Abstract
Introduction
Outcomes after allogeneic stem cell transplant (AHSCT) are influenced by both disease and patient related factors. We hypothesized that combining hematopoietic stem cell transplant comorbidity-age index (HCT-CI/Age) and the refined disease risk index (DRI-R) would better predict survival post-transplant and developed a hematopoietic cell Transplant-composite risk (HCT-CR) model, which we tested in a group of patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) treated at MD Anderson Cancer Center (MDACC).
Methods
The study included consecutively treated patients, 18 years of age or older, with AML and MDS who received first AHSCT at MDACC between 2005-2016. Donors were HLA-matched related (MRD), HLA-matched unrelated (MUD), 9/10 MUD (MMUD), haploidentical (HAPLO) and 9/10 MRD (MMRD).
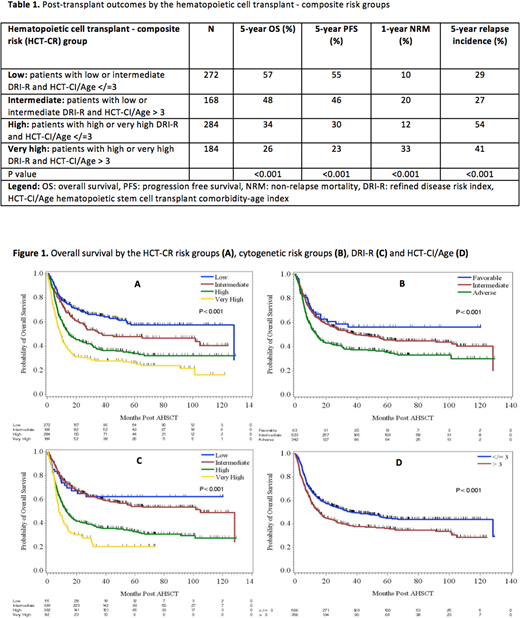
To develop this model, patients were assigned into 4 groups: 1. Patients with low/intermediate DRI-R and HCT-CI/Age </=3 (low-risk); 2. Patients with low/intermediate DRI-R and HCT-CI/Age >3 (intermediate-risk); 3. Patients with high/very high DRI-R and HCT-CI/Age </=3 (high-risk); and 4. Patients with high/very high DRI-R and HCT-CI/Age >3 (very high-risk). Primary endpoint was 5-year overall survival (OS); other outcomes assess were progression-free survival (PFS), non-relapse mortality (NRM) and relapse rate. The stability of the HCT-CR model was tested by bootstrap resampling. The discrimination power of the HCT-CR model on OS was compared with that of the DRI-R, HCT-CI/Age and cytogenetic risk model by the Harrell C-concordance index.
Results
The analysis included 942 patients (492 male and 450 female) with a median age of 53 years (range 18-65 years). Cytogenetic data at diagnosis was available in 928 (98.5%) patients and was favorable, intermediate and adverse cytogenetic risk in 63 (7%), 523 (56%) and 342 (37%), respectively. Fifty-five (6%), 399 (43%), 392 (42%) and 82 (9%) patients had low, intermediate, high and very high DRI-R, respectively. The HCT-CI/Age was available in 922 (98%) patients with the median score of 3 (range 0-18). Donor types included MRD (n=377), MUD (n=416), MMUD (n=68), HAPLO (n=73) and MMRD (N=8).
Using the HCT-CR model, patients were stratified into 4 risk groups: low (N=272), intermediate (N=168), high (N=284) and very high-risk (N=184), with significantly different survival. The 5-year OS rates for patients in low, intermediate, high and very high-risk group were 57%, 48%, 34%, and 26%, respectively (P<0.001) (Figure 1). The probability of 5-year PFS rates were 55%, 46%, 30% and 23% for these 4 risk groups, respectively (P<0.001). Post-transplant outcomes of all 4 HCR-CR groups are summarized in Table 1.
Compared with the low HCT-CR risk group, patients with intermediate, high and very high-risk group had a significantly increased risk of death with HR of 1.42 (95%CI 1.06-1.91; P=0.02), 2.11 (95%CI 1.65-2.70; P<0.001), and 3.02 (95%CI 2.32-3.92; P<0.001), respectively. The significant association between OS and the HCT-CR groups persisted after adjusting for potential confounders with adjusted HR of 1.37 (95%CI 1.02-1.85) for intermediate, 2.08 (95%CI 1.62-2.67) for high and 2.92 (95%CI 2.23-3.82) for very high-risk group when compared with low risk group.
The stability of the hematopoietic cell transplant - composite risk model was confirmed in a bootstrap resampling procedure. Among 500 new datasets, on average, patients in intermediate, high and very high-risk group had significantly increased risk of death after transplant when compared with low risk group with HR of 1.39, 2.11 and 2.98, respectively. Results from the concordance test showed that the HCT-CR model provided better discriminative capacity for prediction of OS when compared with DRI-R, HCT-CI/Age and cytogenetic risk group models with C-index of 0.62 versus 0.60, 0.54 and 0.55, respectively. The goodness of fit test showed that the HCT-CR model fit the data significantly better than the other models (P<0.001).
Conclusion
Combining disease and patient-related factors provides better survival stratification for patients with AML/MDS receiving AHSCT. This novel HCT-CR model will be validated in patients with all diseases undergoing allogeneic hematopoietic stem cell transplantation and results will be presented at the meeting.
Oran:ASTEX: Research Funding; Celgene: Consultancy, Research Funding; AROG pharmaceuticals: Research Funding. Champlin:Otsuka: Research Funding; Sanofi: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal