Background: rVIII-SingleChain is a novel B-domain truncated recombinant Factor VIII comprised of covalently bonded FVIII heavy and light chains designed to have a high binding affinity to von Willebrand factor.
Aims: This multicenter, open-label, phase III extension study investigates the safety and efficacy of rVIII-SingleChain for prophylaxis and on-demand treatment of bleeding episodes in 50 previously untreated patients (PUPs) for at least 50 Exposure Days (EDs). An ITI substudy was implemented to allow the use of rVIII-SingleChain to attempt inhibitor eradication for PUPs who develop an inhibitor to FVIII.
Method: PUPs with severe hemophilia A (no prior exposure to any FVIII product, and endogenous Factor VIII <1%) were assigned by the investigator to a prophylaxis or on-demand treatment regimen. Inhibitors were assessed monthly. Patients diagnosed with an inhibitor to FVIII (two consecutive central laboratory [CL] results of ≥0.6 BU/mL) could be enrolled into the ITI substudy or remain in the main study. The ITI substudy regimens are: 50 IU/kg 3x/week (low), 100 IU/kg daily (medium), or 200 IU/kg daily (high). Inhibitor eradication was defined as two consecutive CL results of <0.6 BU/mL. One subject was withdrawn per protocol due to high titer inhibitor diagnosis prior to ITI substudy implementation.
Results: As of March 28, 2019, 23 PUPs were treated with rVIII-SingleChain. Median age: 1 y (range 0-5). Mean (SD) time on study: 21.6 (12.6) months. Race distribution; Asian 2, Black 7, White 14.
There have been 147 bleeding events treated with rVIII-SingleChain rated for hemostatic efficacy by the investigator. While patients were inhibitor negative, the overall treatment success (rating of excellent/good) was 93%, and the annualized spontaneous bleeding rate (AsBR) was 0.58. The adverse event profile was as expected, as the most frequently occurring adverse events were upper respiratory tract infections and inhibitors.
Twelve subjects (52%) [95% CI 31%, 73%] were diagnosed with an inhibitor to FVIII; 6 (26%) high titer (peak titer ≥5 BU/mL), and 6 (26%) low titer (peak titer <5 BU/mL). Seven of 11 inhibitor negative subjects achieved >50 EDs, 1 achieved 47 EDs, and 3 achieved <20 EDs. The median ED for inhibitor development (initial result) was 10, range 4-23.
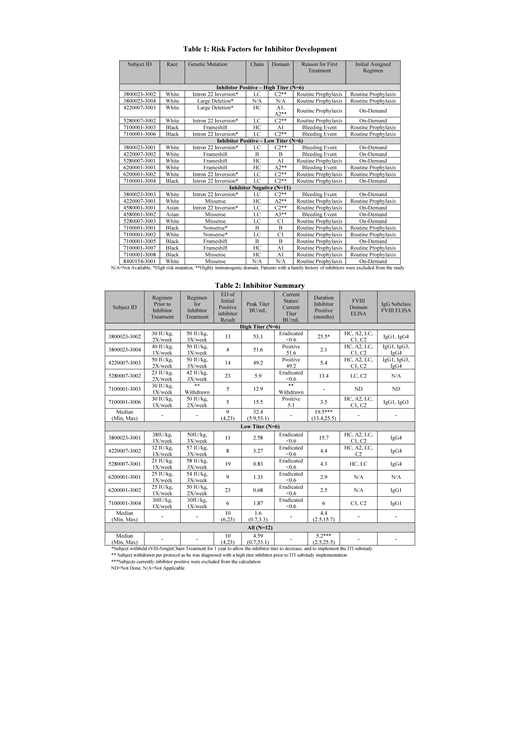
All PUPs enrolled had ≥1 risk factor for inhibitor development (Table 1) including genetic mutation, age of first exposure, initial treatment reason and assigned regimen, as well as bleeding events and infections; inhibitor positive and negative subjects were comparable.
Of the 12 inhibitor positive subjects, 11 continued treatment with rVIII-SingleChain, 7 were treated with approximately 50 IU/kg 3x/week (low dose ITI), 3 with an increased prophylaxis regimen, 1 with no change in regimen, and 0 with high or medium dose ITI regimen (Table 2). Eight of 11 (73%) inhibitor positive subjects (2 high titer, 6 low titer) achieved eradication; 5 low titer subjects were eradicated within 6 months. The clinically relevant inhibitor subjects (2 high titer, and 1 persistent low titer) achieved eradication in a median of 15.7 months, 2 using low dose ITI, and 1 using increased prophylaxis. Eradicated patients were negative for a median of 13.6 months, and no inhibitor relapse was observed. Three remaining inhibitor positive patients are early in their rVIII-SingleChain inhibitor treatment (2.1, 3.5, and 5.4 months).
Detailed analysis of the antibody signature was performed, and revealed epitope isotypes and subclass distribution comparable to other FVIII molecules.
Conclusion: Overall, rVIII-SingleChain demonstrates a positive benefit:risk profile for safety and efficacy in PUPs. The crude high titer inhibitor rate is 26% which is consistent with other rFVIII products, whereas the crude low titer inhibitor rate is currently 26% which is on the higher end in comparison to other rFVIII products. Immunological analyses suggest a low affinity antibody population in subjects with low titer inhibitors. The majority of subjects (73%) who continued treatment with rVIII-SingleChain achieved eradication on a low dose ITI or prophylaxis regimen of approximately 50 IU/kg 3x/week or less. Additional time on study for the currently enrolled subjects is required to determine the final inhibitor and eradication rates in PUPs treated with rVIII-SingleChain.
Santagostino:Pfizer: Consultancy, Speakers Bureau; Bayer: Consultancy, Speakers Bureau; Shire / Takeda: Consultancy, Speakers Bureau. Koenigs:Roche: Consultancy; CSL Behring: Research Funding, Speakers Bureau; Bayer Vital GmbH: Research Funding, Speakers Bureau; Biotest AG: Research Funding, Speakers Bureau; Intersero: Research Funding; Grifols: Speakers Bureau; Pfizer: Research Funding, Speakers Bureau; Shire: Research Funding, Speakers Bureau; Sobi: Research Funding, Speakers Bureau. Djambas Khayat:Novo Nordisk: Consultancy, Speakers Bureau; Pfizer: Speakers Bureau. Lucas:CSL Behring: Employment. Salazar:CSL Behring: Employment. Brainsky:CSL Behring: Employment. Chung:CSL Behring: Employment. Goldstein:CSL Behring: Employment. Mahlangu:Roche: Consultancy, Honoraria, Research Funding, Speakers Bureau; Spark: Consultancy, Honoraria, Research Funding, Speakers Bureau; Biomarin: Research Funding; Novartis: Research Funding; Pfizer: Research Funding; Sanofi: Research Funding; Unique: Research Funding; Catalyst Biosciences: Consultancy, Honoraria, Research Funding, Speakers Bureau; Novo Nordisk: Consultancy, Honoraria, Research Funding, Speakers Bureau; Baxalta: Consultancy, Honoraria, Research Funding, Speakers Bureau; CSL Behring: Consultancy, Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal