Abstract
Since its approval in 2011, the Janus kinase 1/2 (JAK1/2) inhibitor ruxolitinib has evolved to become the centerpiece of therapy for myelofibrosis (MF), and its use in patients with hydroxyurea resistant or intolerant polycythemia vera (PV) is steadily increasing. Several other JAK2 inhibitors have entered clinical testing, but none have been approved and many have been discontinued. Importantly, the activity of these agents is not restricted to patients with JAK2 V617F or exon 12 mutations. Although JAK2 inhibitors provide substantial clinical benefit, their disease-modifying activity is limited, and rational combinations with other targeted agents are needed, particularly in MF, in which survival is short. Many such combinations are being explored, as are other novel agents, some of which could successfully be combined with JAK2 inhibitors in the future. In addition, new JAK2 inhibitors with the potential for less myelosuppression continue to be investigated. Given the proven safety and efficacy of ruxolitinib, it is likely that ruxolitinib-based combinations will be a major way forward in drug development for MF. If approved, less myelosuppressive JAK2 inhibitors such as pacritinib or NS-018 could prove to be very useful additions to the therapeutic armamentarium in MF. In PV, inhibitors of histone deacetylases and human double minute 2 have activity, but their role, if any, in the future treatment algorithm is uncertain, given the availability of ruxolitinib and renewed interest in interferons. Ruxolitinib is in late-phase clinical trials in essential thrombocythemia, in which it could fill an important void for patients with troublesome symptoms.
Introduction
The discovery of the activating V617F mutation in Janus kinase 2 (JAK2) in the majority of patients with the classic Philadelphia chromosome–negative myeloproliferative neoplasms (MPNs)1-4 provided a strong impetus for the development of pharmacologic inhibitors of JAK2, culminating in the regulatory approval of the JAK1/2 inhibitor ruxolitinib for patients with myelofibrosis (MF) in 2011 and hydroxyurea (HU)-resistant or intolerant polycythemia vera (PV) in 2014. The activity of ruxolitinib in patients with MF without regard to JAK2 mutation status5 was an important observation that was vindicated by the finding of universal activation of the JAK signal transducer and activator of transcription (STAT) pathway in MPNs.6,7 Indeed, activating mutations in MPL8,9 (the thrombopoietin receptor) and exon 9 indels in calreticulin10,11 (an endoplasmic reticulin chaperone) underlie the vast majority of JAK2V617F– cases of essential thrombocythemia (ET) and primary myelofibrosis (PMF). Like other cytokine and growth factor receptors,12 MPL is coupled to JAK2, and a requirement for mutant calreticulin to bind to MPL in order to drive myeloproliferation has recently been shown.13-17
Several other JAK2 inhibitors have entered the clinic for patients with MPNs, but many have been discontinued, mainly because of toxicity. A prominent example is fedratinib, which was associated with 4 cases of Wernicke’s encephalopathy in a positive (for spleen and symptoms) placebo-controlled randomized trial (Table 1),18 and uncertainty persists regarding the future of those drugs still in development.19,20 A major advance in therapy for MF would be a JAK2 inhibitor that is less myelosuppressive than ruxolitinib or, better still, one that could improve cytopenias, particularly anemia,29 while preserving, if not improving upon the efficacy of ruxolitinib on splenomegaly and symptoms associated with MPN. Ruxolitinib is also currently being developed for use in HU-resistant or intolerant patients with ET.30,31 Despite the remarkable benefits of ruxolitinib in terms of reduction of splenomegaly and MPN-associated symptoms, there are still questions regarding whether this agent is truly disease modifying in MF.32,33 Another area of debate is whether ruxolitinib should be used earlier in the disease course of patients with MF. An attempt to address this question was made in a placebo-controlled randomized clinical trial in Europe, but that trial was recently terminated because of poor accrual (discussed further below).34 Finally, efforts continue to rationally combine ruxolitinib with other targeted agents in MF on the basis of laboratory evidence of synergism, and with agents that may ameliorate aspects of the disease not optimally addressed or even worsened by ruxolitinib, such as bone marrow (BM) fibrosis and anemia.
Phase 3 trials of JAK2 inhibitors in JAK2 inhibitor–naïve patients with MPNs
| Drug . | Trial . | Reference . | Patient population studied . | No. of patients . | Comparator . | Efficacy . | Safety . |
|---|---|---|---|---|---|---|---|
| Ruxolitinib | COMFORT-1 | 21 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥100 × 109/L, and <10% circulating blasts | 309 | Placebo (1:1) | ≥35% SVR (41.9% vs 0.7%); ≥50% TSS reduction (45.9% vs 5.3%), both at 24 weeks; overall survival advantage for ruxolitinib | Anemia, thrombocytopenia, ecchymoses, dizziness, headache more frequent with ruxolitinib |
| Ruxolitinib | COMFORT-2 | 22 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥100 × 109/L, and <10% circulating blasts | 219 | BAT (2:1) | ≥35% SVR (28% at 48 weeks and 32% at 24 weeks) in ruxolitinib arm; 0% and 0% in best available therapy arm; ↑ EORTC QLQ-C30 and FACT-Lymphoma scores with ruxolitinib, ↓ with B; overall survival benefit for ruxolitinib emerged at 3 years23 | Anemia, thrombocytopenia, diarrhea, abdominal pain, weight gain more frequent with ruxolitinib |
| Pacritinib | PERSIST-1 | 24 | Symptomatic* adults with intermediate-/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, absolute neutrophil count >0.5 × 109/L, and <10% circulating blasts | 327 | BAT excluding JAK2 inhibitors (2:1) | ≥35% SVR (19% vs 5% at 24 weeks); ≥50% TSS reduction (no difference at 24 weeks; did emerge at 48 weeks in intention-to-treat population but 36% vs 14% in evaluable population); 25% vs 0% achieved transfusion independence | Diarrhea, nausea, vomiting more frequent in pacritinib group |
| Momelotinib | SIMPLIFY-1 | 25 | Adults with intermediate-2/high-risk† PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥50 × 109/L, absolute neutrophil count ≥0.75 × 109/L, <10% circulating blasts, and no grade ≥2 peripheral neuropathy | 432 | Ruxolitinib (1:1) | ≥35% SVR (26.5% [momelotinib] vs 29% [ruxolitinib]); ≥50% TSS reduction (28.4% [momelotinib] vs 42.2% [ruxolitinib]); transfusion independence rate, 66.5% (momelotinib) vs 49.3% (ruxolitinib); transfusion dependence rate, 30.2% (momelotinib) vs 40.1% (ruxolitinib) | Thrombocytopenia, diarrhea, headache, dizziness frequent in both arms; also nausea with momelotinib and anemia with ruxolitinib; peripheral neuropathy reported by 10% of momelotinib and 5% of ruxolitinib patients |
| Fedratinib | JAKARTA | 18 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM and platelets ≥50 × 109/L | 289 | Placebo (1:1:1); 2 fedratinib doses were compared (400 and 500 mg) | ≥35% SVR (36%, 40%, and 1%); ≥50% TSS reduction (36%, 34%, and 7%, all at 24 weeks) | Anemia, thrombocytopenia, leukopenia, neutropenia, diarrhea, nausea, vomiting, elevated AST/ALT more frequent with fedratinib than placebo |
| Ruxolitinib | RESPONSE | 26 | Adults with PV requiring phlebotomy, spleen volume ≥450 cm3 and resistance or intolerance to HU27 | 222 | Standard therapy (1:1) | Composite 1º end point (hematocrit control + ≥35% SVR), 20.9% vs 0.9%; hematocrit control, 60% vs 19.6%; ≥35% SVR, 38.2% vs 0.9%; complete hematologic response, 23.6% vs 8.9%; ≥50% TSS reduction, 49% vs. 5%; all at 32 weeks | Higher rates of herpes zoster and nonmelanoma skin cancers in ruxolitinib group |
| Ruxolitinib | RESPONSE-2 | 28 | Adults with PV requiring phlebotomy, no palpable splenomegaly, and resistance or intolerance to HU27 | 149 | Standard therapy (1:1) | Hematocrit control, 62% vs 19%; complete hematologic response, 23% vs 5%; ≥50% TSS reduction, 45% vs 23%, all at 28 weeks | Headache, constipation, hypertension, weight gain, and anemia common in ruxolitinib group |
| Drug . | Trial . | Reference . | Patient population studied . | No. of patients . | Comparator . | Efficacy . | Safety . |
|---|---|---|---|---|---|---|---|
| Ruxolitinib | COMFORT-1 | 21 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥100 × 109/L, and <10% circulating blasts | 309 | Placebo (1:1) | ≥35% SVR (41.9% vs 0.7%); ≥50% TSS reduction (45.9% vs 5.3%), both at 24 weeks; overall survival advantage for ruxolitinib | Anemia, thrombocytopenia, ecchymoses, dizziness, headache more frequent with ruxolitinib |
| Ruxolitinib | COMFORT-2 | 22 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥100 × 109/L, and <10% circulating blasts | 219 | BAT (2:1) | ≥35% SVR (28% at 48 weeks and 32% at 24 weeks) in ruxolitinib arm; 0% and 0% in best available therapy arm; ↑ EORTC QLQ-C30 and FACT-Lymphoma scores with ruxolitinib, ↓ with B; overall survival benefit for ruxolitinib emerged at 3 years23 | Anemia, thrombocytopenia, diarrhea, abdominal pain, weight gain more frequent with ruxolitinib |
| Pacritinib | PERSIST-1 | 24 | Symptomatic* adults with intermediate-/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, absolute neutrophil count >0.5 × 109/L, and <10% circulating blasts | 327 | BAT excluding JAK2 inhibitors (2:1) | ≥35% SVR (19% vs 5% at 24 weeks); ≥50% TSS reduction (no difference at 24 weeks; did emerge at 48 weeks in intention-to-treat population but 36% vs 14% in evaluable population); 25% vs 0% achieved transfusion independence | Diarrhea, nausea, vomiting more frequent in pacritinib group |
| Momelotinib | SIMPLIFY-1 | 25 | Adults with intermediate-2/high-risk† PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM, platelets ≥50 × 109/L, absolute neutrophil count ≥0.75 × 109/L, <10% circulating blasts, and no grade ≥2 peripheral neuropathy | 432 | Ruxolitinib (1:1) | ≥35% SVR (26.5% [momelotinib] vs 29% [ruxolitinib]); ≥50% TSS reduction (28.4% [momelotinib] vs 42.2% [ruxolitinib]); transfusion independence rate, 66.5% (momelotinib) vs 49.3% (ruxolitinib); transfusion dependence rate, 30.2% (momelotinib) vs 40.1% (ruxolitinib) | Thrombocytopenia, diarrhea, headache, dizziness frequent in both arms; also nausea with momelotinib and anemia with ruxolitinib; peripheral neuropathy reported by 10% of momelotinib and 5% of ruxolitinib patients |
| Fedratinib | JAKARTA | 18 | Adults with intermediate-2/high-risk PMF or post-PV/ET MF with palpable splenomegaly ≥5 cm below LCM and platelets ≥50 × 109/L | 289 | Placebo (1:1:1); 2 fedratinib doses were compared (400 and 500 mg) | ≥35% SVR (36%, 40%, and 1%); ≥50% TSS reduction (36%, 34%, and 7%, all at 24 weeks) | Anemia, thrombocytopenia, leukopenia, neutropenia, diarrhea, nausea, vomiting, elevated AST/ALT more frequent with fedratinib than placebo |
| Ruxolitinib | RESPONSE | 26 | Adults with PV requiring phlebotomy, spleen volume ≥450 cm3 and resistance or intolerance to HU27 | 222 | Standard therapy (1:1) | Composite 1º end point (hematocrit control + ≥35% SVR), 20.9% vs 0.9%; hematocrit control, 60% vs 19.6%; ≥35% SVR, 38.2% vs 0.9%; complete hematologic response, 23.6% vs 8.9%; ≥50% TSS reduction, 49% vs. 5%; all at 32 weeks | Higher rates of herpes zoster and nonmelanoma skin cancers in ruxolitinib group |
| Ruxolitinib | RESPONSE-2 | 28 | Adults with PV requiring phlebotomy, no palpable splenomegaly, and resistance or intolerance to HU27 | 149 | Standard therapy (1:1) | Hematocrit control, 62% vs 19%; complete hematologic response, 23% vs 5%; ≥50% TSS reduction, 45% vs 23%, all at 28 weeks | Headache, constipation, hypertension, weight gain, and anemia common in ruxolitinib group |
ALT, alanine aminotransferase; AST, aspartate aminotransferase; EORTC QLQ-C30, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire C30; FACT-Lymphoma, Functional Assessment of Cancer Therapy-Lymphoma; LCM, left costal margin.
Defined as a score ≥3 for at least 2 symptoms or a score >4 for at least 1 symptom other than fatigue on the original MPN-Symptom Assessment Form (MPN-SAF) TSS, or a TSS ≥13 on the MPN-SAF TSS 2.0.
Patients with intermediate-1 risk were eligible if they had symptomatic hepatomegaly, splenomegaly, or anemia and/or were unresponsive to available therapy.
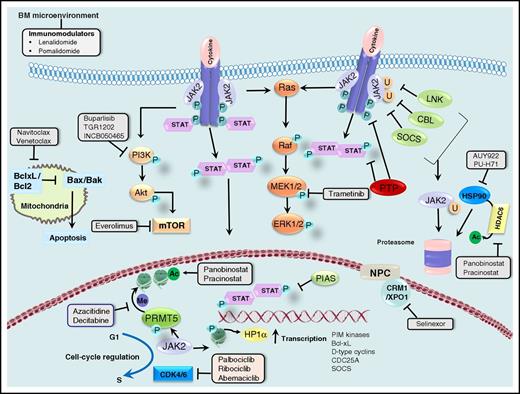
Canonical and noncanonical actions of JAK2 and opportunities for therapeutic targeting. JAK2 transduces cytokine and growth factor signals from membrane-bound receptors through phosphorylation of the STAT family of transcription factors. Negative regulators of JAK2, such as LNK, CBL, and SOCS, lead to ubiquitinylation and proteasomal degradation of JAK2, whereas protein tyrosine phosphatases (PTPs) dephosphorylate cytokine receptors, JAKs, and STATs. The protein inhibitor of STATs (PIAS) prevents the binding of STATs to target DNA. JAK2 is a client of the chaperone protein HSP90, and HSP90 inhibitors and HDAC6 inhibitors (through acetylation and disruption of HSP90 function) promote degradation of JAK2. JAK2 signals downstream to the PI3K/Akt/mTOR and Ras/Raf/MEK/ERK signaling cascades, which provides opportunities for combined inhibition of JAK2 and PI3K, mTOR, or MEK1/2. BH3 mimetics promote mitochondrial apoptosis, and synergism with ruxolitinib in MPN cells and animal models has been shown. Synergism between ruxolitinib and the selective inhibitor of nuclear export selinexor has also been demonstrated preclinically. Activated JAK2 promotes cell cycle progression through several mechanisms discussed in the text, making combined inhibition of JAK2 and CDK4/6 a rational approach. Finally, nuclear JAK2 phosphorylates PRMT5 and histone H3, activating transcription of many genes, including those encoding the PIM kinases, Bcl-xL, D-type cyclins, the cell cycle phosphatase CDC25A, and SOCS (negative feedback). Epigenetic deregulation is frequent in MPNs, and combinations of ruxolitinib with epigenetic modifiers such as azacitidine and panobinostat have shown promise in the clinic. Combinations of ruxolitinib with immunomodulatory drugs are also being pursued. Adapted, with permission, from Meyer and Levine.160
Canonical and noncanonical actions of JAK2 and opportunities for therapeutic targeting. JAK2 transduces cytokine and growth factor signals from membrane-bound receptors through phosphorylation of the STAT family of transcription factors. Negative regulators of JAK2, such as LNK, CBL, and SOCS, lead to ubiquitinylation and proteasomal degradation of JAK2, whereas protein tyrosine phosphatases (PTPs) dephosphorylate cytokine receptors, JAKs, and STATs. The protein inhibitor of STATs (PIAS) prevents the binding of STATs to target DNA. JAK2 is a client of the chaperone protein HSP90, and HSP90 inhibitors and HDAC6 inhibitors (through acetylation and disruption of HSP90 function) promote degradation of JAK2. JAK2 signals downstream to the PI3K/Akt/mTOR and Ras/Raf/MEK/ERK signaling cascades, which provides opportunities for combined inhibition of JAK2 and PI3K, mTOR, or MEK1/2. BH3 mimetics promote mitochondrial apoptosis, and synergism with ruxolitinib in MPN cells and animal models has been shown. Synergism between ruxolitinib and the selective inhibitor of nuclear export selinexor has also been demonstrated preclinically. Activated JAK2 promotes cell cycle progression through several mechanisms discussed in the text, making combined inhibition of JAK2 and CDK4/6 a rational approach. Finally, nuclear JAK2 phosphorylates PRMT5 and histone H3, activating transcription of many genes, including those encoding the PIM kinases, Bcl-xL, D-type cyclins, the cell cycle phosphatase CDC25A, and SOCS (negative feedback). Epigenetic deregulation is frequent in MPNs, and combinations of ruxolitinib with epigenetic modifiers such as azacitidine and panobinostat have shown promise in the clinic. Combinations of ruxolitinib with immunomodulatory drugs are also being pursued. Adapted, with permission, from Meyer and Levine.160
Ruxolitinib in MF: 5 years later
Ruxolitinib was approved for the treatment of patients with intermediate- or high-risk MF in the United States and for patients with MF and splenomegaly and/or symptoms in Europe on the basis of the results of the pivotal COMFORT-1 and COMFORT-2 trials (Table 1), which compared it to placebo and best available therapy (BAT), respectively.21,22 In both trials, ruxolitinib provided robust benefits as measured by the rates of ≥35% spleen volume reduction (SVR) at 24 and 48 weeks. In addition, nearly half the patients assigned to receive ruxolitinib in COMFORT-1 achieved ≥50% reduction in their total symptom score (TSS)35 on the MF Symptom Assessment Form at 24 weeks.21 Similar marked symptom benefits from ruxolitinib were observed in COMFORT-2.22 Multiple analyses of these trials with progressively longer periods of follow-up23,36-39 demonstrated that the benefits of ruxolitinib were durable; median spleen response duration was 3.2 years in both COMFORT-138 and COMFORT-2.39 In COMFORT-1, 59.4% of patients originally randomly assigned to ruxolitinib achieved ≥35% SVR at any time during the study, although the proportion of patients with ≥35% SVR progressively declined from 42% at 24 weeks to 18.5% at week 264; the median change in spleen volume from baseline at week 240 was –40.8%.38 In COMFORT-2, 53.4% of patients in the ruxolitinib arm achieved ≥35% SVR at any time on the study, and the probability of maintaining response at 5 years was 0.48.39 Adverse events (AEs) resulting in discontinuation of ruxolitinib occurred in approximately one-third of patients in COMFORT-1 and one-quarter of patients in COMFORT-2.38,39 In COMFORT-1, anemia (98.7%) and thrombocytopenia (83.9%) occurred in the vast majority of patients randomly assigned to ruxolitinib, and herpes zoster infections occurred significantly more frequently than in the placebo group (similar to the experience in PV),26 whereas pneumonia was the most common late-onset grade 3 to 4 AE (after 48 months of ruxolitinib therapy).38 Pneumonia (13.1%) and herpes zoster (11.5%) were common among ruxolitinib-treated patients in COMFORT-2 as well, as were urinary tract infections (24.6%), whereas grade 3 to 4 anemia was reported in 22.5% and thrombocytopenia was reported in 15.2%.39 Although ruxolitinib was well tolerated overall, immunosuppression has been a concern, given evidence of impairment of dendritic cell,40 T-cell,41 and natural killer cell42 function. There are also anecdotal reports of hepatitis B reactivation,43 cytomegalovirus retinitis,44 and even progressive multifocal leukoencephalopathy45 in patients treated with ruxolitinib, whereas tuberculosis occurred in 1% of patients receiving ruxolitinib in COMFORT-2.39 Of interest, a comparison of the placebo and BAT groups across the COMFORT trials showed no meaningful differences.46
Although the COMFORT trials studied International Prognostic Scoring System47 intermediate-2 and high-risk patients, a substantial body of evidence supports the use of ruxolitinib in intermediate-1 risk patients with MF as well.48 Recently released National Comprehensive Cancer Network guidelines also recognize ruxolitinib as an option for low-risk MF patients with troublesome symptoms and/or splenomegaly.49 In an analysis of the phase 3b expanded access JUMP trial, which included 163 patients with intermediate-1 risk MF who had received ruxolitinib therapy for at least 1 year, more than 60% had achieved a ≥50% reduction from baseline in palpable spleen length at 24 and 48 weeks, and another ∼20% to 25% achieved a <50% reduction.50 The median time to a ≥50% reduction in palpable splenomegaly was 4.7 weeks. Approximately 20% of patients had discontinued ruxolitinib at the time of data cutoff, mostly (11%) as a result of AEs. Anemia (54%; grade 3 to 4, 24.5%) and thrombocytopenia (40.5%; grade 3 to 4, 11%) were common. Platelet counts stabilized after declining over the first 4 weeks, whereas hemoglobin levels decreased during the first 8 to 12 weeks and then gradually rose to near-baseline levels (after week 24), as in COMFORT-1 and COMFORT-2. Infections occurring in ≥5% of patients included herpes zoster (8%) and bronchitis (6.1%). Spleen responses to ruxolitinib were dose-dependent and correlated with survival,51,52 underscoring the importance of careful dose optimization while managing drug-related cytopenias.53 Importantly, pooled analyses of the COMFORT studies have shown that ruxolitinib-induced anemia, unlike disease-related anemia, does not have an impact on survival.54,55 Although many variables have been evaluated,5 only the presence of ≥3 mutations (using a panel of 28 genes recurrently mutated in hematologic malignancies) has been found thus far to predict lower odds of a spleen response to ruxolitinib, a shorter time to treatment discontinuation, and inferior survival.56
Whether or not ruxolitinib prolongs the survival of MF patients continues to be a contentious issue.32,33 Although statistically significantly superior survival among patients randomly assigned to ruxolitinib was noted after 1,21 2,36 and 538 years of follow-up of the COMFORT-1 trial, and after 323 and 539 years of follow-up of the COMFORT-2 trial, these trials were not powered to measure differences in survival.33 At 5 years, the median survival times of patients assigned to receive ruxolitinib in both trials had not been reached, whereas those assigned to receive placebo or BAT fared similarly (median, 3.8 and 4.1 years, respectively).38,39 Ruxolitinib-treated patients in the original phase 1/2 trial51 and in COMFORT-257 have also been shown to have improved survival in comparison with historical cohorts, but mismatches in the proportions of patients with post-PV/ET MF (who may have better outcomes than those with PMF)58 or thrombocytopenia and advanced age (both well-recognized adverse prognostic features in PMF)47,59,60 have been cited as inherent flaws in these studies.33 In terms of disease modification, the effects of ruxolitinib are rather modest as assessed by improvement in BM fibrosis (15.8% in COMFORT-2)39 and mutant JAK2 allele burden reduction (26 of 236 evaluable patients in COMFORT-1 achieved partial or complete molecular responses).61 For these reasons, it has been proposed62 that the extension of survival of patients with intermediate-2–risk or high-risk MF by ruxolitinib in the COMFORT trials may be attributable to improvements in appetite, weight, cachexia, and hypocholesterolemia as a result of the drug’s broad suppressive effect on inflammatory cytokines.63 An Italian consensus panel recently concluded that, on the basis of current evidence, ruxolitinib should not be used solely for its survival benefit.64 However, the survival advantage seen for ruxolitinib in the COMFORT trials, despite complete crossover and the greater mutant JAK2 allele burden reduction observed in patients with shorter disease duration,61 raise the possibility that this is truly a disease-modifying agent and should, therefore, be used in less advanced stages of the disease. The phase 3 ReTHINK trial (NCT02598297), a comparison of ruxolitinib with placebo in patients with high molecular risk (ie, mutations in 1 or more of the ASXL1, SRSF2, EZH2, IDH1, or IDH2 genes)65 MF but no significant splenomegaly or disease-associated symptoms, was designed to explore the possibility that ruxolitinib may delay or prevent progression when used earlier in the disease course,34 but that trial had to be closed recently because of poor accrual (low incidence of high-molecular-risk mutations among individuals without substantial splenomegaly or symptoms).
Pacritinib and momelotinib for MF: where do we stand?
JAK2 is essential for normal hematopoiesis,66 but myelosuppression limits dose escalation of ruxolitinib in MF.63 Thus, there was substantial enthusiasm for pacritinib, a nonmyelosuppressive JAK2 selective inhibitor, on the basis of promising phase 2 results in patients with MF and any degree of cytopenia.67,68 Pacritinib was compared with BAT (excluding ruxolitinib) in 327 JAK inhibitor–naïve patients with MF in the PERSIST-1 trial (patients with platelets <100 000/µL were excluded from the COMFORT trials),24 32% of whom had baseline platelets <100 000/µL and 15% of whom had baseline platelets <50 000/µL. At week 24 in the intention-to-treat analysis, significantly greater proportions of patients assigned to pacritinib than of those assigned to BAT achieved ≥35% SVR, the primary end point, as well as ≥50% TSS reduction (considering the symptom cluster of fatigue/tiredness, early satiety, abdominal discomfort, night sweats, pruritus, and bone pain). Significant differences in favor of pacritinib in terms of SVR were also noted in the subgroups of patients with baseline platelets <100 000/µL and <50 000/µL; 25% of red blood cell (RBC) transfusion-dependent (TD) patients receiving pacritinib achieved transfusion independence (TI) compared with none receiving BAT (Table 1).24 Two doses of pacritinib (200 mg twice per day and 400 mg once per day) were compared with BAT (which could include ruxolitinib) in thrombocytopenic (platelets <100 000/µL) patients with MF in the PERSIST-2 trial.69 Of the 72 intention-to-treat patients in the BAT arm, 32 (44%) received ruxolitinib at some point in the trial. Prior receipt of JAK2 inhibitors was also allowed, and similar proportions of patients in the pooled pacritinib groups and the BAT group (41% to 46%) had previously received ruxolitinib. The primary end point of superior SVR (by ≥35%) in the pooled pacritinib arms was met (18% vs 3%; P = .001), but the co-primary end point of superior TSS reduction (by ≥50%) was not (25% vs 14%; P = .079). In secondary analyses, pacritinib at 200 mg twice per day outperformed BAT with respect to both SVR (22% vs 3%; P = .001) and TSS reduction (32% vs 14%; P = .011), whereas pacritinib at 200 mg once per day was superior to BAT only in terms of SVR (15% vs 3%; P = .017).69 Accrual to and analysis of the PERSIST-2 trial were affected by the imposition of a full clinical hold19 by the US Food and Drug Administration on pacritinib clinical trials because of excess cardiovascular and hemorrhagic events in pacritinib-treated patients in both PERSIST trials. This hold has since been lifted.20 After discussions with the Food and Drug Administration, CTI BioPharma will conduct additional dose exploration studies of pacritinib in MF.
Momelotinib is a JAK1/2 inhibitor that (at least in a rat model of anemia of chronic disease) improves anemia through inhibition of activin A receptor type 1 and reduction of hepcidin production by the liver.70 Encouraging activity, encompassing spleen, symptom, and anemia responses, was observed in phase 1 and 2 trials of momelotinib in patients with MF, with both once-per-day and twice-per-day dosing.71,72 These findings led to the design of two phase 3 trials, SIMPLIFY-1 and SIMPLIFY-2.25,73 SIMPLIFY-1 (Table 1) was a noninferiority study comparing momelotinib head-to-head with ruxolitinib in JAK inhibitor–naïve patients with MF.25 Noninferiority was achieved with respect to the primary end point of ≥35% SVR at 24 weeks but not for the key secondary end point of TSS reduction. In SIMPLIFY-2, momelotinib was compared with BAT in patients with MF who were pretreated with ruxolitinib,73 and continuation of ruxolitinib was allowed in the BAT arm (in fact, 88% of patients randomly assigned to BAT continued to receive ruxolitinib, a much higher proportion than in PERSIST-2). This study did not meet its primary end point of superiority of momelotinib over BAT in terms of SVR at 24 weeks. Both trials showed differences in favor of momelotinib with respect to anemia-related end points, and SIMPLIFY-2 also in terms of TSS reduction. On the basis of the overall negative findings of these 2 registration-directed trials, the development of momelotinib has been discontinued.
Other JAK inhibitors in development for MF
The JAK2/Src inhibitor NS-01874 is currently being tested in a phase 1/2 trial75 in patients with MF. In vitro, this agent is 4.3-fold more selective for JAK2V617F than wild-type JAK2, which translated to reduction of leukocytosis and splenomegaly, improvement in BM fibrosis, and prolonged survival in a mouse model of MF without a decrease in circulating RBCs or platelet counts.76 In the phase 1 portion of the ongoing trial,77 a ≥50% reduction in palpable splenomegaly was achieved in more than half the patients, 48% of whom had previously been exposed to JAK inhibitors. Fourteen of 36 evaluable patients (39%) had International Working Group for Myelofibrosis Research and Treatment–defined78 clinical improvement in splenomegaly (sustained for ≥8 weeks).77 The rates of anemia and thrombocytopenia were 15% and 27% in phase 1,77 and 21% and 14% in phase 2 (preliminary results), respectively.75 Enrollment in the phase 2 portion was restricted to patients for whom prior JAK2 inhibitor therapy had failed. Results on 29 patients have been presented thus far.75 Three of 26 evaluable patients (12%) achieved ≥35% SVR and 9 (35%) experienced ≥50% reduction in TSS; all 3 spleen responses were durable (for ≥6 months). Nausea, diarrhea, fatigue, and neurologic AEs (dizziness in the majority) each occurred in 17% of patients.
The JAK1-selective inhibitor INCB039110 has been tested in patients with MF79 because selectively targeting JAK1 through suppression of cytokines may provide significant symptom benefits to patients with minimal myelosuppression.80 SVRs with this agent were expectedly modest, although they were often associated with some clinical benefit for patients.79 Approximately one-third of patients achieved the primary end point of ≥50% reduction in TSS at week 12 in the 2 higher-dose cohorts, and 53.8% of patients requiring RBC transfusions during the preceding 12 weeks achieved a ≥50% reduction in the number of units transfused during the first 24 weeks on study.79
Conventional (type 1) adenosine triphosphate–competitive JAK inhibitors bind to and stabilize the active conformation of JAKs, whereas type 2 inhibitors bind to and stabilize the inactive conformation, preventing the kinase from cycling to the active conformation.81 The JAK2 inhibitor persistence is a phenomenon in which JAK-STAT signaling is reactivated despite the presence of a type 1 JAK2 inhibitor via heterodimerization between activated JAK2 and other JAKs, such as JAK1 or TYK2.82 CHZ868 is a type 2 JAK2 inhibitor that can reverse type 1 JAK2 inhibitor persistence and is active in vivo, inducing greater reductions in the mutant allele burden than type 1 JAK2 inhibitors in murine models of MPNs.83 However, this agent is not a clinical candidate.
Rational ruxolitinib-based combinations for MF: is this the way forward?
Given the discontinuation of development of a number of JAK inhibitors, persistent uncertainty over the future of pacritinib, and the established safety and efficacy of ruxolitinib as well as its limitations, finding optimal partner drugs to combine with ruxolitinib seems to be the most logical approach to drug development in MF. Naturally, the design of such combinations needs to take into consideration the clinical unmet needs, strength of the translational science, and long-term tolerability.
Combining ruxolitinib with drugs that improve anemia in MF is intuitive, but a trial evaluating the combination of ruxolitinib and danazol was terminated prematurely because of lack of benefit,84 and excessive myelosuppression limits the concurrent administration of lenalidomide and ruxolitinib.85 Although pomalidomide was not better than placebo in terms of TI rates in a phase 3 randomized controlled trial in RBC-TD patients with MF,86 this agent is being studied in combination with ruxolitinib in anemic patients with MF.87 After a median of eight 4-week cycles of treatment, 3 of 24 patients experienced clinical improvement of their anemia, two of whom had increases in hemoglobin levels.87 A study of the combination of ruxolitinib and thalidomide in patients with MF (NCT03069326) has recently begun enrolling patients.
Sotatercept is a first-in-class, activin receptor type IIA ligand trap that may relieve stromal inhibition of terminal erythropoiesis by sequestering ligands of the transforming growth factor β superfamily such as growth and differentiation factor 11.88 In the first report of an ongoing trial (NCT01712308) in patients with MF and anemia, sotatercept led to a 36% objective response rate (ORR), which included achievement of TI in some RBC-TD patients.89 This well-tolerated agent lends itself particularly well to combination with ruxolitinib, and if proven effective in the combination arm of this trial, this class of agents could prove to be a useful addition to the therapeutic armamentarium for anemia of MF, enabling more patients to stay on and receive an optimal dose of ruxolitinib.
Abnormalities of methylation are frequent in MPNs,90 and JAK2 powerfully influences gene transcription through its noncanonical nuclear actions (eg, phosphorylation of histone H391 and the arginine methyltransferase PRMT5).92 Azacitidine93 and decitabine94 have limited single-agent activity in MF. A trial of ruxolitinib and azacitidine (n = 41; with azacitidine begun after 3 months of ruxolitinib alone) reported an ORR of 69% and a median time to response of 1 month.95 Of 29 patients with palpable splenomegaly ≥5 cm at baseline, 14 (48%) achieved a >50% reduction in palpable spleen length at 24 weeks, and 23 (79%) achieved this at any time on the study. The JAK2V617F allele burden decreased in 87% of serially evaluable JAK2-mutated responders, and the European Myelofibrosis Network BM fibrosis score96 decreased in 41% of responders.93 These results are noteworthy because PMF may be best considered a myelodysplastic syndrome/myeloproliferative neoplasm (MDS/MPN),97 and this regimen may be particularly useful, for example, in patients with higher BM blast counts.
Among many other mechanisms of action in MPNs,98 an important effect of histone deacetylase (HDAC) inhibitors is downregulation of JAK2 via interference with the chaperone function of HSP90 (through acetylation).99 In general, these agents have been more efficacious clinically in PV/ET than in MF, and their chronic, predominantly low-grade toxicities such as fatigue, nausea, diarrhea, and thrombocytopenia can be challenging for long-term use,98 which seems to be necessary before disease-modifying effects emerge in MF.100 Synergism between JAK2 inhibitors and HDAC inhibitors against MPN cells has been demonstrated preclinically.101 Three clinical trials of HDAC inhibitors in combination with ruxolitinib in MF are presently enrolling patients or have completed accrual (NCT01693601, NCT01433445, NCT02267278). Preliminary results from one of these (NCT01433445) showed ≥35% SVR in 57% and 39% of patients in the expansion phase (n = 23) at weeks 24 and 48, respectively, improved BM fibrosis in 4 patients, and a ≥20% decline in the JAK2V617F allele burden in 5 patients by week 48, results that seem to compare favorably with those expected with ruxolitinib alone.102
JAK2V617F is associated with significantly increased levels of phospho-Akt in hematopoietic cells,103,104 and multiple groups have shown synergism between JAK2 inhibitors and agents that interrupt the phosphatidylinositol-3-kinase/Akt/mammalian target of rapamycin (PI3K/Akt/mTOR) axis in MPNs, both in vitro and in vivo.105,106 Promising results were obtained with single-agent everolimus in patients with MF, particularly with regard to relief of MPN-associated symptoms.107 However, the combination of ruxolitinib and the pan-PI3K inhibitor buparlisib was relatively disappointing.108 More recent efforts in this area have focused on drugs that selectively target the δ isoform of PI3K109 ; however, the experience with idelalisib in B-cell malignancies tells a cautionary tale, with reports of induction of genomic instability in B cells110 and of high rates of immune-mediated hepatotoxicity in previously untreated patients with chronic lymphocytic leukemia.111
PRM-151 is recombinant human pentraxin-2, a naturally occurring plasma protein also known as serum amyloid P that inhibits fibrocyte differentiation.112 It has been demonstrated that clonal, neoplastic fibrocytes contribute to the induction of BM fibrosis in PMF.113 PRM-151 was well tolerated in patients with MF, both alone and in combination with ruxolitinib, and several BM fibrosis responses were seen.114 Among 13 patients who completed at least 72 weeks of treatment, the majority had improvements in BM fibrosis, hemoglobin levels, platelet counts (including achievement of TI in patients who were TD), symptoms, and splenomegaly.115 A pivotal phase 2 trial of PRM-151 monotherapy in patients with MF who are not candidates for ruxolitinib (NCT01981850) has completed accrual. Regulatory approval of this agent could potentially pave the way for more definitive combination studies with ruxolitinib.
The use of interferon preparations is recommended in professional society guidelines for the first-line treatment of high-risk young patients with PV or ET, given their lack of leukemogenicity.116 Both pegylated interferon alfa-2a (administered once per week) and ropeginterferon alfa-2b (administered once every 2 weeks) are associated with high rates of hematologic and molecular response in patients with PV and ET117-119 ; however, at least in the case of pegylated interferon alfa-2a, discontinuation rates as a result of toxicity can still be substantial.120 In MF, the role of interferon monotherapy is essentially limited to some patients with early disease.121,122 A 90% ORR to the combination of interferon alfa-2 and ruxolitinib was reported in a small trial (n = 30; 20 PV, 7 PMF, 3 post-PV MF; 27 previously treated with interferon alfa-2),123 with marked improvements in symptoms, palpable splenomegaly, and hematocrit control without phlebotomy, suggesting that such combinatorial approaches should be pursued, particularly in light of renewed interest in interferon after the demonstration of noninferiority of ropeginterferon alfa-2b to HU in patients with PV.124
There is a particularly strong rationale for combining ruxolitinib with pharmacologic inhibitors of cyclin-dependent kinases 4 and 6 (CDK4/6) which, in complex with D-type cyclins, drive cell cycle progression from G0 to and during G1. JAK2V617F translationally upregulates the CDC25A phosphatase that regulates the G1/S transition and S-phase entry.125 JAK2V617F also downregulates and inhibits the CDK inhibitory function of p27.126 Finally, the transcription of D-type cyclins is promoted by JAK-STAT.127,128 Synergism between ruxolitinib and the CDK4/6 inhibitor ribociclib was recently demonstrated in vivo in MPN models and was shown to be further enhanced by the addition of PIM kinase inhibitor PIM447.129 JAK2-mediated growth factor signaling induces the expression of PIM.130 Accordingly, a clinical trial of this triple-drug combination in MF is underway in Europe (NCT02370706).
Inhibitors of hedgehog signaling have thus far exhibited insufficient clinical activity on their own in MF,131 and on the basis of laboratory evidence of synergism,132 some have been combined with ruxolitinib.133 However, the results presented thus far do not suggest a clear incremental benefit to this approach over ruxolitinib alone.133 Other agents that seem promising in preclinical studies as potential partners for ruxolitinib include HSP90 inhibitors,134 which degrade JAK2 (this strategy may also overcome JAK2 inhibitor persistence),135 the mitogen activated protein kinase kinase (MEK) inhibitor trametinib (ruxolitinib induced extracellular signal-regulated kinase (ERK) activation in a mouse model of mutant calreticulin),136 the selective inhibitor of nuclear export selinexor,137 and the BCL-2/-xL antagonist ABT-737138 ; however, these agents/combinations are not yet in clinical trials in MPN, except for a small trial (NCT01668173) of monotherapy with the HSP90 inhibitor AUY922, in which 6 patients were treated before the trial was terminated because of toxicity.139
Ruxolitinib for PV and ET
Ruxolitinib was approved for HU-resistant or intolerant27 patients with PV on the basis of the findings of the RESPONSE trial (Table 1), in which it outperformed BAT (which was still HU in the majority of patients, reflecting the paucity of options for this population) with respect to hematocrit control, ≥35% SVR, complete hematologic response rate, and improvement in TSS at week 32, accompanied by a trend toward fewer thromboembolic events.26 The benefits of ruxolitinib were maintained after a minimum of 80 weeks of follow-up140 and recapitulated in a very similarly designed trial in HU-resistant or intolerant127 PV patients without significant splenomegaly (RESPONSE-2; Table 1).28 However, in the phase 3b RELIEF trial, which evaluated switching to ruxolitinib in patients with PV and persistent symptoms despite good hematocrit control on a stable dose of HU, this strategy was not statistically significantly superior to continuing on HU, as assessed by the rate of ≥50% reduction in the TSS cytokine symptom cluster (the sum of tiredness, itching, muscle aches, night sweats, and sweats while awake) at week 16.141
Several trials are currently evaluating ruxolitinib in high risk-ET (NCT02962388, NCT02577926). Specifically, strategies aimed at regulatory approval in both the United States and Europe are focusing on a second-line indication in patients resistant to or intolerant of HU,142 comparing the drug with anagrelide or interferon alfa (eg, NCT03123588, NCT02962388). Long-term, noncomparative phase 2 data with ruxolitinib in this setting have been presented.31 In general, ruxolitinib was well tolerated and led to improvements in thrombocytosis, leukocytosis, splenomegaly, and symptoms of pruritus, bone pain, night sweats, asthenia, and paresthesia in most patients.31 In addition, the MAJIC trial compared ruxolitinib to BAT in ET patients with HU resistance or intolerance.30 Ruxolitinib was not superior to BAT in that trial in terms of complete hematologic response at 12 months (the primary end point) or partial hematologic response, but molecular responses were restricted to ruxolitinib-treated patients, who also had significantly lower mean MPN-10 TSS35 and individual symptoms of early satiety and itching during the first 12 months.30
Ruxolitinib in other MPNs and MDS/MPN overlap syndromes
Activating mutations in the gene encoding the colony-stimulating factor 3 receptor (CSF3R) are common in patients with chronic neutrophilic leukemia (CNL) and atypical chronic myeloid leukemia (aCML).143 Membrane proximal CSF3R mutants (eg, T618I) are sensitive to ruxolitinib in vitro143 as well as in vivo,144 and there are several anecdotal reports of clinical efficacy of ruxolitinib in patients with CNL or aCML harboring this mutation, in some cases, accompanied by temporary reductions in the mutant allele burden.145-147 Accordingly, a clinical trial (NCT02092324) of ruxolitinib for patients with CNL or aCML is underway: on the basis of experience in MF, patients do not need to have an activating CSF3R mutation to be eligible. Chronic myelomonocytic leukemia (CMML), one of the MDS/MPNs, is characterized by dependence on aberrant signaling through the granulocyte-macrophage colony-stimulating factor pathway, which requires JAK-STAT.148,149 The efficacy of JAK2 inhibition in CMML has been demonstrated preclinically.150 In a phase 1 trial (n = 20), the recommended phase 2 dose of ruxolitinib in CMML was found to be 20 mg twice per day, and no dose-limiting toxicities were identified. Objective responses using MDS response criteria (mostly hematologic improvement) occurred in 5 patients, and the majority of patients had improvement or resolution in splenomegaly and disease-related symptoms.151 The combination of ruxolitinib and azacitidine seems promising in patients with unclassifiable MDS/MPNs.152
Perspectives and conclusion
More than 5 years after ruxolitinib was licensed for use in patients with MPNs, it remains the only JAK inhibitor registered for hematologic indications. During this time, much has been learned about the efficacy and safety profile of this and other JAK inhibitors in MPNs.153 Several JAK inhibitors tested in patients with MF failed because of neurotoxicity, and although the underlying mechanisms remain purely speculative, possible relationships to interference with erythropoietin154 and leptin155 signaling and, in the case of fedratinib, inhibition of thiamine uptake,156 have been suggested. Although it has been associated with a high incidence of grade 1 to 2 peripheral neuropathy, momelotinib was recently discontinued because of insufficient efficacy. Given that this agent had the unique and clinically important property of improving MF-associated anemia, this raises the question of whether rates of SVR and TSS improvement are always optimal endpoints in pivotal trials of MF drugs and if such trials should be powered for survival instead.
Although ruxolitinib has brought enormous benefits to patients, evidence that JAK inhibition is truly disease modifying, at least in MF, remains weak. This is consistent with observations that the phenotype-driving mutations may not, in fact, be the disease-initiating mutations in PMF.97 Allogeneic hematopoietic cell transplantation (allo-HCT), still the only treatment modality with curative potential, should be offered to all patients with higher-risk MF.157,158 Although the timing of allo-HCT in patients receiving ruxolitinib is not known with certainty, retrospective studies suggest that prior ruxolitinib therapy does not compromise allo-HCT outcomes and that the drug should be continued almost up until the time of conditioning.159 A plethora of other targeted agents are being studied in MPNs, mostly in MF, both alone and in combination with ruxolitinib. Thus far, some of the combinations have been more promising than others, but in general, both insufficient activity (over that expected with ruxolitinib alone) and additive or overlapping toxicity are of concern. Having a lead-in phase of ruxolitinib alone for about 12 weeks (the period within which patients derive maximum benefit from ruxolitinib monotherapy) before the second drug is introduced (eg, NCT01787487, NCT02267278) may be optimal. Nonetheless, advances in our understanding of the biology of MPNs and the diverse strategies being explored raise hope that the next chapter in MPN therapy will soon begin, building upon the gains made in the JAK inhibitor era.
Acknowledgments
The authors gratefully acknowledge Rita Assi, Kate J. Newberry, and Naval Daver for their assistance in preparing the figure.
This work was supported in part by a Cancer Center Support Grant (P30 CA016672) from the National Institutes of Health, National Cancer Institute (MD Anderson Cancer Center).
Authorship
Contribution: P.B. and S.V. reviewed the literature and wrote the review article.
Conflict-of-interest disclosure: S.V. received research funding for conducting clinical trials from Incyte, Gilead Sciences, Promedior, CTI BioPharma, and NS Pharma. P.B. received honoraria from Incyte for participation in an advisory board.
Correspondence: Srdan Verstovsek, University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd, Unit 428, Houston, TX 77030; e-mail: sverstov@mdanderson.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal