
Introduction: In November 2015, conditional approval of blinatumomab was granted for adults with relapsed and/or refractory Philadelphia chromosome-negative B-cell precursor acute lymphoblastic leukemia (R/R Ph- BCP-ALL). Prior to country-specific reimbursement, blinatumomab was made available to patients (pts) who met pre-specified criteria via an expanded access program in specific countries: this included both adults and pediatric pts with diagnosis of R/R Ph- BCP-ALL, R/R Ph+ BCP-ALL, or minimal residual disease (MRD)-positive Ph-/Ph+ ALL. Here, we report on adults with MRD+ BCP-ALL enrolled in this expanded access program with reference to their characteristics and both blinatumomab usage and effectiveness.
Methods: The retrospective observational study (NEUF) recruited pts who initiated blinatumomab in the available expanded access setting between 2014 and 2016. Pts were followed from blinatumomab initiation until death, entry into a clinical trial, end of follow-up, or the end of the study period (30 June 2017), whichever occurred first. Efficacy analyses were undertaken on a MRD intention-to-treat basis. MRD response was defined as MRD level <10-4 within first cycle and within the first 2 cycles. MRD assessment was undertaken as per local clinical practice, including flow cytometry and polymerase chain reaction (PCR): MRD status was then extracted from the patient medical record. Median disease-free survival (DFS) was defined as time from initiation of blinatumomab until date of relapse (blasts in bone marrow >5% or extramedullary relapse after documented response) or death, whichever occurred first. Adverse events were reported separately, according to local regulations.
Results: Out of 373 enrolled pts, 109 MRD positive adult ALLs (83 Ph-; 26 Ph+) were included in Italy (53), France (23), Spain (20), Russia (11), and the UK (2). Most patients (76%, n=83) were Ph-. Forty-one percent (n=45) were female and median age was 43 years (interquartile range [IQR]: 27, 55).
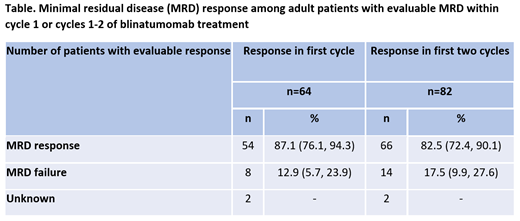
In their medical history, 16% (n=17 out of 109 MRD positive patients) had a prior allogeneic hematopoietic stem cell transplant (HSCT). The median number of prior salvage therapies was 0 (IQR: 0.0, 1.0). Among blinatumomab co-medications, almost 90% were treated with dexamethasone, 35% (n=36) as pre-phase and 87% (n=92) as pre-medication. Of the 82 pts with evaluable MRD within two cycles of blinatumomab, 83% (n=66) had a MRD response (Table), including 48 with non-detectable MRD and 18 with MRD <10-4.
Following blinatumomab initiation, 68% (n=74 out of 109) of pts proceeded to HSCT: 65 (88%) of these patients had documented complete remission with full/partial/incomplete recovery of peripheral blood counts before transplant. The median time from complete response (CR) to HSCT was 2.4 months (range: 1.6, 5.3), with median follow-up time being 18.5 months (IQR: 14.0, 27.7).
The median DFS was 27.6 months (IQR: 7.4, not estimable [NE]). Censoring for HSCT increased DFS to 33.0 months (IQR: 8.9, NE). At 24 months following blinatumomab initiation, overall survival ((OS) was 65% (95% confidence interval [CI]: 52.8, 74.2): when censoring for HSCT, OS was 77.6% (95% CI: 52.8, 88.9); median follow-up time was 4.0 months (IQR: 2.6, 13.7). The Kaplan-Meier estimate of the non-relapse mortality following HSCT post-blinatumomab was 6% (95% CI: 1.9, 16.4) at 3 months and 10% (95% CI: 4.6, 22.3) at 12 months.
Conclusions: In this large multi-country, multi-site study, blinatumomab was shown to induce molecular response within two cycles in the majority of patients with evaluable MRD. Furthermore, the median DFS was over two years, while two-thirds of pts were still alive 24 months after initiation. This study demonstrates the real-world effectiveness of blinatumomab and is consistent with results from clinical studies (BLAST).
Boissel:NOVARTIS: Consultancy. Bassan:Shire: Honoraria; Incyte: Honoraria; Amgen Inc.: Honoraria; Pfizer: Honoraria. Chiaretti:Pfizer: Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees; Shire: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees. Foà:Celltrion: Membership on an entity's Board of Directors or advisory committees; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Amgen Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Amgen Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Shire: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Roche: Consultancy, Speakers Bureau; Shire: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Consultancy, Speakers Bureau; Roche: Consultancy, Speakers Bureau; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celltrion: Membership on an entity's Board of Directors or advisory committees. Papayannidis:Novartis: Honoraria; Incyte: Honoraria; Teva: Honoraria; Shire: Honoraria; Amgen: Honoraria; Pfizer: Honoraria. Alam:Amgen: Employment, Equity Ownership. Brescianini:Amgen: Employment, Equity Ownership. Pezzani:Amgen: Employment, Equity Ownership. Kreuzbauer:Amgen: Employment, Equity Ownership. Rambaldi:Celgene: Consultancy, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Pfizer: Consultancy, Speakers Bureau; Amgen: Consultancy, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal