Background: The combination of lenalidomide+rituximab (R2) recently showed superior efficacy vs R-placebo in patients (pts) with R/R iNHL (Leonard et al. J Clin Oncol 2019). Based on these AUGMENT study results, R2 was approved by the US FDA for treatment of adult pts with previously treated follicular lymphoma (FL) or marginal zone lymphoma (MZL). Advanced age at diagnosis is a risk factor in pts with iNHL. We performed post-hoc subgroup analyses by age from AUGMENT and data here focus on pts age ≥ 70 y.
Methods: AUGMENT (NCT01938001) is a multicenter, double-blind, randomized phase III study of R2 vs R-placebo in pts with FL grade 1-3a or MZL previously treated with ≥ 1 systemic therapy with R/R disease but not refractory to rituximab. Pts were randomized 1:1 to R2 or R-placebo. R2 was oral lenalidomide 20 mg/d, d1-21/28 for 12c plus rituximab IV 375 mg/m2 weekly in c1 and d1 of c2-5. R-placebo was rituximab+placebo on the same schedule. The primary endpoint was progression-free survival (PFS) per 2007 IWG response criteria (without PET) as assessed by IRC (central review). Secondary endpoints included overall response rate (ORR), complete response (CR), time to next anti-lymphoma treatment (TTNLT) and safety. Post-hoc analyses were performed by dividing pts into age < 70 y and ≥ 70 y subgroups, the latter group considered unfit for chemotherapy.
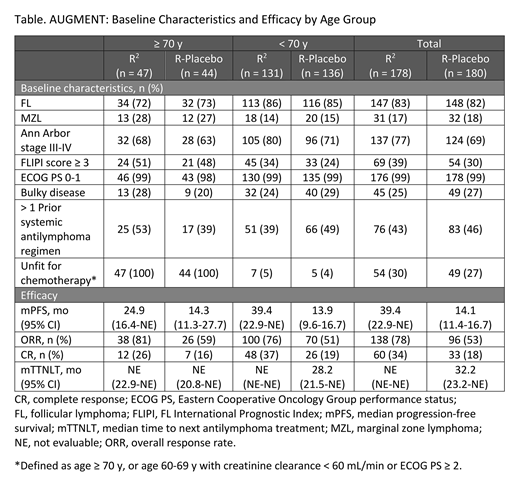
Results: Of 358 pts randomized (R2, n = 178; R-placebo, n = 180), 267 pts were age < 70 y (R2, n = 131; R-placebo, n = 136), and 91 pts were age ≥ 70 y (R2, n = 47; R-placebo, n = 44). Baseline characteristics including histology, disease status, and prior treatments are shown in the table and were similar across treatment arms in pts ≥ 70 y. At a median follow-up of 28.3 mo, the study met its primary endpoint of PFS, with a hazard ratio (HR) of 0.46 (95% CI, 0.34-0.62; P < 0.0001) in the overall population. R2 had superior PFS vs R-placebo in both < 70 and ≥ 70 y subgroups, with HR of 0.41 (95% CI, 0.29-0.59) and HR of 0.66 (95% CI, 0.37-1.18), respectively. In pts ≥ 70 y, median PFS with R2 vs R-placebo was 24.9 vs 14.3 mo; ORR/CR was 81%/26% vs 59%/16%; and TTNLT was not reached in either arm. Efficacy results for all pts and those < 70 y are reported in the table; notably in pts receiving R2, mPFS was longer in pts < 70 y vs ≥ 70 y (39.4 mo [95% CI, 22.9-NE] vs 24.9 mo [95% CI, 16.4-NE]). In pts ≥ 70 y, any-grade adverse events (AEs) with a ≥ 10 % difference between R2 vs R-placebo included neutropenia (63% vs 11%), constipation (33% vs 16%), cough (33% vs 16%), leukopenia (26% vs 2%), anemia (24% vs 9%), pyrexia (24% vs 9%), pruritus/pruritus generalized (24% vs 2%), muscle spasms (22% vs 11%), rash/rash maculopapular (22% vs 5%), headache (20% vs 9%), thrombocytopenia (17% vs 2%), dyspepsia (13% vs 2%), influenza (13% vs 2%), back pain (7% vs 18%), and nasopharyngitis (4% vs 16%). Also, tumor flare was reported in 9% vs 0% of pts, respectively. In pts ≥ 70 y, 75% of R2 pts vs 36% of R-placebo pts had ≥ 1 grade 3/4 AE, mainly due to neutropenia (50% vs 7%). All other grade 3/4 AEs occurred in < 10% of pts ≥ 70 y in both treatment arms. One grade 5 AE occurred in pts ≥ 70 y (R-placebo arm). In the R2 arm, the median number of treatment cycles was 12 for both the < 70 y vs ≥ 70 y subgroups; however, fewer older pts completed 12 cycles of lenalidomide (76% vs 57%), and more started lenalidomide at the lower dose of 10 mg (6% vs 35%) because of low creatinine clearance, respectively. In the R2 < 70 y and ≥ 70 y subgroups, the average daily dose of lenalidomide was 17.9 mg/d (range, 5.6-20.0) and 14.4 mg/d (range 4.2-20.0), and median relative dose intensity was 95% and 86%, respectively.
Conclusions: Similar to the results in the original population, R2 showed superior efficacy vs rituximab monotherapy (plus placebo) as measured by the primary end point of PFS and secondary end points of ORR and CR in pts with R/R FL grade 1-3a and MZL irrespective of age. The efficacy and safety profiles of R2 and R-placebo in pts ≥ 70 y were similar to those reported in the overall population. Older pts treated with R2 vs R-placebo had superior mPFS (24.9 vs 14.3 mo). They were more likely to start lenalidomide at a lower dose and had lower median dose intensity which may have contributed to their shorter mPFS vs younger pts receiving R2. These data show that R2 maintained efficacy improvements vs R-placebo in pts ≥ 70 y, despite higher unfit status and lower overall lenalidomide treatment/exposure. Thus, R2 is an effective and available treatment option for pts with iNHL, including those with advanced age.
Trněný:Amgen: Consultancy, Honoraria; F. Hoffmann-La Roche: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Celgene: Consultancy; MorphoSys: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; Gilead Sciences: Consultancy, Honoraria. Leonard:Nordic Nanovector: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy; AstraZeneca: Consultancy; Celgene: Consultancy; Bayer Corporation: Consultancy; Epizyme, Inc: Consultancy; Karyopharm Therapeutics: Consultancy; Sutro Biopharma: Consultancy; Merck: Consultancy; Gilead: Consultancy; BeiGene: Consultancy; ADC Therapeutics: Consultancy; Sandoz: Consultancy; Akcea Therapeutics: Consultancy; Miltenyi: Consultancy; MorphoSys: Consultancy. Nowakowski:F. Hoffmann-La Roche Ltd: Research Funding; NanoString: Research Funding; Selvita: Membership on an entity's Board of Directors or advisory committees; Curis: Research Funding; Bayer: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Genentech, Inc.: Research Funding; MorphoSys: Consultancy, Research Funding. Izutsu:Chugai, Celgene, Daiichi Sankyo, Astra Zeneca, Eisai, Symbio, Ono, Bayer, Solasia, Zenyaku, Incyte, Novartis, Sanofi, HUYA Bioscience, MSD, Astellas Amgen, Abbvie, ARIAD, Takeda, Pfizer: Research Funding; Kyowa Kirin, Eisai, Takeda, MSD, Chugai, Nihon Medi-physics, Janssen, Ono, Abbvie, Dainihon Sumitomo, Bayer, Astra Zeneca, HUYA Japan, Novartis, Bristol-Byers Squibb, Mundi, Otsuka, Daiichi Sankyo, Astellas, Asahi Kasei: Honoraria; Celgene: Consultancy; Eisai, Symbio, Chugai, Zenyaku: Research Funding; Eisai, Chugai, Zenyaku: Honoraria. Fowler:TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding. Thieblemont:Roche: Honoraria, Research Funding; Gilead: Honoraria; Novartis: Honoraria; Kyte: Honoraria; Janssen: Honoraria; Celgene: Honoraria; Cellectis: Membership on an entity's Board of Directors or advisory committees. Zinzani:Verastem: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; MSD: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Eusapharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Consultancy; Celltrion: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen-Cilag: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Servier: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sandoz: Membership on an entity's Board of Directors or advisory committees; Immune Design: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Portola: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Kyowa Kirin: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TG Therapeutics: Honoraria, Speakers Bureau. Kalambakas:Celgene Corporation: Employment, Equity Ownership. Czuczman:Celgene Corporation: Employment, Equity Ownership. Fustier:Celgene Corporation: Employment, Equity Ownership. Wu:Celgene Corporation: Employment, Equity Ownership. Gribben:Janssen: Consultancy, Honoraria, Research Funding; Acerta/Astra Zeneca: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal