Abstract
Background:
In CLL/SLL, the iwCLL PR category is heterogeneous and complex to evaluate. Changes in clinical staging (Binet, Rai) have been proposed as a method to evaluate response in CLL (Montserrat, Cancer 1985; iwCLL, Ann Intern Med 1989). Ibrutinib (ibr), a first-in-class, once-daily inhibitor of Bruton's tyrosine kinase, is indicated by the FDA for the treatment of pts with CLL/SLL and allows for treatment without chemotherapy. Robust improvements in progression-free survival (PFS) and overall survival (OS) among ibr-treated pts in the phase 3 RESONATE trial demonstrated superiority of ibr over ofatumumab (ofa) in heavily pretreated CLL, regardless of prognostic features. As the majority of ibr pts achieved iwCLL PR or PR-L manifested by improved hematologic function and/or disease burden, we analyzed RESONATE pts with PR or PR-L by Binet stage to determine if clinical downstaging captures different, clinically meaningful subgroups among patients in iwCLL PR/PR-L.
Methods:
Among 391 previously treated CLL pts randomized to receive oral ibr (n=195) or intravenous ofa (n=196), those who achieved a best overall response of PR or PR-L were assessed for Binet Stage at time of first response. Stage A was defined as no anemia or thrombocytopenia and <3/5 sites of involvement (unilateral or bilateral cervical, axillary, and inguinal lymph nodes, liver, spleen); Stage B as no anemia or thrombocytopenia and ≥3/5 involved sites; and Stage C as anemia (hemoglobin <10 g/dL) and/or thrombocytopenia (platelets <100,000/µL) regardless of lymph node/organ site involvement. Binet stage data were considered missing if any one component of the staging criteria was not available. Kaplan-Meier analyses were performed based on Binet Stage at time of first response. Data were analyzed as of the interim analysis (Byrd, N Engl J Med 2014), and p-values are descriptive.
Results:
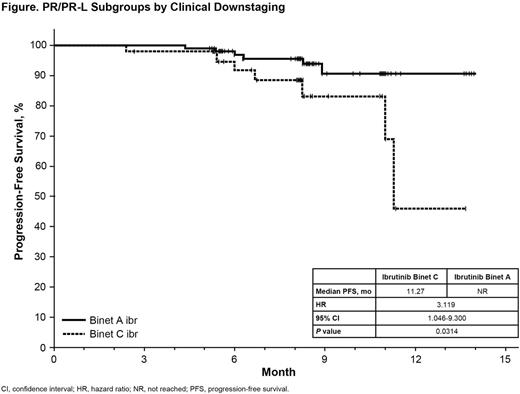
A total of 162 (83%) ibr pts and 45 (23%) ofa pts achieved PR/PR-L as best overall response as assessed by investigator; 53% of ibr responders were PR and 46% were PR-L at first response. Baseline characteristics (age, prior therapy, marrow infiltration, unmutated IGHV) were balanced between arms. A majority of PR/PR-L ibr pts were downstaged to Stage A reflecting improvement in lymphadenopathy/organomegaly and/or cytopenias with treatment. At time of first PR/PR-L, the proportion of Stage C pts decreased from 44% (n=72) at baseline to 27% (n=44) for the ibr arm and from 42% (n=19) to 33% (n=15) for the ofa arm. While maintaining PR/PR-L, 48% of ibr-treated and 40% of ofa-treated pts shifted from Stage C to A (or to B/A). The efficacy of ibr over ofa in PR/PR-L pts was similar to that observed in the intent-to-treat population for median PFS (not reached [NR] for ibr vs. 8.48 mo for ofa, P<0.0001). A treatment effect between ibr vs. ofa was also observed for duration of response (DOR) in PR/PR-L pts (NR for ibr vs. 5.52 mo for ofa, P<0.0001). Responders with PR/Stage A showed a longer PFS outcome than responders with PR/Stage C at time of first response to ibr (P=0.0314, Figure). The estimated median PFS for Stage C should be interpreted with caution as it occurred at the tail end of the curve with 2 pts at risk. Within the ofa arm, no difference in PFS between PR/Stage A vs. PR/Stage C was observed, although a limited sample size of responders prohibits robust analysis of this arm. For the ibr arm, both OS (P=0.0281) and DOR (P=0.0431) suggest a trend for improved outcome for PR/Stage A vs. PR/Stage C. Subset analysis (e.g., pts with del17p; PR vs PR-L outcomes) were not feasible because of the small number of pts in these categories.
Conclusions:
This ad-hoc analysis shows that changes in clinical stage break down the PR/PR-L (iwCLL) category into different, clinically meaningful subgroups, and reinforces the notion that improving cytopenias is a desirable goal of CLL therapy with ibrutinib (Barrientos, ASH 2014). Altogether, this study demonstrates that changes in clinical stage could be a useful and non-costly method to evaluate treatment response at different time points over the course of the disease, a concept with potential clinical implications that requires validation in prospective clinical trials.
Delgado:Janssen: Consultancy, Honoraria; Novartis/GSK: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Roche: Consultancy, Honoraria, Research Funding; Infinity: Research Funding. Suzuki:AbbVie: Equity Ownership; Pharmacyclics, LLC, an Abbie Company: Employment, Other: Leadership, travel, accommodations, expenses. Hsu:AbbVie: Equity Ownership; Pharmacyclics, LLC, an Abbie Company: Employment. James:Pharmacyclics, LLC, an AbbVie Company: Employment; AbbVie: Equity Ownership. Montserrat:Vivia Biotech: Equity Ownership; Pharmacyclics: Consultancy; Janssen: Honoraria, Other: travel, accommodations, expenses; Morphosys: Other: Expert Testimony; Gilead: Consultancy, Other: Expert Testimony.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal