Introduction
N6-methyladenosine (m6A) RNA methylation is the most abundant epitranscriptomic modification, dynamically installed by the m6A methyltransferases (termed as "writers"), reverted by the demethylases (termed as "erasers"), and recognized by m6A binding proteins (termed as "readers"). Emerging evidence suggests that m6A RNA methylation regulates RNA stability, and participates in the pathogenesis of multiple diseases including cancers. Nevertheless, the role of m6A RNA methylation in chronic lymphocytic leukemia (CLL) remains to be unveiled. Herein, we hypothesized that m6A RNA methylation contributed to the tumorigenesis and maintenance of CLL. Moreover, the risk-prediction model integrated with the m6A regulators could serve as a novel and effective prognostic indicator in CLL. This study aimed to identify robust m6A RNA methylation-associated fingerprints for risk stratification in patients with CLL.
Methods
A total of 714 de novo CLL patients from 4 cohorts (China, Spain, Germany and Italy) were enrolled with informed consents. EpiQuik m6A RNA methylation colorimetric quantification assay was utilized to assess m6A RNA methylation levels. LASSO Cox regression algorithm was performed to calculate m6A RNA methylation-associated risk score (short for "m6A risk score") in R software. Besides, Kaplan-Meier survival analysis with log-rank test, univariate and multivariate Cox regression analyses and ROC curve analysis of overall survival (OS) were conduct to explore the prognostic value of m6A signature in CLL. Furthermore, RNA-seq, MeRIP-seq, Ribo-seq, functional enrichment analyses in silico and preclinical experiments ex vivo were applied to confirm the biological mechanism of the m6A regulators in CLL.
Results
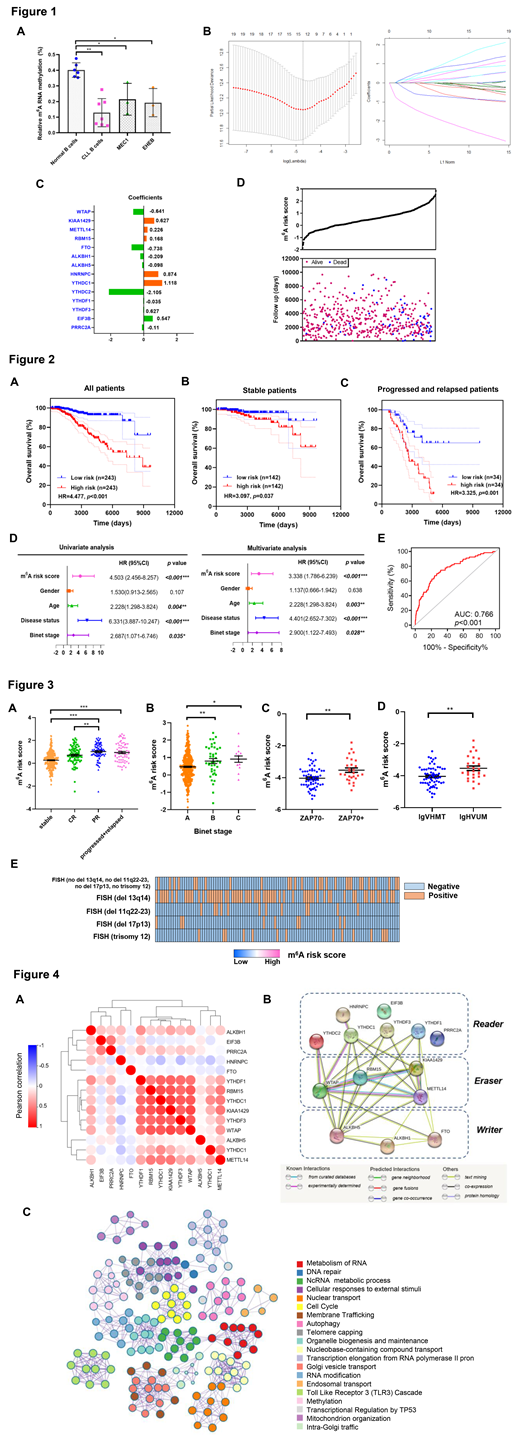
In the present study, we performed a comprehensive analysis to dissect the role of m6A RNA methylation regulators in CLL. Compared with normal B cells from healthy donors, obvious decreased level of m6A RNA methylation was observed in primary CLL cells (p<0.01; Figure 1A). In addition, down-regulated m6A RNA methylation was also detected in CLL cell lines MEC1 and EHEB (p<0.05; Figure 1A).
Then, we further investigated the association of the m6A RNA methylation regulators with clinical outcomes of CLL patients. By LASSO Cox regression analysis in 486 CLL patients, the m6A risk score was established with the coefficients of fourteen m6A regulators at the minimum lambda value of 0.00892 (Figure 1B-C). Based on the median risk score as the cut-off value, a clear distribution pattern was delineated in CLL patients (Figure 1D).
Kaplan-Meier curves showed stratified high-risk patients presented significantly shorter OS versus the low-risk group (HR=4.477, p<0.001; Figure 2A). Besides, m6A risk score also predicts inferior prognosis in stable subgroup (HR=3.097, p=0.037; Figure 2B), and progressed/ relapsed subgroup (HR=3.325, p=0.001; Figure 2C). Moreover, univariate, multivariate cox regression analyses and ROC curve confirmed high m6A risk score as an independent survival predictor in CLL patients (p<0.001; Figure 2D-E).
Thereafter, the clinicopathological relevance and underlying mechanism of m6A risk score were explored. Significant elevated m6A risk score was detected in patients with unfavorable treatment responses compared with stable status (p<0.001; Figure 3A). Furthermore, CLL patients with advanced Binet stage, positive ZAP-70 and unmutated IGHV present increased m6A risk score (p<0.05; Figure 3B-C). Intriguingly, we also observed the significantly negative correlation between highrisk score and 13q14 deletion, in accordance with patients' inferior outcome (p=0.047; Figure 3D). Moreover, Pearson correlation analysis, STRING interactive network and functional enrichment analyses deciphered that the m6A regulators exerted crucial roles in CLL progression potentially via modulating RNA metabolism and oncogenic pathways (Figure 4A-C).
Conclusion
To date, our study provides evidence for the first time that reduced m6A RNA methylation contributes to the tumorigenesis of CLL. Distinct m6A risk scoreis demonstrated as an efficient tool facilitating prognosis evaluation in CLL patients. However, validation of the signature in more independent cohorts are warranted. Further interrogations will be elucidated on the biological mechanism of m6A regulators, highlighting insights into pathogenesis and therapy strategy of CLL.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal