Abstract
INTRODUCTION:
Cell population data (CPD) measured by hematology analyzers (HA) describe morphological parameters of white blood cell (WBC) subpopulations and are reported to be as useful parameters in the screening of several hematological and non-hematological diseases. The Sysmex XN is an HA that uses fluorescence flow cytometry for WBC differential, combining forward scatter (FSC), side scatter (SSC) and fluorescence intensity (SFL) to identify WBC populations. These three measures, along with their Distribution Width (DW), are used to provide CPD of Neutrophils (NE), Lymphocytes (LY) and Monocytes (MO). Recent articles have shown that neutrophils CPD parameters (NE-SSC, NE-SFL) may be useful in the diagnosis of myelodysplastic syndromes [1,2] and sepsis [3]. However, little is known on their analytical quality specification, as the comparability of CPD between instruments and laboratories. The aim of the study was to evaluate the between-analyzers consistency of CPD provided by Sysmex XN comparing the results obtained in five different XN modules installed in the same laboratory. Here we present the results for NE-SSC and NE- SFL, the CPD parameters mainly used to support the diagnosis of suspected sepsis and myelodysplasia
M&M
Peripheral blood samples collected in K3EDTA tubes (Becton Dickinson, NJ) in three different days from 30 healthy blood donors (HBD, 10 samples/daily) were analyzed within 30 minutes on each XN module. Additionally, all normal samples analyzed by the lab were selected from the routine workload of one month (November 2015) consisting of a total of 35.500 samples. The inclusion criteria (that led to a selection of 6,702 normal routine samples, NRS) were as follows: no morphological flags, WBC from 4.0 to 10.0 x 109/L; Hgb from 120 to 170 g/L; MCV from 84 to 98 fL, PLT from 150 to 450 x 109/L; RDW-CV < 14.0%; NRBC=0.00 x 109/L; NE from 2.00 to 5.60 x 109/L, LY from 1.50 to 3.50 x 109/L, MO from 0.30 to 0.80 x 109/L, EOS from 0.10 to 0.60 x 109/L, BASO <0.20 x 109/L. Finally, the daily results of XN-Chex control blood (Normal level) were included in the evaluation.
For each group of samples (HBD, NRS, XN-Check) we compared the median values of CPD parameter obtained on each XN modules. The between-analyzers consistency of CPD parameter was assessed calculating the percentage of Bias (Bias%) between the median value of each XN module and the overall median value of all the five analyzers.
To evaluate the stability over the time of CPD parameters, the same study on 30 HBD, 6,690 NRS and XN-Chex control blood was repeated after two months (January 2016).
RESULTS
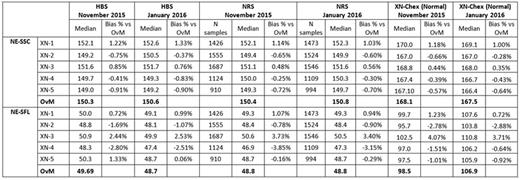
The median values of NE-SSC and NE-SFL obtained in each group of samples are shown in Table 1. The results highlight a good agreement between analyzers, with a maximum Bias of -3.85%. Interestingly, the median values of NE-SSC and NE-SFL were practically the same in HBD and NRS while, as expected, they were significantly different on XN-Chex (due to the presence of stabilized cells). However, the Bias observed for each module XN on control blood was similar to that observed on fresh blood (HBS and NRS groups). Median values of NE-SSC and NE-SFL were practically the same after two months both in HBD and NRS, proving that CPD parameters are stable over the time. Similar results were obtained on all the other CPD parameters provided by Sysmex XN (data not shown)
CONCLUSIONS
CPD parameters are stable over the time and their inclusion on the automated QC of the analyzer based on the moving average of normal may be a reliable and inexpensive method to assess the between-analyzers consistency of CPD parameters. Providing the expected values of CPD parameters, XN-Check could be useful for the daily QC, to assess the need of calibration and to improve the inter-laboratory comparability being the Neutrophils the best WBC population to control using XN-Check control blood. The moving average can be used too as a way to monitor the calibration and to decide what to do when the QC materials give a result out of range (moving average).The results of this study show the possibility to calibrate the CPD parameters. The availability of a calibration material also for CPD parameters would guarantee a further improvement of standardization and their use as reportable diagnostic parameters.
1. Le Roux G, Vlad A, Eclache V et al. Int J Lab Hematol 2010;32:237-243
2. Furundarena JR, Araiz M, Uranga M et al. Int J Lab Hematol 2010;32:360-366
3. Park SH, Park CJ, Lee BR et al. Int J Lab Hematol 2014;37
Simon-Lopez:Sysmex Corporation: Consultancy, Honoraria. Buoro:Sysmex EMEA: Research Funding. Pacioni:DASIT Diagnostica SpA: Employment, Other: Scientific Manager of DASIT (Distributor of Sysmex in Italy). Seghezzi:Sysmex EMEA: Research Funding. Manenti:Sysmex EMEA: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal