Introduction. Our TT regimens for newly diagnosed multiple myeloma (MM) incorporate novel agents into a sequential treatment program comprising induction, tandem autologous stem cell transplantation and consolidation followed by 3 years of maintenance. Herein, we report the very long-term results in a large cohort of 1986 patients treated on successive TT protocols, the most mature of which (TT1, 2, and 3a) have a median follow-up ranging from 12.8 to 23.1 yrs.
Methods. TT1 (1990) was followed by TT2 (1998), which introduced Thalidomide (T) in a randomized fashion. TT3 used bortezomib (V) throughout, with TT3a (2003) and 3b (2006) having different maintenance. TT3a used in year 1 of maintenance V, T and dexamethasone (D) and in years 2 and 3 TD. TT3b introduced lenalidomide (R) during maintenance for 3 years together with V and D. TT4 (2009) only enrolled patients with GEP-defined low risk disease and randomized patients to a standard arm or light arm using a similar regimen as TT3b. TT5 (2009) was specifically designed for patients who have a high 70-gene score and employed a dose dense treatment approach. Finally, TT6 (2009) accrued previously treated, patients irrespective of GEP-defined risk using a treatment schema similar to that used in TT5. Gene expression profiling was used to assign molecular classifications. These include HY (hyperdiploidy), LB (gene expression patterns frequently seen in patients with fewer focal bone lesions), MF (spikes in MAF and MAFB expression), MS (hyperactivation of MMSET +/- FGFR3), PR (over-expression of proliferation-related genes), and CD-1 or CD-2 (different forms of aberrant CCND1 and CCND3 expression). A mixed parametric cure model was used to estimate the proportion of patients with long-term, event-free survival, or the "cure fraction." When using progression free survival (PFS) in the model, the cure fraction is the percent of patients who are likely to never experience relapse based on trends in the survival times that have been observed. When using complete remission duration (CRD) in the model, the model estimates the cure fraction among patients who achieved complete response.
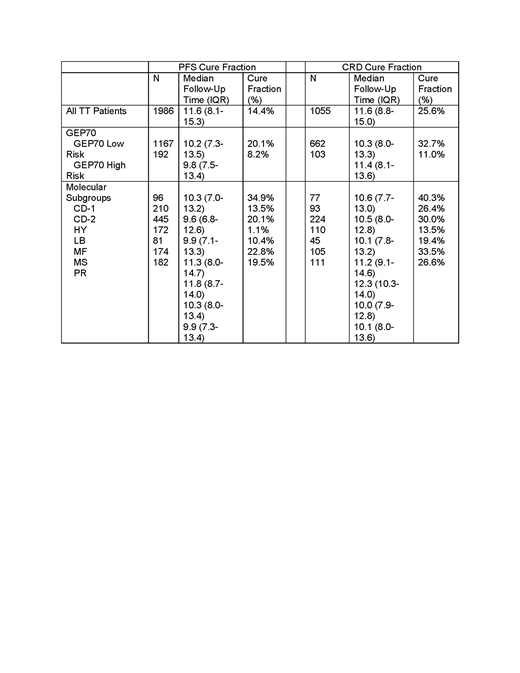
Results. The median follow-up on the entire cohort patients was 11.6 years (range: 0.0-27.6) The median overall survival was 9.2 years, with 79.3% and 48.0% having an event-free survival greater than 3 and 10 years, respectively. Overall, patients with GEP70 low risk MM had estimated PFS and CRD cure fractions of 20.1% and 32.7%, respectively. GEP70 high risk MM patients fared much worse with estimated cure fractions of only 8.2 and 11.0%. The estimated PFS- and-CRD based cure fractions increased over time with successive protocols (PFS-cure: 6.0% in TT1 to 27.7% in TT4; CRD-cure: 9.3 to 49.8%). These cure fractions were consistent with the early plateau in the PFS and CRD curves seen at 9 years in TT4 patients. The highest cure fractions were seen in the CD-1 molecular group (34.9 and 40.3%) with intermediate outcomes in the HY (20.1 and 30.0%) and MS (22.8 and 33.5%) groups (Table 1). Surprisingly, low cure fractions were observed in the LB (1.1 and 13.5%) and CD-2 groups (13.5 and 26.4%). CD-1, LB and CD-2 groups had similar 5-yr PFS rates of 60, 60 and 63% respectively, but a steady low rate of relapse was observed in the CD-2 and especially the LB group. These findings were confirmed in a 5-yr landmark analysis showing high PFS and CRD cure fractions in the CD-1 group of 62.7 and 72.3% respectively contrasting to much lower cure fractions in the CD-2 (47.2 and 49.2%) and LB (30.8 and 45.0%) groups.
Conclusions. We report excellent long-term outcomes in patients with GEP70 low risk MM and cure fractions in the range of 20-30%. Patients with LB and CD-2 subgroups have lower overall cure rates, despites similar initial 5-yr PFS rates compared to the superior performing CD-1 group, which can be explained by the occurrence of late relapses.
van Rhee:EUSA: Consultancy; Adicet Bio: Consultancy; Takeda: Consultancy; Sanofi Genzyme: Consultancy; Kite Pharma: Consultancy; Karyopharm Therapeutics: Consultancy; Castleman Disease Collaborative Network: Consultancy. Walker:Celgene: Research Funding. Davies:Janssen, Celgene: Other: Research Grant, Research Funding; Amgen, Celgene, Janssen, Oncopeptides, Roche, Takeda: Membership on an entity's Board of Directors or advisory committees, Other: Consultant/Advisor. Morgan:Amgen, Roche, Abbvie, Takeda, Celgene, Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Other: research grant, Research Funding.
anti-CD38 monoclonal antibody targeting myeloma
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal