Background:
Chromosomal rearrangements involving the 11q23 locus, resulting in fusions of the Mixed Lineage Leukemia (MLL) gene, are found in 5-10% of adult patients with acute myeloid leukemia and can represent a poor prognostic feature. Patients with this subtype of AML often respond well to standard induction chemotherapy but frequently relapse, even after allogeneic hematopoietic stem cell transplantation. Effective therapy options in the relapsed / refractory setting for this high-risk group represent an urgent unmet clinical need.
In MLL-rearranged AML, recruitment of disruptor of telomeric silencing 1-like (DOT1L), a histone H3 lysine 79 (H3K79) methyltransferase, and subsequent changes in methylation of downstream targets HOXA9 and Meis1 is central to leukemogenesis. DOT1L-mediated expression of MLL target genes is critical to developing leukemia in a murine model and inhibition of DOT1L suppressed downstream expression of MLL target genes. Pinometostat is a potent and selective small molecule inhibitor of DOT1L methyltransferase activity. Use of pinometostat in a continuous IV infusion in a rat xenograft model of MLL-rearranged leukemia showed complete, sustained tumor regressions without significant appreciable toxicities.
A recent single-agent, phase I trial evaluated pinometostat in R/R patients with MLL-R myeloid malignancies. This study included 43 patients with MLL-R AML and observed that the drug was generally well-tolerated. Responses to pinometostat included one patient with morphologic CR, one with cytogenetic CR, three with resolution of leukemia cutis, and nine with signs of differentiation / leukocytosis.
Promoter hypermethylation contributes to the dysregulation of MLL-R target genes HOXA9 and Meis1, and this effect was reversed upon treatment with the hypomethylating agent azacitidine. As such, this combination may lead to a more robust response in this patient population. We now seek to combine the novel, targeted agent pinometostat with azacitidine in R/R MLL-R AML.
Study design & methods:
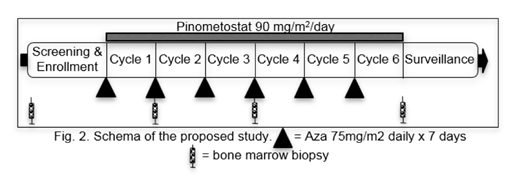
We are conducting an open-label, single-arm, phase Ib / II study that will enroll 36-48 patients with R/R MLL-R AML to evaluate the tolerability and preliminary efficacy of pinometostat in combination with azacitidine. Since MLL-R AML is a rare disease and is seen in only 5-10% of patients with AML, we are looking to collaborate with other centers through the National Cancer Institute Experimental Therapeutics Clinical Trials Network (ETCTN) to meet our accrual target. The study is active nationally and enrolling patients (NCT03701295).
The presence of the MLL-R will be confirmed locally by FISH or cytogenetics. Adult patients who are refractory to two courses of induction, relapse after CR, or elect not to pursue induction therapy are considered eligible. Patients who have previously undergone HSCT or who have well-controlled HIV are also candidates.
Pinometostat will be given by continuous IV infusion via portable pump. Azacitidine will be administered at 75mg/m2 daily over 7 days at the start of each 28-day cycle. Patients will be followed on trial for a total of 6 cycles, with bone marrow biopsies done for assessment after cycles 1, 3, and 6.
The dose of pinometostat will be escalated following a standard 3+3 design, and the primary endpoint for the phase Ib portion will be safety and tolerability. The primary endpoint for the phase II will be response to combination therapy as defined by the 2017 European Leukemia Network guidelines. Accrual will proceed as per a Simon two-stage minimax design.
Integrative correlative analyses will include genomics, changes in DOT1L-mediated methylation by H3K79 ELISA, and qPCR of HOXA9 and Meis1. PK studies and azacitidine incorporation and DNA methylation studies will also be performed.
Cai:Imago Biosciences, Inc.: Consultancy. Armstrong:AstraZeneca: Research Funding; Epizyme, Inc.: Consultancy, Equity Ownership; Imago Biosciences, Inc.: Consultancy, Equity Ownership; Cyteir Therapeutics: Consultancy, Equity Ownership; C4 Therapeutics: Consultancy, Equity Ownership; Syros Pharmaceuticals: Consultancy, Equity Ownership; OxStem Oncology: Consultancy, Equity Ownership; Accent Therapeutics: Consultancy, Equity Ownership; Mana Therapeutics: Consultancy, Equity Ownership; Novartis: Research Funding; Janssen: Research Funding. Rudek:RenovoRX: Research Funding; Taiho: Research Funding; Celgene: Research Funding; Cullinan Apollo: Research Funding. Tallman:Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; UpToDate: Patents & Royalties; UpToDate: Patents & Royalties; ADC Therapeutics: Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; UpToDate: Patents & Royalties; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; Cellerant: Research Funding; Cellerant: Research Funding; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Research Funding; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; UpToDate: Patents & Royalties; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Cellerant: Research Funding; ADC Therapeutics: Research Funding; ADC Therapeutics: Research Funding; ADC Therapeutics: Research Funding; ADC Therapeutics: Research Funding; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Biosight: Research Funding; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Biosight: Research Funding; Biosight: Research Funding; Biosight: Research Funding; Biosight: Research Funding; Cellerant: Research Funding; Cellerant: Research Funding; Nohla: Consultancy, Membership on an entity's Board of Directors or advisory committees; Biosight: Research Funding; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; BioLineRx: Consultancy, Membership on an entity's Board of Directors or advisory committees; Daiichi-Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Orsenix: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; UpToDate: Patents & Royalties; Jazz Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; KAHR: Consultancy, Membership on an entity's Board of Directors or advisory committees; Tetraphase: Consultancy, Membership on an entity's Board of Directors or advisory committees; UpToDate: Patents & Royalties; Oncolyze: Consultancy, Membership on an entity's Board of Directors or advisory committees; Delta Fly Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees. Stein:PTC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Agios: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astellas Pharma US, Inc: Membership on an entity's Board of Directors or advisory committees; Celgene Corporation: Membership on an entity's Board of Directors or advisory committees; Daiichi Sankyo, Inc.: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Syros: Membership on an entity's Board of Directors or advisory committees; Bioline: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal