjusted for multiple comparisons.
Results:
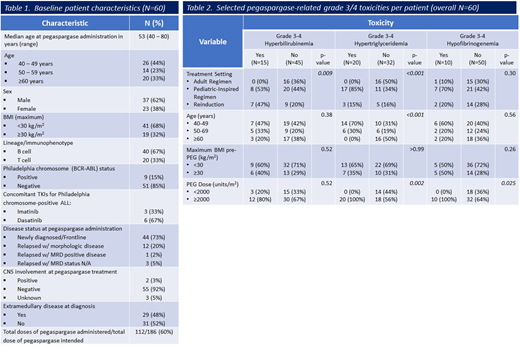
We identified 60 pts with ALL (40 B-ALL and 20 T-ALL) who received at least one dose of PEG-ASP. Nine pts were Ph+. The median pt age at initiation of the treatment was 53, (range, 40 to 80), and 19 pts had a BMI ≥30 kg/m2. Forty-four pts received treatment for newly diagnosed ALL, and 16 pts for relapsed disease. Table 1 lists pt baseline characteristics. Among the 44 pts with newly diagnosed ALL, 27 pts received PEG-ASP as part of pediatric or pediatric-inspired regimens at doses of 2000 - 2500 units/m2, and 1 pt received a modified dose of 1000 units/m2 due to age. The remaining 16 pts received PEG-ASP at doses of 1000 - 2000 units/m2 for consolidation, per established adult regimens (ALL-2 and L-20; Lamanna, et al., Cancer, 2013). Grade 3/4 ASP-related toxicities with a >10% incidence included: hyperbilirubinemia, transaminitis, hypoalbuminemia, hyperglycemia, hypofibrinogenemia, and hypertriglyceridemia. Frontline treatment regimens in which PEG-ASP was used in consolidation cycles only (ALL-2, L-20) were associated w/ a lower incidence of hyperbilirubinemia (p=0.009) and hypertriglyceridemia (p<0.001) compared to those regimens that included PEG-ASP during induction (pediatric/pediatric-inspired regimens) (Table 2). Younger age (40-59 vs. ≥60 years) was associated with a greater risk of hypertriglyceridemia (p<0.001) and higher PEG-ASP dose (≥2000 vs. <2000 units/m2) was associated with a greater risk of hypertriglyceridemia and hypofibrinogenemia (p=0.002 and p=0.025, respectively). Thirty-eight pts (63%) received all intended doses of PEG-ASP. Six pts stopped PEG-ASP to proceed to allogeneic hematopoietic stem cell transplantation (5 in CR1, 1 in CR2), and 7 pts stopped for hypersensitivity reactions. Hepatotoxicity was the only ASP-related toxicity that led to PEG-ASP discontinuation occurring in 5 pts (hyperbilirubinemia, N=4; transaminitis, N=1). The total number of intended doses of PEG-ASP based on regimens used was 186, and 112 were administered.
Conclusion:
PEG-ASP was incorporated into the treatment of 60 adult ALL pts age ≥40, with manageable toxicity. Seven pts discontinued PEG-ASP due to hypersensitivity reactions and 5 discontinued due to hepatotoxicity, but other reported toxicities did not lead to PEG-ASP discontinuation and the majority of the pts completed all intended doses of PEG-ASP. This study suggests that with careful monitoring, PEG-ASP can safely be administered in adults ≥40 years of age.
Rajeeve:ASH-HONORS Grant: Research Funding. Tallman:UpToDate: Patents & Royalties; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Cellerant: Research Funding; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; ADC Therapeutics: Research Funding; Biosight: Research Funding; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees. Geyer:Dava Oncology: Honoraria; Amgen: Research Funding. Park:Takeda: Consultancy; Allogene: Consultancy; Amgen: Consultancy; AstraZeneca: Consultancy; Autolus: Consultancy; GSK: Consultancy; Incyte: Consultancy; Kite Pharma: Consultancy; Novartis: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal