Abstract
Introduction
Local radiation therapy (RT) is commonly employed to treat bone lesions and/or extramedullary manifestations of multiple myeloma (MM) or solitary bone plasmacytoma (SBP). Besides providing sufficient control of local tumor growth, radiation therapy may also contribute to broader anti-tumor immunity by generating off-site cellular immune responses. Here, we put a focus on tumor-associated antigens (TAAs) well known for their expression in MM such as Wilms' Tumor Protein 1 (WT1), New York esophageal squamous cell carcinoma 1 (NY-ESO-1), human telomerase reverse transcriptase (hTERT), mucin 1 (MUC1), and preferentially expressed antigen of melanoma (PRAME) and assessed CD8-mediated immune responses before and during radiation.
Methods
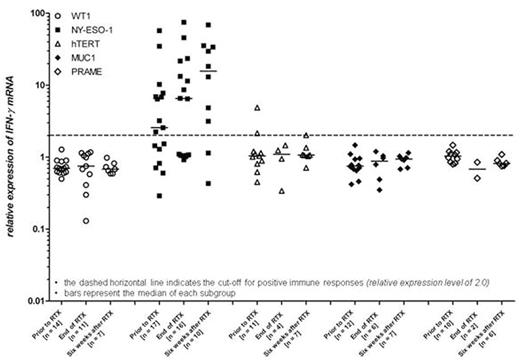
We prospectively recruited HLA-A*02:01-positive patients with multiple myeloma or solitary plasmacytoma requiring local radiation therapy for bone and extramedullary lesions in this scientific, non-interventional study, supported by a grant of the Wilhelm Sander Foundation, Germany, and approved by our local ethical committee. All patients provided written informed consent. Immune responses against HLA-A*02:01-restricted peptides of WT1, NY-ESO-1, hTERT, MUC1, and PRAME were assessed at three different time points: prior to RT, end of RT and six to eight weeks after RT. CD8+ T lymphocytes were isolated from peripheral blood and stimulated with irradiated, peptide-pulsed T2 cells (174 x CEM.T2; ATCC® CRL-1992™). After extraction of total RNA, Interferon gamma (IFNγ) mRNA expression was analyzed by RT-qPCR. IFNγ mRNA levels were normalized to CD8 mRNA levels. IFNγ mRNA expression of cells stimulated with the irrelevant melanoma antigen Glycoprotein 100 (gp100) served as calibrator. An expression level of 2.0 or more as compared to gp100 was considered as a positive result. Furthermore, an intracellular cytokine staining assay to detect IFNγ release in peptide-stimulated cells was employed to confirm PCR results on a protein level. Peptide-pulsed T2 cells also served as targets in CD107a degranulation assays.
Results
To date, 19 patients including 16 MM and three SBP patients received fractionated local RT with or without concomitant systemic therapy. 13 patients were previously untreated; three patients received prior allogeneic and five prior autologous hematopoietic stem cell transplantation. At baseline, positive immune responses were frequently detected against NY-ESO-1 (10 out of 17 assessed patients; 58.8%) but interestingly not against hTERT, WT1, MUC1 or PRAME (see Figure). Consequently, baseline immune responses against NY-ESO-1 were significantly higher (p<0.001) as compared to all other TAAs investigated (Kruskal Wallis test). These median response levels against NY-ESO-1 further increased in the course of radiation therapy and were confirmed by flow cytometry on a protein level. The functionality and specificity of these responses could be demonstrated in degranulation assays suggesting the ability to deliver efficient anti-tumor immunity.
Conclusions
Immunity against NY-ESO-1 was preexisting in the majority of patients and was undergoing marked expansion under local radiotherapy. This phenomenon was not found for any other TAA investigated suggesting that NY-ESO-1 carries a unique immunogenicity justifying most recent attempts to employ NY-ESO-1 as a target for genetically engineered T lymphocytes. Finally, this tumor-specific immunity is likely to contribute to immunosurveillance of multiple myeloma additionally supported by immune modulatory drugs applied for treatment such as IMiDs and proteasome inhibitors.
Knop:Takeda: Consultancy. Einsele:Celgene: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Speakers Bureau. Mielke:MSD: Consultancy, Other: Travel grants; Gilead: Other: Travel grants; Novartis: Consultancy; Celgene: Other: Travel grants, Speakers Bureau; JAZZ Pharma: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal