Abstract
Background: The natural history of CLL/small lymphocytic lymphoma (SLL) treated with chemo-immunotherapy included enrichment for del(17p) with each progression and evolution to Richter transformation (RT) in some patients (pts) with refractory disease.In early phase clinical trials, the BCL2 inhibitor venetoclax achieves objective responses in ~80% of pts with heavily pre-treated relapsed/refractory CLL/SLL, irrespective of disease bulk, chemo-refractoriness or del(17p)/TP53 aberrations. However, the nature of progressions during ongoing therapy has not yet been well characterised. We report the clinicopathological features, outcomes and dominant predictors of progression on venetoclax for CLL/SLL.
Methods: We retrospectively reviewed data from 67 pts treated with venetoclax for relapsed/refractory CLL/SLL at two institutions in Australia between June 2011 and March 2016. Pts were enrolled in 1 of 3 ongoing trials: Phase 1 venetoclax monotherapy (NCT01328626), Phase 1b venetoclax + rituximab (NCT01682616), or Phase 2 venetoclax monotherapy in del(17p) CLL (NCT01889186). Pts received 150Ð1200mg/day of venetoclax ± 6 doses of rituximab (n=16).Forty-nine received the approved dose of 400mg/day or higher.Pts were investigated for RT at progression with PET scans and biopsies. There was no mandated systematic screening for RT at trial entry. Univariate Kaplan Meier, Cox proportional hazards multivariate and Classification and Regression Tree (CART) analyses were used to identify risk factors for progression.
Results: Median age was 68 (range 20Ð87) years; pts had received a median 3 (1Ð12) prior therapies. CLL/SLL was fludarabine refractory (F-refr; defined as no response or progression within 6 months) in 51%. With median follow up of 23 (2Ð46) months, 25 pts (37%) had progressed; 17 (68%) with RT (14 DLBCL, 3 Hodgkin-like) and 8 (32%) with CLL/SLL. Median time-to-progression (TTP) was 8 (1Ð23) months for RT and 23 (7Ð38) months for CLL/SLL (p = 0.0033). PET scans were performed in 12/17 cases of RT. High FDG-avidity disease (SUVmax > 10) was multifocal in 9 cases (median 4 sites (2 Ð 10)),unifocal in 2 and negative in 1pt with histologically confirmed DLBCL RT. All 13 cases of DLBCL RT tested for BCL2 protein expression by IHC were positive.
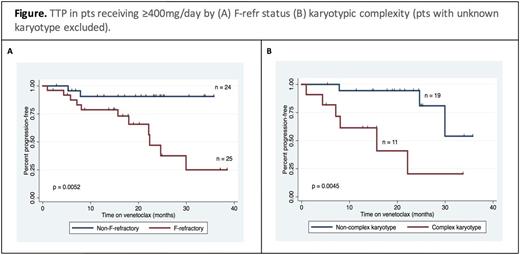
TTP was closely related to best iwCLL response (median: not reached, 25 and 6 months for CR, PR and SD, respectively; p<0.0001). Univariate analysis examined putative factors associated with modulation of risk of progression in pts treated at _400mg/day (n=49) including: age, number of prior therapies, disease bulk (>5 cm), presence of del(17p), presence of del(17p) and/or TP53 mutation, del(11q), and concurrent rituximab therapy. None were statistically significant (p>0.1). F-refr disease and complex karyotype (defined as ³3 cytogenetic abnormalities on conventional karyotype) were associated with risk of progression by univariate analysis (HR 6.1, p=0.0052;and HR 6.6, p=0.0045, respectively; see Figure). A limited power multivariate and CART analysis supported independence between these variables.
Median overall survival after progression was 11.4 months (32% at 2-years). Salvage chemotherapy was used in 16/17 pts with RT, followed byautograftsor allografts in 2 cases each. Seven pts with RT remain alive (response to salvage: 5 CR, 2 PR), including all 3 pts with Hodgkin-like RT (22, 23 and 43 months post progression). Three pts with DLBCL RT who responded to salvage (2 CR, 1 PR) subsequently progressed with CLL/SLL and remain alive on BTK inhibitors (BTKIs) at 30, 34 and 38 months. Six of 8 pts with progressive CLL/SLL onvenetoclaxwere treated withibrutinib(5 PR, 1 SD) and 3 remain alive on therapy at last follow up (6, 6 and 9 months).
Conclusions: F-refractoriness and complex karyotype are the dominant risk factors for progression onvenetoclax, which may presentearly as RT, as might be expected in heavily pretreated patients with these risk factors, or later as CLL/SLL, with PET features being discriminatory. A minority of pts with RT progression can attain durable disease control with multimodality therapy and progressive CLL/SLL can respond to BTKIs, even if after RT. Patients with CLL/SLL that isF-refr or has complex karyotype should have clinically occult RTexcluded before treatment with venetoclax monotherapy.
Anderson:Walter and Eliza Hall Institute of Medical Research: Other: Walter and Eliza Hall Institute of Medical Research receives milestone payments for the development of venetoclax. Tam:janssen: Honoraria, Research Funding; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees. Huang:Walter and Eliza Hall Institute of Medical Research: Other: Walter and Eliza Hall Institute of Medical Research receives milestone payments for the development of venetoclax. Seymour:Genentech: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie Inc.: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees. Roberts:Walter and Eliza Hall Institute of Medical Research: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal